Research Articles
Analytical Techniques for Measuring Hormone Bioavailability: From Formulation Challenges to Regulatory Validation
This article provides a comprehensive overview of modern analytical techniques for assessing hormone bioavailability across diverse formulations.
Standardized Protocols for Compounded Bioidentical HRT: Bridging the Evidence Gap in Regulatory Science and Clinical Practice
This article addresses the critical lack of standardized protocols for compounded bioidentical hormone replacement therapy (cBHT), a significant challenge in regulatory science and pharmaceutical development.
Hormone Replacement Therapy in Premature Ovarian Insufficiency: A Comprehensive Review of Mechanisms, Clinical Management, and Future Research Directions
Premature ovarian insufficiency (POI), a condition of ovarian failure before age 40 with a recently updated prevalence of 3.5%, presents profound clinical challenges extending beyond infertility to encompass significant risks...
2025 Clinical Guidelines for Initiating Menopausal Hormone Therapy: Evidence-Based Protocols for Researchers and Developers
This article provides a comprehensive analysis of contemporary clinical guidelines for initiating menopausal hormone therapy (MHT), synthesizing recent regulatory updates, evidence-based protocols, and emerging research.
Serum Hormone Monitoring in HRT: Analytical Methods, Clinical Applications, and Research Frontiers
This article provides a comprehensive resource for researchers, scientists, and drug development professionals on serum hormone monitoring during Hormone Replacement Therapy (HRT).
Optimizing Progesterone in HRT: From Molecular Mechanisms to Advanced Clinical Protocols for Researchers
This article provides a comprehensive analysis of progesterone supplementation within Hormone Replacement Therapy (HRT), tailored for researchers, scientists, and drug development professionals.
Advanced Methodologies for Endometrial Thickness Assessment in HRT Cycles: From Clinical Protocols to AI Innovations
This article provides a comprehensive analysis of endometrial thickness (ET) assessment methodologies within Hormone Replacement Therapy (HRT) cycles, tailored for researchers, scientists, and drug development professionals.
HRT Protocols in Frozen Embryo Transfer: A 2025 Review of Efficacy, Safety, and Future Directions for Clinical Research
This article provides a comprehensive analysis of Hormone Replacement Therapy (HRT) protocols for Frozen Embryo Transfer (FET), tailored for researchers and drug development professionals.
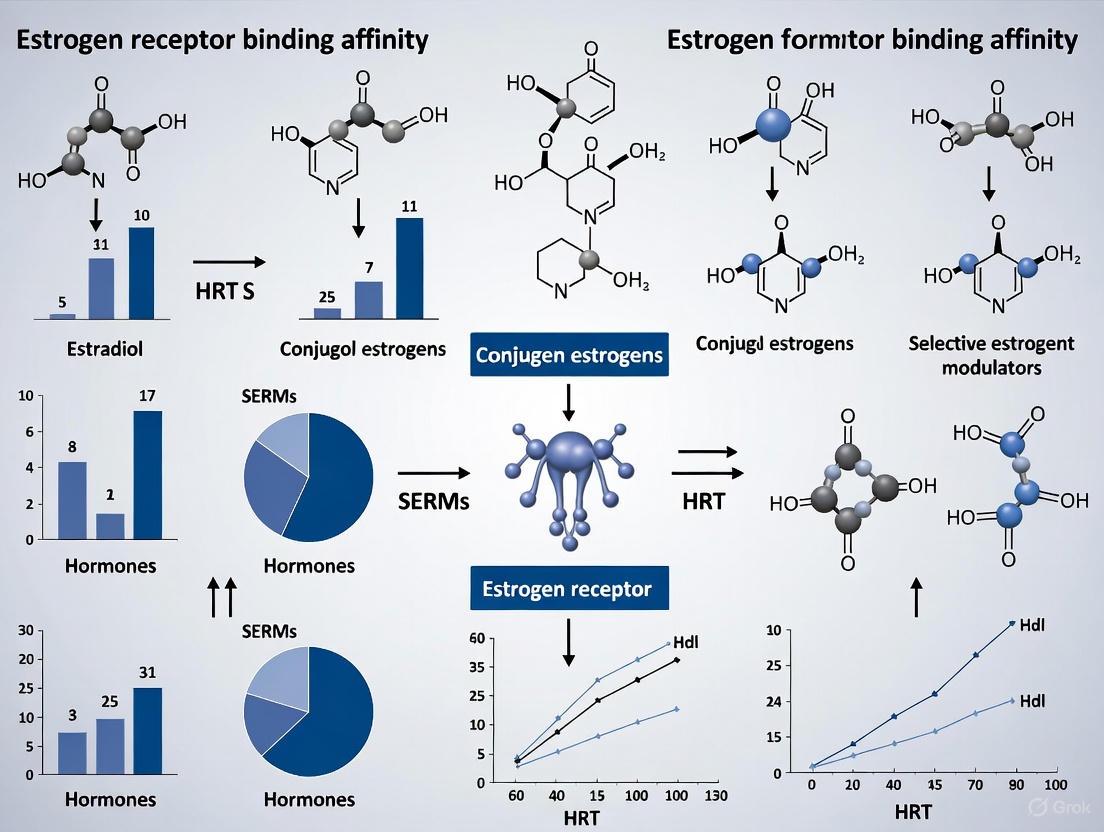
Estrogen Receptor Binding Affinity: A Critical Evaluation of HRT Formulations for Research and Development
This article provides a comprehensive analysis of estrogen receptor (ER) binding affinity across hormone replacement therapy (HRT) formulations, tailored for researchers and drug development professionals.
Progestogen Type and Breast Cancer Risk: Molecular Mechanisms, Clinical Evidence, and Therapeutic Implications
This article provides a comprehensive analysis for researchers and drug development professionals on the critical impact of progestogen type on breast cancer risk.