Research Articles
The Placenta's Secret: How Pregnancy Rewires Drug Metabolism
Exploring how pregnancy dramatically alters drug metabolism through changes in phase II enzymes and transporters, with implications for medication safety.
The Hunger Gatekeeper: How Ghrelin Knocks on Your Brain's Door
Explore how ghrelin, the hunger hormone, navigates the brain's strict barriers to stimulate appetite and its implications for health and disease.
The Stress Paradox: How Madagascar's Red-Bellied Lemurs Reveal the Hidden Costs of Forest Disturbance
Research on red-bellied lemurs in Madagascar reveals surprising findings about stress hormone levels in disturbed vs. undisturbed forests.
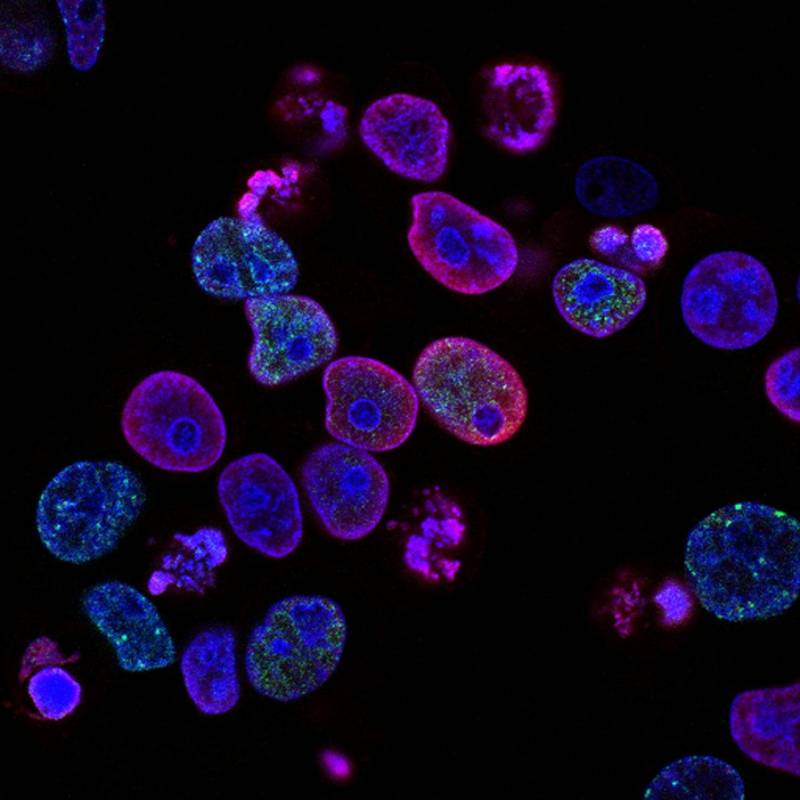
Beyond the Microscope: How Gene Profiling is Revolutionizing Breast Cancer Treatment
How gene profiling is revolutionizing breast cancer treatment with focus on PAM50 Risk of Recurrence Assay
The Great Thyroid Relay: How the Placenta Fuels Fetal Brain Development
Exploring how the placenta regulates thyroid hormone delivery to support fetal brain development and adapts in complicated pregnancies.
The Golden Seed Secret: How Melinjo's Lipid Gel Brightens Skin Safely
Discover how Melinjo seed extract in lipid nanoparticle gels offers a safe, effective solution for skin brightening, backed by clinical research.
The Hidden Architects: Unlocking Rice Fertility Through Anther Wall Secrets
Discover how transcriptome analysis reveals the molecular mechanisms controlling rice fertility through anther wall development, with implications for global food security.
The Menopause Supplement Dilemma: Separating Hope from Hype in Herbal Remedies
Examining the scientific evidence behind popular menopause supplements and providing evidence-based recommendations for symptom management.
The PTEN Puzzle: How a Tiny Protein Influences PCOS and Fertility
Exploring how PTEN protein influences PCOS and fertility through rat studies
Dual Strike: How Blocking Two Hormone Receptors Could Revolutionize Prostate Cancer Treatment
Exploring the potential of simultaneous androgen and estrogen receptor blockade in prostate cancer treatment using natural compounds like carnosol.