Neuroendocrine Profiles Compared: Oral Contraceptives vs. Naturally Cycling Women in Biomedical Research
This article synthesizes current research on the distinct neuroendocrine profiles of oral contraceptive (OC) users and naturally cycling (NC) women, a critical consideration for drug development and clinical research.
Neuroendocrine Profiles Compared: Oral Contraceptives vs. Naturally Cycling Women in Biomedical Research
Abstract
This article synthesizes current research on the distinct neuroendocrine profiles of oral contraceptive (OC) users and naturally cycling (NC) women, a critical consideration for drug development and clinical research. It explores the foundational science of how synthetic hormones in OCs create a hypogonadal or mixed endocrine state, differing significantly from the dynamic hormonal fluctuations of the natural menstrual cycle. The content details methodological best practices for accounting for these profiles in study design, addresses common research challenges such as OC formulation variability and participant screening, and validates findings by comparing cognitive, affective, and neural outcomes between groups. Aimed at researchers and drug development professionals, this review provides a framework for incorporating female-specific endocrine status into rigorous biomedical science.
Endocrine Foundations: Modeling the Hypothalamic-Pituitary-Ovarian Axis in OC Users and Naturally Cycling Women
Oral contraceptives (OCs) represent one of the most significant pharmacologic interventions in human reproductive biology. Their mechanism of action centers on the strategic suppression of endogenous hormone production to prevent ovulation. Both combined oral contraceptives (COCs) and progestin-only pills (POPs) operate primarily through negative feedback on the hypothalamic-pituitary-gonadal (HPG) axis, albeit with differing emphases and efficacies [1]. Understanding these mechanisms is fundamental for researchers investigating endocrine disruption, drug development professionals designing next-generation formulations, and clinicians interpreting the physiologic impacts of these widely used medications. This guide provides a structured comparison of how these two contraceptive classes achieve their effects, with particular focus on their differential suppression of endogenous hormone production compared to naturally cycling hormone profiles.
Comparative Mechanisms of Action
The following table summarizes the core mechanisms by which COCs and POPs suppress endogenous hormone production and prevent pregnancy.
Table 1: Comparative Mechanisms of Combined and Progestin-Only Oral Contraceptives
| Feature | Combined Oral Contraceptives (COCs) | Progestin-Only Pills (POPs) |
|---|---|---|
| Primary Mechanism | Suppression of the hypothalamic-pituitary-ovarian (HPO) axis to prevent ovulation via synergistic action of estrogen and progestin [1] [2] | Multiple complementary mechanisms, with primary effect varying by formulation: cervical mucus thickening and variable ovulation suppression [3] [4] |
| Hypothalamic Effect | Suppresses gonadotropin-releasing hormone (GnRH) pulse frequency [1] [2] | Suppresses GnRH pulse frequency, though generally to a lesser extent than COCs [1] |
| Pituitary Effect | Marked suppression of Follicle-Stimulating Hormone (FSH) and Luteinizing Hormone (LH); prevents mid-cycle LH surge [1] [2] [5] | Variable suppression of FSH and LH; may not consistently prevent the LH surge in all users [3] |
| Ovarian Effect | Inhibits follicular development and prevents ovulation as a primary, consistent effect [1] [6] | Inhibits ovulation in a variable percentage of cycles (approximately 50-60%, depending on formulation) [3] |
| Cervical Mucus Effect | Progestin component thickens cervical mucus, impeding sperm penetration [1] [2] | Primary mechanism for many formulations; consistently thickens cervical mucus to create a sperm barrier [4] [7] |
| Endometrial Effect | Causes endometrial atrophy, creating an unfavorable environment for implantation [2] [8] | Alters endometrial lining and reduces glandular development, potentially hindering implantation [9] [4] |
| Typical Use Failure Rate | 9% per year [1] [2] [6] | Varies by formulation; typical use failure rates are similar to COCs (approximately 9%) [7] |
Visualizing the Endocrine Disruption Mechanisms
The following diagrams illustrate the distinct pathways through which COCs and POPs disrupt the natural menstrual cycle.
Combined Oral Contraceptive (COC) Action on the HPG Axis
Progestin-Only Pill (POP) Action on the HPG Axis
Experimental Protocols for Investigating OC Mechanisms
Research into the mechanisms of oral contraceptives requires specialized methodologies to quantify hormonal suppression and its physiological consequences.
Protocol for Assessing Hormonal Suppression
Objective: To quantitatively measure the suppression of endogenous FSH, LH, estradiol, and progesterone in users of COCs versus POPs compared to naturally cycling controls.
- Participant Recruitment: Recruit three matched cohorts: COC users (monophasic formulation), POP users (norethindrone or drospirenone), and naturally cycling women (confirmed ovulatory cycles). Exclude participants with endocrine disorders, recent hormonal medication use, or BMI >35 kg/m².
- Blood Sampling Protocol: For all participants, collect serial blood samples:
- COC Group: On days 1 (first active pill), 7, 14, and 21 of the packet.
- POP Group: On days 1, 7, 14, and 28 of the packet (continuous daily dosing).
- Control Group: On menstrual cycle days 3 (baseline), 7, 14 (peri-ovulatory), and 21 (mid-luteal).
- Hormone Assay: Process samples to obtain serum. Quantify hormone levels using electrochemiluminescence immunoassays (ECLIA) for FSH, LH, estradiol, and progesterone. Run all samples from a single participant in the same assay batch to minimize inter-assay variability.
- Ovarian Ultrasound: For the control group only, perform transvaginal ultrasonography on sampling days to track follicular development and confirm ovulation.
- Data Analysis: Compare mean hormone concentrations between groups at equivalent time points using ANOVA with post-hoc testing. The primary endpoint is the absence of an LH surge and low, stable estradiol/progesterone levels in OC groups, indicating effective suppression.
Protocol for Evaluating Cervical Mucus Changes
Objective: To objectively assess the anti-permeability effects of progestin on cervical mucus as a barrier to sperm penetration.
- Mucus Collection: Collect endocervical mucus from participants in each group (COC, POP, Control) using a specialized aspiration device during the estimated fertile window (control group) or a comparable cycle day.
- Post-Coital Test (PCT): A subset of participants can undergo an in vivo PCT, where mucus is examined microscopically 2-12 hours after unprotected intercourse to quantify the number of motile sperm per high-power field.
- Capillary Tube Test: Using an in vitro approach, place a sample of cervical mucus in a capillary tube and introduce a prepared sample of motile sperm at one end. Measure the distance traveled by the vanguard sperm after 30 minutes.
- Mucus Scoring: Use a standardized scoring system (e.g., Insler Score) to rate mucus quantity, ferning, spinnbarkeit (stretchability), and cellularity.
- Analysis: Compare PCT results, sperm penetration distances, and mucus scores between groups. Successful mechanism is indicated by scant, thick, cellular mucus with poor spinnbarkeit and negligible sperm motility or penetration in the COC and POP groups.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Investigating Oral Contraceptive Mechanisms
| Reagent / Material | Function in Research |
|---|---|
| Ethinyl Estradiol (EE) | The synthetic estrogen standard in most COCs; used in in vitro studies to model the estrogenic component of contraceptive action on cell lines [2] [7]. |
| Progestins (e.g., Levonorgestrel, Norethindrone, Drospirenone) | Synthetic progestogens for studying the progestin-specific effects in both COCs and POPs, including receptor binding affinity and gene regulation experiments [1] [7]. |
| Electrochemiluminescence Immunoassay (ECLIA) Kits | For the precise, high-throughput quantitative measurement of FSH, LH, estradiol, and progesterone in serum/plasma samples from clinical trials [10]. |
| GnRH Receptor Antagonists | Pharmacologic tools used in controlled experiments to dissect the specific role of GnRH suppression in the overall mechanism of action of OCs. |
| Human Pituitary Cell Cultures | An in vitro model system for directly testing the effects of OC hormones on FSH and LH synthesis and secretion, independent of hypothalamic input. |
| Cervical Mucus Simulants | Synthetic hydrogels with tunable properties used for standardized in vitro testing of sperm motility and penetration under conditions mimicking OC use. |
Combined and progestin-only oral contraceptives achieve their primary objective through the strategic suppression of endogenous reproductive hormones, but they employ distinct mechanistic pathways. COCs act as a powerful, centralized suppressant of the HPG axis, consistently inhibiting ovulation through the synergistic action of estrogen and progestin. In contrast, POPs employ a more nuanced, multi-faceted approach, where the primary mechanism can be a potent local effect (cervical mucus thickening) supported by variable degrees of central suppression. For researchers, these differences are not merely academic; they inform the design of clinical trials, the interpretation of endocrine profiles in study participants, and the development of novel compounds with improved side-effect profiles. The choice of experimental model and endpoint measurement is critically dependent on which specific mechanism is under investigation, whether it be the suppression of the LH surge, the alteration of cervical mucus biophysics, or the creation of an unreceptive endometrium.
Oral contraceptives (OCs) represent one of the most widely used pharmacological interventions globally, with approximately 150 million women using them worldwide [11]. Despite six decades of clinical use, the neurobiological mechanisms underpinning their effects on the brain remain incompletely characterized. Two primary conceptual frameworks have emerged to explain how OCs influence brain structure and function: the hypogonadal state model, which emphasizes the suppression of endogenous hormonal fluctuations, and the hyperprogestogenic state model, which focuses on the direct effects of synthetic progestins on the central nervous system [12] [13]. This review systematically compares these competing models by synthesizing current preclinical and clinical evidence, with particular emphasis on neuroimaging findings, neuroactive steroid concentrations, and emotional and cognitive outcomes. Understanding the distinct pathways through which OCs exert their cerebral effects is crucial for researchers and drug development professionals aiming to develop next-generation contraceptives with optimized neurological side-effect profiles.
Core Conceptual Models: Mechanisms and Pathways
The Hypogonadal State Model
The hypogonadal state model posits that the primary mechanism of OC action on the brain is through the suppression of the hypothalamic-pituitary-gonadal (HPG) axis. Combined oral contraceptives (COCs) suppress the release of gonadotropin-releasing hormone (GnRH) from the hypothalamus, which subsequently inhibits the pulsatile secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the pituitary gland [12] [14]. This suppression prevents ovulation and dramatically reduces the production of endogenous ovarian hormones, including estradiol and progesterone [12].
The resulting endocrine environment is characterized by abolished cyclical fluctuations of endogenous sex steroids and their replacement with constant, low levels of synthetic hormones [11]. This model conceptualizes OC users as being in a functional state of central hypogonadism, similar to other conditions where GnRH neuronal activity is impaired [14]. The brain, particularly regions rich in steroid hormone receptors such as the hippocampus, amygdala, and prefrontal cortex, is consequently exposed to an attenuated, non-cyclical hormonal milieu [11] [15].
The Hyperprogestogenic State Model
In contrast, the hyperprogestogenic state model emphasizes the direct neurological impact of the synthetic progestins contained within OCs. This model highlights that progestins are not a homogeneous drug class but rather comprise compounds with diverse pharmacological profiles, including varying affinities for progesterone receptors, androgen receptors, mineralocorticoid receptors, and glucocorticoid receptors [12] [13].
The model proposes that the brain is exposed to significantly different hormonal environments depending on the specific progestin type, dose, and regimen [13]. For instance, levonorgestrel (LNG) exhibits strong androgenic properties and binds with high affinity to progesterone, androgen, and mineralocorticoid receptors [12], whereas drospirenone has anti-androgenic activity and spironolactone-like effects [12] [13]. These differential receptor interactions trigger distinct downstream neurobiological effects, including alterations in neurosteroid synthesis (particularly allopregnanolone), GABAergic and serotonergic neurotransmission, and ultimately, brain network organization and function [13].
Table 1: Core Characteristics of Conceptual Models for OC Effects on the Brain
| Feature | Hypogonadal State Model | Hyperprogestogenic State Model |
|---|---|---|
| Primary Mechanism | Suppression of HPG axis and endogenous hormone cyclicity [12] [14] | Direct action of synthetic progestins on neural receptors and pathways [12] [13] |
| Key Hormonal Changes | ↓ Endogenous Estradiol, ↓ Progesterone, abolished cyclicity [12] [11] | ↑ Potent synthetic progestins; variable androgenic/anti-androgenic activity [12] [13] |
| Theoretical OC State | Functional central hypogonadism [14] | Pharmacologically-induced progestin dominance [13] |
| Primary Brain Targets | Hypothalamus, pituitary; brain regions with high steroid receptor density (e.g., hippocampus) [12] [11] | Distributed neural systems based on progestin receptor binding and neurosteroid conversion [12] [13] |
The following diagram illustrates the distinct neuroendocrine pathways proposed by these two models:
Neuroimaging Evidence for Distinct Neural Effects
Brain Structure and Functional Connectivity
Neuroimaging studies provide compelling evidence for OC-induced alterations in brain organization, with patterns that may reflect contributions from both conceptual models.
Structural Changes: Studies have documented modified volumes in specific brain regions between OC users and naturally cycling women, including areas within the prefrontal cortex and hippocampus [12]. Furthermore, increased mean diffusivity in the fornix, a key white matter tract, has been observed in OC users compared to non-users, suggesting microstructural changes [12].
Functional Connectivity: A randomized, double-blind, placebo-controlled crossover study investigating effects of OCs on brain networks revealed widespread network-level changes, particularly within subcortical, executive, and somatomotor circuits [16]. This study also found that OCs increased between-subject similarity in functional connectomes, suggesting a loss of individual neurological "idiosyncrasy" during use [16]. The specific networks affected included the default mode, executive, limbic, salience, somatomotor, and subcortical networks [16].
A rigorous single-subject serial imaging study conducted over two complete cycles—one natural and one on COCs—found that modularity, system segregation, and characteristic path length were statistically significantly higher across the natural cycle [17]. This suggests a more network-structured brain connectivity architecture during the natural cycle, whereas OC use was associated with a generally increased connectivity structure evidenced by lower characteristic path length, potentially reflecting a less optimized or specialized neural organization [17].
Table 2: Key Neuroimaging Findings in OC Users Compared to Naturally Cycling Women
| Brain Metric | Key Findings in OC Users | Potential Model Alignment |
|---|---|---|
| Regional Brain Volume | Altered volumes in prefrontal regions, hippocampus [12] | Both Models |
| White Matter Microstructure | Increased mean diffusivity in the fornix [12] | Hypogonadal |
| Functional Network Organization | ↓ Modularity, ↓ System Segregation, ↓ Characteristic Path Length [17] | Hypogonadal |
| Between-Subject Similarity | ↑ Similarity in functional connectomes (loss of idiosyncrasy) [16] | Hypogonadal |
| Specific Network Alterations | Changes in default mode, executive, limbic, salience, somatomotor, subcortical networks [16] | Both Models |
Methodological Protocols in Key Neuroimaging Studies
Randomized Controlled Trial Protocol (Citation 5):
- Design: Double-blind, placebo-controlled crossover.
- Participants: Healthy females of reproductive age.
- Intervention: Active combined OCPs vs. matched placebo.
- MRI Acquisition: Resting-state functional MRI (rs-fMRI) for functional connectivity.
- Analysis:
- Seed-based connectivity analysis targeting amygdala, putamen, and dorsal anterior cingulate.
- Functional connectome fingerprinting (a multivariate, data-driven method) to identify whole-brain patterns distinguishing individuals.
- Calculation of between-subject similarity (Iother) in functional connectomes.
- Correlation of connectivity changes with Daily Record of Severity of Problems (DRSP) symptom scores to link neural changes with mood.
Serial Single-Subject Study Protocol (Citation 7):
- Design: Intensive longitudinal case study; the "28andMe" project.
- Participant: A single healthy 23-year-old female.
- Phases:
- Natural Cycle: rs-fMRI daily for 30 consecutive days during a naturally occurring menstrual cycle.
- OC Cycle: The same protocol repeated 12 months later while using a monophasic COC (20 μg ethinylestradiol + 100 μg levonorgestrel).
- Hormone Monitoring: Daily blood measurements for hormone levels.
- Analysis:
- Graph Theory Metrics: Calculation of modularity, system segregation, and characteristic path length from rs-fMRI data.
- Dynamic Functional Connectivity (dFC): Using Leading Eigenvector Dynamic Analysis (LEiDA) to identify and track discrete, time-varying brain states.
The experimental workflow for a comprehensive neuroimaging study integrating these elements is depicted below:
Neurobiological Mechanisms and Signaling Pathways
Neuroendocrine and Neurotransmitter Systems
Preclinical and clinical studies reveal that OCs exert complex effects on multiple neurobiological systems, with mechanisms aligning with both conceptual models.
Neuroactive Steroids: HC use is associated with significantly altered concentrations of neuroactive steroids in the brain. Preclinical studies in rats show that chronic administration of ethinyl estradiol (EE) and levonorgestrel (LNG) decreases the concentrations of pregnenolone, progesterone, and its metabolite allopregnanolone in the frontal cortex and hippocampus [12]. Allopregnanolone is a potent positive allosteric modulator of GABA-A receptors, and its reduction has significant implications for mood and anxiety regulation [13].
Neurotransmitter Systems: OC effects on key neurotransmitter systems include:
- GABAergic System: Reductions in cortical allopregnanolone suggest a downstream dampening of GABAergic inhibition, which may contribute to affective symptoms reported by some users [12] [13].
- Serotonergic System: Estrogens are known to increase serotonergic activity, and thus suppression of endogenous estradiol via the hypogonadal state may reduce serotonin signaling, potentially influencing mood and emotion processing [15].
- Glutamatergic System: Animal studies indicate that EE increases the expression of glutamate decarboxylase (GAD) in the perirhinal cortex, suggesting enhanced conversion of glutamate to GABA and a shift in excitatory-inhibitory balance [11].
Stress Response System: OC use is associated with a blunted cortisol response to stress, which may alter the vulnerability of users to mood disorders [11]. This effect on the hypothalamic-pituitary-adrenal (HPA) axis may represent an interaction between the hypogonadal state and synthetic hormone administration.
The following diagram synthesizes the key neurobiological pathways affected by OC use:
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Investigating OC Effects on the Brain
| Reagent/Material | Function/Application | Example Use in Field |
|---|---|---|
| Combined OC Formulations | Create specific hormonal milieus for intervention studies; vary by progestin type and EE dose [12] [13] | Comparing androgenic vs. anti-androgenic progestins (e.g., LNG vs. drospirenone) [13] |
| Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) | High-specificity measurement of endogenous and synthetic hormones in saliva or serum [13] | Quantifying E2, EE, P4, testosterone, and progestins concurrently in participant samples [13] |
| Validated Mood & Cognitive Batteries | Standardized assessment of behavioral outcomes (e.g., DRSP, Beck Depression Inventory) [16] [18] | Correlating neural changes with negative affect, depression, and anxiety scores [11] [16] |
| High-Resolution MRI Scanner (3T+) | Acquisition of structural, functional (rs-fMRI), and diffusion-weighted images [16] [17] | Mapping brain volume, functional connectivity, and white matter microstructure [12] [16] |
| Graph Theory Analysis Pipelines | Quantification of global and nodal brain network properties from rs-fMRI data [17] | Calculating modularity, characteristic path length, and system segregation [17] |
| Dynamic Functional Connectivity Algorithms (e.g., LEiDA) | Identification of time-varying brain states and their dynamics during rest [17] | Tracking shifts in the prevalence of discrete brain states across cycles or conditions [17] |
Behavioral and Cognitive Correlates
Emotional and Mood Outcomes
The effects of OCs on mood and affective processing represent one of the most variable and clinically relevant outcomes, with evidence supporting both conceptual models.
Mood Disorders and Depression: Large epidemiological studies have found higher rates of depression in OC users, particularly in adolescents and during the first few months of use [19]. However, responses are highly individual, with some women experiencing improved and stabilized mood, while others report mood worsening and depressive symptoms [11] [19]. This variability suggests that individual differences in hormonal sensitivity may interact with both the hypogonadal state (e.g., removal of cyclical fluctuations) and the hyperprogestogenic state (e.g., specific progestin type) to determine affective outcomes [13].
Fear and Emotional Processing: A recent fear conditioning and extinction study found that both current and past COC users displayed greater fear return in safe contexts compared to non-users, indicating impaired fear regulation [13]. Exploratory analyses linked this impairment to higher ethinyl estradiol doses and specific progestins, providing support for the hyperprogestogenic model, where formulation-specific effects drive behavioral outcomes [13].
Dimensional Hormone Sensitivity Framework: The dimensional hormone sensitivity framework posits that individuals differ not only in whether they are behaviorally sensitive to hormones, but also to which specific hormonal changes they are sensitive (e.g., progesterone metabolite surges, estradiol withdrawal) [13]. This framework helps explain why some women with premenstrual dysphoric disorder (PMDD) benefit from the mood-stabilizing effects of certain OCs (particularly those with anti-androgenic activity), while others experience adverse mood effects [11] [13].
Cognitive Performance
Research on cognitive outcomes has yielded mixed results, though some patterns are emerging that may be formulation-dependent.
Spatial and Verbal Abilities: A systematic review concluded that the most consistent cognitive finding is a moderate increase in memory tasks, while results regarding spatial performance are inconclusive but may depend on the hormonal contraceptive formulation used [11]. Estrogenic potency appears to have a more substantial impact on spatial abilities than progestin androgenicity, with EE potentially diminishing spatial performance [11]. Effects on verbal fluency also show complex patterns, with some studies observing moderate increases during active pill intake, while others found a negative association with longer duration of OC use, suggesting differential effects of short- versus long-term use [11].
Table 4: Summary of Key Behavioral and Cognitive Findings Associated with OC Use
| Domain | Key Findings | Implications for Conceptual Models |
|---|---|---|
| Mood & Depression | Highly variable response; some experience improvement, others worsening, especially early use [11] [19]. | Supports interaction between individual sensitivity (Dimensional Framework) and both models [13]. |
| Fear Regulation | Impaired fear extinction recall and greater fear return in safe contexts [13]. | Hyperprogestogenic (formulation-dependent, linked to EE dose/progestin type) [13]. |
| Spatial Abilities | Inconclusive results, potentially modulated by estrogenic potency more than progestin androgenicity [11]. | Both Models (EE effect suggests hypogonadal; progestin effect suggests hyperprogestogenic). |
| Verbal Memory/Fluency | Moderate increases in some tasks; negative association with long-term use in others [11]. | Both Models (complex, timing-dependent effects). |
| Stress Response | Blunted cortisol response to stress [11]. | Hypogonadal (HPG-HPA axis interaction). |
The evidence synthesized in this review indicates that both the hypogonadal and hyperprogestogenic models provide valuable, non-mutually exclusive frameworks for understanding the effects of OCs on the brain. The hypogonadal model is strongly supported by neuroimaging evidence showing widespread alterations in functional network organization, including reduced modularity and loss of individual connectome idiosyncrasy, which likely reflect the profound suppression of endogenous hormonal cyclicity [16] [17]. In contrast, the hyperprogestogenic model effectively explains the formulation-dependent differences in outcomes, particularly regarding mood, fear regulation, and certain cognitive functions, which vary significantly with progestin type and dose [12] [13].
Future research should move beyond simply comparing OC users to non-users and instead adopt precision medicine approaches that account for the vast pharmacological heterogeneity of OCs and individual differences in hormonal sensitivity. Key priorities include head-to-head comparisons of formulations with distinct neurobiological profiles (e.g., differing effects on allopregnanolone), longitudinal studies capturing transition periods (initiation, withdrawal), and the integration of high-specificity hormone assays with multi-modal neuroimaging. By dissecting the relative contributions of endogenous hormone suppression versus synthetic hormone actions, researchers and drug developers can work toward a new generation of contraceptives that minimize adverse neurological effects while maintaining efficacy.
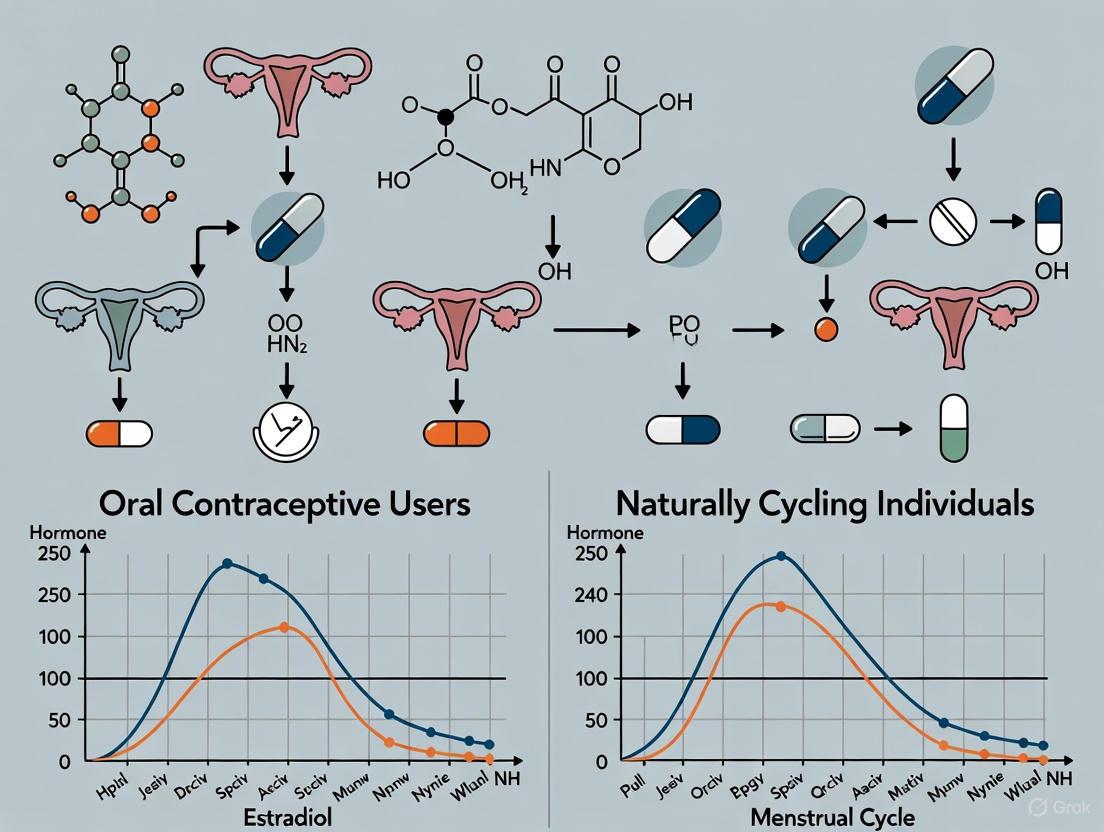
The human endocrine system regulates numerous physiological processes, with sex hormones playing a pivotal role in female reproductive health and beyond. Research contrasts two distinct hormonal profiles: the naturally cycling woman (NCW) experiencing rhythmic hormonal fluctuations, and the oral contraceptive (OC) user maintaining steady-state synthetic hormone levels [20]. This comparison is fundamental for understanding how these different hormonal environments affect physiological and cognitive outcomes, informing both clinical practice and pharmaceutical development.
In natural cycles, hormones fluctuate in a precise pattern throughout the menstrual cycle, driven by the hypothalamic-pituitary-ovarian (HPO) axis [21]. Conversely, combined oral contraceptives (COCs) typically contain synthetic estrogen (commonly ethinylestradiol) and a synthetic progestin, which suppress the HPO axis, leading to low, stable levels of these synthetic hormones and the suppression of endogenous hormonal fluctuations [20] [21]. This article provides a comparative analysis of these two states, supported by experimental data and visualization of the underlying physiological mechanisms.
Physiological and Cognitive Comparisons
Experimental Data on Cognitive and Physiological Outcomes
Table 1: Comparative Cognitive Performance in Natural Cycling vs. Oral Contraceptive Users
| Cognitive Domain | Natural Cycling (NCW) | Oral Contraceptive (OC) Users | Research Findings |
|---|---|---|---|
| Topographic Memory (Learning) | Variable performance across cycle phases [20] | Consistently better performance in learning 8-step path [20] | OC users showed an advantage in topographic learning compared to NCW [20] |
| Topographic Memory (Delayed Recall) | No significant difference between groups [20] | No significant difference between groups [20] | Both groups performed similarly in recalling a path after a 5-minute delay [20] |
| Verbal Memory | Slightly lower overall recall trend [22] | Slightly better recall trend for short-term & long-term tasks [22] | No significant negative impact of COCs; a trend suggesting slightly better recall in users [22] |
| Psychological Well-being Variability | Higher day-to-day variability in agitation, risk-taking, attractiveness, energy [23] | Lowered variability across several well-being domains [23] | OC use associated with "emotional blunting" or stabilized well-being markers [23] |
| Psychological Well-being Levels | Higher happiness, attractiveness, risk-taking, energy ratings [23] | Lower ratings on some well-being domains [23] | OC users reported more relaxation, sexual desire, and better sleep quality [23] |
| Endometrial Thickness (in FET) | Significantly thicker endometria [24] | Thinner endometria [24] | Natural cycle protocol yielded thicker endometrial linings [24] |
| Obstetric Outcome (in FET) | Lower risk of caesarean section [24] | Higher risk of caesarean section [24] | HRT cycle associated with an increased risk of caesarean section [24] |
Detailed Experimental Protocols
To ensure reproducibility and critical appraisal, this section outlines the methodologies from key studies cited in the comparison tables.
2.2.1 Protocol: Topographic Memory Assessment (Walking Corsi Test) [20]
- Objective: To investigate the effects of endogenous and exogenous hormonal fluctuations on topographic memory.
- Participants: 51 healthy women (25 NCW and 26 OC users). NCW were tested in the early follicular phase (days 4-5) and the mid-luteal phase (days 20-21). OC users were tested during the active pill phase (days 20-21) and the inactive pill phase (days 2-4).
- Tool: The Walking Corsi Test (WalCT), which assesses the ability to learn and recall a pathway in a real-world setting.
- Procedure:
- Participants were instructed to learn an eight-step sequence path.
- After learning the path, recall was tested immediately.
- Delayed recall was tested five minutes later.
- Analysis: Performance was compared between the two groups (NCW vs. OC) and, for NCW, between the two menstrual cycle phases.
2.2.2 Protocol: Verbal Affective Memory Task (VAMT) [22]
- Objective: To investigate the effect of COC use on verbal memory and affective bias in a large sample of healthy women.
- Participants: 205 healthy premenopausal women (90 COC users and 115 non-users) from the CIMBI database.
- Tool: The Verbal Affective Memory Task (VAMT). Data were pooled from 24-word and 26-word versions, which include positive, negative, and neutral words.
- Procedure:
- Participants were presented with the word list.
- Memory was assessed across three conditions: immediate learning, short-term recall, and long-term recall.
- Primary Outcome: The percentage of correctly recalled words (Total Word Recall-%) across all valence categories and conditions.
- Secondary Outcome: Affective memory bias, calculated as the percentage of correctly recalled positive words minus the percentage of correctly recalled negative words.
- Analysis: Multiple linear regression was used to determine group differences between COC users and non-users.
2.2.3 Protocol: Frozen Embryo Transfer (FET) Outcomes [24]
- Objective: To compare pregnancy outcomes and perinatal obstetric complications between natural cycle and hormone replacement therapy (HRT) cycle protocols for endometrial preparation in frozen embryo transfer.
- Study Design: Secondary analysis of a multicenter randomized controlled trial.
- Participants: 908 patients who were transferred two cleavage-stage (day 2 or day 3) frozen embryos.
- Groups:
- Natural Cycle: Participants were monitored for ovulation via ultrasound. Luteal phase support began on the day of ovulation.
- HRT Cycle: Oral estradiol valerate (4-8 mg/day) was started on days 1-3 of the period. When endometrial thickness reached ≥7 mm, progesterone was added.
- Outcomes: Endometrial thickness, implantation rate, clinical pregnancy, live birth, and obstetric complications (e.g., caesarean section).
- Analysis: Statistical comparisons using Student's t-tests, Mann-Whitney U tests, and Chi-squared tests as appropriate.
Signaling Pathways and Hormonal Regulation
The Hypothalamic-Pituitary-Ovarian (HPO) Axis
The following diagram illustrates the key hormonal interactions in the natural menstrual cycle, which are suppressed by combined oral contraceptives.
Mechanism of Combined Oral Contraceptives
The following diagram shows how synthetic hormones in COCs interrupt the natural HPO axis to prevent ovulation.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents and Tools for Hormonal Cycle Research
| Reagent / Tool | Function in Research | Example Application |
|---|---|---|
| Salivary Hormone Kits | Measures bioavailable estradiol, progesterone, and testosterone levels non-invasively [23] | Tracking daily hormonal fluctuations across the menstrual cycle in longitudinal studies [23] |
| Walking Corsi Test (WalCT) | Assesses topographic memory and spatial navigation in a real-world setting [20] | Comparing learning and recall of pathways between NCW and OC users [20] |
| Verbal Affective Memory Task (VAMT) | Evaluates verbal memory and affective bias using valenced words [22] | Investigating immediate, short-term, and long-term verbal recall in different hormonal groups [22] |
| Radioimmunoassay (RIA) | Quantifies hormone levels in plasma or serum with high sensitivity [23] | Validating phase-specific hormone concentrations (e.g., low E2/P4 in follicular phase) [23] |
| Transvaginal Ultrasound | Measures endometrial thickness and tracks follicular development [24] | Monitoring endometrial preparation in natural vs. HRT cycles for frozen embryo transfer [24] |
| Profile of Mood States (POMS) | Self-report questionnaire assessing transient, distinct mood states [22] | Quantifying subclinical mood symptoms as a covariate in cognitive studies [22] |
The evidence demonstrates a clear trade-off between dynamic fluctuation and steady-state hormonal profiles. Natural cycling is characterized by a complex, rhythmic interplay of hormones that supports certain physiological endpoints like endometrial development but introduces variability in cognitive and psychological domains [24] [23]. In contrast, oral contraceptives create a stable, suppressed hormonal environment that can enhance performance in specific memory tasks like topographic learning and reduce emotional variability, albeit sometimes at the cost of lowered well-being in other areas and altered obstetric outcomes [20] [24] [23]. These findings underscore the importance of context—whether for contraceptive development, therapeutic intervention, or cognitive research—in evaluating the implications of these distinct hormonal landscapes. Future research should continue to elucidate the mechanisms behind these differences to better tailor interventions to individual needs.
The synthetic progestins contained in combined oral contraceptives (COCs) exhibit diverse pharmacological profiles, with their inherent androgenic activity representing a critical differentiator in clinical performance and research outcomes. Androgenicity refers to the capacity of a progestin to bind to and activate androgen receptors, producing effects similar to male sex hormones. This property stems primarily from the molecular structure of the progestin compound, with some derived from testosterone retaining significant androgenic potential, while others synthesized from progesterone or spironolactone demonstrate anti-androgenic properties [25]. The spectrum of progestin androgenicity creates distinct endocrine environments for oral contraceptive users compared to naturally cycling women, necessitating careful consideration in both clinical practice and research design [26] [25].
Understanding these differential androgenic profiles is essential for personalized contraceptive selection and for interpreting neuroscientific and endocrine research comparing hormonal contraceptive users with naturally cycling populations. This analysis systematically compares androgenic and anti-androgenic progestin formulations based on experimental data, detailing their mechanisms, clinical effects, and methodological considerations for research applications.
Progestin Classification and Pharmacological Profiles
Progestins in combined oral contraceptives are commonly categorized by generation and androgenicity, with significant clinical implications. Second-generation progestins like levonorgestrel (LNG) exhibit higher androgenic potential, while third-generation progestins (desogestrel, gestodene) and drospirenone (DRSP) demonstrate lower or anti-androgenic activity [27] [25]. The androgenic effect primarily occurs through direct binding to androgen receptors, with some progestins also potentially lowering sex hormone-binding globulin (SHBG), thereby increasing the bioavailability of free testosterone [25].
Table 1: Classification and Properties of Common Progestins
| Progestin | Generation | Androgenicity Profile | Molecular Derivative | Receptor Binding Affinity |
|---|---|---|---|---|
| Levonorgestrel (LNG) | Second | Moderately to Highly Androgenic | Testosterone | High progesterone receptor, moderate androgen receptor |
| Desogestrel (DSG) | Third | Low/Non-Androgenic | Testosterone | High progesterone receptor specificity |
| Gestodene (GSD) | Third | Low/Non-Androgenic | Testosterone | High progesterone receptor binding |
| Drospirenone (DRSP) | Fourth | Anti-Androgenic | Spironolactone | Progesterone receptor agonist, androgen receptor antagonist |
The structural basis for these differences lies in their molecular derivatives. Androgenic progestins typically retain the basic steroid nucleus of testosterone with minimal modifications that preserve androgen receptor affinity. In contrast, anti-androgenic progestins like drospirenone incorporate structural elements that allow them to bind androgen receptors without activating them, functioning as competitive antagonists [25]. This fundamental pharmacological difference translates to distinct clinical profiles and research implications.
Experimental Evidence: Comparative Performance Data
A 2025 network meta-analysis of 18 randomized controlled trials directly compared four progestins—gestodene (GSD), desogestrel (DSG), drospirenone (DRSP), and levonorgestrel (LNG)—across multiple efficacy and safety parameters [27]. The findings demonstrate how androgenicity influences clinical outcomes:
Table 2: Comparative Efficacy and Safety Profiles of Different Progestins [27]
| Progestin | Breakthrough Bleeding (OR) | Irregular Bleeding (OR) | Contraceptive Efficacy (SUCRA%) | Adverse Events (SUCRA%) | Key Clinical Strengths |
|---|---|---|---|---|---|
| Gestodene (GSD) | 0.41 (0.26, 0.66) | 0.67 (0.52, 0.86) | 48.1% | 22.1% | Superior bleeding control |
| Desogestrel (DSG) | 0.98 (0.75, 1.29) | 1.02 (0.81, 1.29) | 51.3% | 45.8% | Balanced efficacy/safety |
| Drospirenone (DRSP) | 1.21 (0.91, 1.61) | 1.15 (0.89, 1.49) | 49.8% | 66.9% | Minimizing androgenic effects |
| Levonorgestrel (LNG) | 1.32 (0.99, 1.76) | 1.24 (0.96, 1.60) | 41.8% | 55.2% | Emergency contraception |
This comprehensive analysis revealed that progestins with lower androgenic activity (GSD, DSG) demonstrated superior bleeding profiles, while the anti-androgenic DRSP showed the most favorable adverse event profile [27]. Androgenic progestins like LNG, while effective for emergency contraception, exhibited less favorable bleeding patterns and higher discontinuation rates in some studies.
Cognitive and Spatial Performance Differential
The androgenic properties of progestins significantly moderate the relationship between masculine self-perception and spatial skills. Research involving 141 naturally cycling women and 229 OC users revealed that the positive association between self-perceived masculinity and 3D mental rotations performance present in naturally cycling women was accentuated in users of OCs with relatively low estrogenic and high progestational activity [25]. This moderation effect underscores how exogenous hormonal profiles interact with psychological factors to influence cognitive outcomes.
Notably, the androgenic activity of progestins impacts brain structure and function differently. Neuroimaging studies have identified differential effects on gray matter volume in frontal and temporal regions between users of androgenic versus anti-androgenic OCs [28]. These structural differences may underlie observed cognitive variations and highlight the importance of considering progestin androgenicity in neuroscientific research.
Androgen Receptor Signaling Pathways: Mechanisms of Action
The differential effects of androgenic and anti-androgenic progestins occur primarily through modulation of the androgen receptor (AR) signaling pathway. Understanding this mechanism is fundamental to comprehending their distinct clinical profiles.
Figure 1: Androgen Receptor Signaling Modulation by Progestins. Androgenic progestins activate transcription, while anti-androgenic progestins competitively inhibit and recruit corepressors.
The diagram illustrates how androgenic progestins function as AR agonists, promoting the transcription of androgen-responsive genes, while anti-androgenic progestins act as competitive antagonists, blocking native androgen binding and potentially recruiting corepressors to suppress gene transcription [25]. This fundamental difference in molecular mechanism translates to the clinically observable effects.
Research Methodologies: Experimental Protocols for Androgenicity Assessment
Clinical Comparison Studies
The network meta-analysis methodology provides a robust framework for direct and indirect comparison of progestin effects [27]. Key experimental parameters include:
Population: Reproductive-aged women (typically 18-45) without contraindications to COCs. Sample sizes ranging from 55-740 participants per treatment arm across studies. Intervention: Monophasic COC formulations with standardized ethinyl estradiol doses (typically 20-30μg) combined with different progestins. Comparator: Active controls consisting of COCs with different progestins, allowing for direct comparison. Outcomes: Primary endpoints include breakthrough bleeding incidence, irregular bleeding rates, withdrawal bleeding patterns, and contraceptive efficacy (Pearl Index). Secondary endpoints encompass androgenic side effects (acne, hirsutism), metabolic parameters, and subjectively reported adverse events. Assessment Schedule: Cycle-controlled documentation over minimum 6-13 treatment cycles, with specific cycle days designated for outcome assessment to ensure standardization [27].
This methodology enables both pairwise and network meta-analysis, generating odds ratios with 95% confidence intervals for dichotomous outcomes and standardized mean differences for continuous variables, with statistical analysis performed using specialized software (STATA, GeMTC) [27].
Cognitive Research Protocols
Studies investigating cognitive effects employ distinct methodologies sensitive to hormonal manipulations:
Population: Healthy premenopausal women, including both naturally cycling women and established OC users (minimum 3 months continuous use). Group Matching: Careful matching for age, education, and socioeconomic status to control for potential confounds [28] [25]. Cognitive Assessment: Standardized spatial tasks, particularly 3D mental rotations tests, administered consistently across participants. Hormonal Activity Quantification: Novel coding schemes quantifying the estrogenic, progestational, and androgenic activity of OC formulations based on their pharmacological profiles [25]. Control for Menstrual Cycle Phase: For naturally cycling women, testing scheduled during early follicular (low hormone) and mid-luteal (high progesterone) phases to account for endogenous fluctuations [25] [29].
This methodological approach allows researchers to disentangle the specific contribution of progestin androgenicity to cognitive performance while controlling for other formulation variables.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for Androgenicity Studies
| Reagent/Material | Research Application | Function/Justification |
|---|---|---|
| STATA 18.0 with net-meta package | Statistical analysis | Network meta-analysis incorporating direct and indirect treatment comparisons |
| GeMTC 14.3 software | Bayesian network meta-analysis | Enables complex treatment network modeling with random effects |
| CINeMA (Confidence in NMA) framework | Evidence quality assessment | Evaluates certainty of evidence across multiple domains in network meta-analysis |
| Walking Corsi Test (WalCT) | Topographic memory assessment | Evaluates spatial learning and memory in environmental context |
| 3D Mental Rotations Tests | Spatial cognition measurement | Assesses visuospatial ability sensitive to hormonal manipulations |
| Modified Downs and Black Checklist | Study quality assessment | Evaluates methodological rigor of included studies in systematic reviews |
| Androgen Response Element (ARE) Reporter Assays | Molecular mechanism studies | Quantifies androgen receptor activation/inhibition in response to progestins |
| Big Five Personality Inventory | Participant characterization | Controls for potential personality confounds between OC users and non-users |
This toolkit enables comprehensive investigation across multiple levels of analysis, from molecular mechanisms to cognitive and clinical outcomes. The statistical software packages are particularly critical for synthesizing evidence across multiple study designs, while the cognitive assessments provide sensitive measures of neurocognitive effects related to androgenicity [27] [25] [29].
The androgenic properties of progestins represent a fundamental dimension differentiating combined oral contraceptives, with demonstrable effects on clinical outcomes, cognitive performance, and physiological parameters. Androgenic progestins like levonorgestrel remain valuable for specific applications such as emergency contraception but exhibit less favorable bleeding profiles. Conversely, anti-androgenic options like drospirenone offer advantages for women concerned with androgenic side effects such as acne or hirsutism [27] [25].
For researchers investigating hormonal contraceptive effects, these findings underscore the critical importance of disaggregating OC users by progestin type and specifically considering androgenicity rather than treating OC users as a homogeneous population. Future research should prioritize head-to-head comparisons of specific formulations, incorporate longitudinal designs to track long-term effects, and further elucidate the molecular mechanisms through which progestin androgenicity modulates neural function and clinical outcomes. Such detailed characterization will advance both personalized contraceptive selection and our fundamental understanding of hormone-cognition interactions.
The widespread use of synthetic hormones, particularly in oral contraceptive pills (OCPs), represents a significant neuroendocrine intervention, with approximately 150 million users globally as of 2020 [30]. For researchers and drug development professionals, understanding how these synthetic compounds influence brain organization is crucial for optimizing therapeutic efficacy and minimizing neurological side effects. This review synthesizes recent neuroimaging evidence comparing the effects of synthetic hormones and natural hormonal cycling on brain structure and functional connectivity, providing a foundation for developing more targeted and brain-compatible hormonal therapies.
The neuroendocrine system exhibits remarkable sensitivity to both endogenous and exogenous hormones, with gonadal hormone receptors widely distributed throughout the brain [31]. While natural hormonal cycling creates a rhythmic pattern of estrogen and progesterone fluctuation across menstrual phases, synthetic hormones in OCPs fundamentally alter this dynamic by suppressing endogenous hormone production and introducing steady-state levels of synthetic equivalents [20]. This distinction in hormonal profiles between natural cycling and OCP use provides a powerful comparative framework for investigating hormone-brain relationships, with implications for drug development, personalized medicine, and the treatment of hormone-sensitive neurological conditions.
Structural Brain Changes: Natural Cycling Versus Oral Contraceptive Use
Whole-Brain Structural Dynamics Across Natural Menstrual Cycles
Emerging evidence from densely-sampled neuroimaging studies reveals that the natural menstrual cycle involves coordinated structural changes across the entire brain rather than isolated region-specific effects. In typical cycling, spatiotemporal patterns of brain volume changes are significantly associated with serum progesterone levels, demonstrating the hormone's widespread influence on brain morphology [31]. These structural fluctuations follow the rhythmic hormonal patterns characteristic of the menstrual cycle, with the follicular phase marked by rising estradiol and low progesterone, and the luteal phase characterized by elevated progesterone and a secondary estradiol peak [31].
Advanced precision imaging techniques have enabled researchers to track these dynamic changes with unprecedented temporal resolution. The distribution of estrogen and progesterone receptors throughout cerebral and cerebellar tissues provides a biological basis for these widespread structural effects [31]. Notably, estradiol and progesterone play pivotal roles in synaptogenesis, myelination processes, and modulation of spine density, suggesting potential mechanisms through which natural hormonal fluctuations drive cyclic structural brain changes [31].
Structural Modulation During Oral Contraceptive Use
In contrast to natural cycling, OCP use creates a distinctly different hormonal milieu characterized by suppressed endogenous progesterone and altered estradiol dynamics. Structural brain patterns during OCP use are primarily associated with serum estradiol levels rather than progesterone, reflecting the fundamental shift in hormonal regulation [31]. This selective suppression of progesterone in OCP users creates an estradiol-dominant state similar to that observed in endocrine conditions like endometriosis, providing insights into how synthetic hormonal profiles differentially influence brain structure [31].
The structural changes observed during OCP use appear more widely distributed rather than region-specific, suggesting that synthetic hormones exert a coordinated influence on brain morphology, though through different hormonal mechanisms than natural cycling [31]. This widespread structural effect aligns with the broad distribution of hormone receptors throughout the brain and underscores the global impact of synthetic hormonal interventions on brain architecture.
Table 1: Comparative Structural Brain Changes in Natural Cycling Versus OCP Use
| Structural Feature | Natural Cycling Pattern | OCP Use Pattern | Key Associated Hormones |
|---|---|---|---|
| Whole-brain volume dynamics | Spatiotemporal patterns across menstrual cycle | Widespread, coordinated changes | Natural: Progesterone; OCP: Estradiol |
| Gray matter volume | Cyclic fluctuations in specific regions | Altered prefrontal volume | Varies by study design |
| Hippocampal structure | Subfield volume associations with hormone levels | Limited direct evidence | Estradiol, Progesterone |
| Cortical thickness | Spatiotemporal patterns across cycle | Not specifically reported | Progesterone in natural cycling |
Functional Connectivity and Network Organization
Methodological Approaches in Functional Connectivity Research
Resting-state functional magnetic resonance imaging (rs-fMRI) has emerged as the primary methodological tool for investigating hormone-mediated changes in brain network organization. Seed-based connectivity analysis represents a hypothesis-driven approach that examines functional connectivity between predetermined regions of interest and the rest of the brain [30]. This method has been particularly valuable for investigating brain circuits involving the amygdala, putamen, and dorsal anterior cingulate cortex (dACC), regions with known sensitivity to hormonal fluctuations [30].
More recently, functional connectome fingerprinting has provided a multivariate, data-driven alternative that allows simultaneous interrogation of connectivity changes across the entire brain [30]. This approach detects whole-brain connectivity patterns that distinguish individuals from one another and has proven particularly sensitive for identifying network-level changes during OCP use [30]. Additional analytical frameworks include brain entropy (BEN) quantification, which measures irregularity and disorder in brain activity, and has been linked to hormonal influences on cognitive and behavioral traits [32].
Table 2: Key Methodological Approaches in Hormonal Neuroimaging Research
| Method | Primary Application | Key Advantages | Limitations |
|---|---|---|---|
| Seed-based connectivity | Focused investigation of specific neural circuits | Hypothesis-driven, high sensitivity to regional effects | Limited whole-brain perspective |
| Functional connectome fingerprinting | Multivariate, whole-brain network analysis | Data-driven, detects distributed network changes | Complex interpretation |
| Brain entropy (BEN) mapping | Quantification of neural activity irregularity | Links neural activity patterns to behavior | Emerging methodology |
| Dense-sampling designs | Individualized tracking of hormone-brain dynamics | High temporal resolution, within-subject sensitivity | Logistically challenging |
OCP-Induced Alterations in Functional Connectivity
Randomized, double-blind, placebo-controlled crossover studies have revealed that OCPs induce widespread alterations in functional connectivity patterns, particularly within subcortical, executive, and somatomotor circuits [30]. A particularly significant finding is that OCP use increases between-subject similarity in functional connectomes, suggesting a loss of individual idiosyncrasy during synthetic hormone exposure [30]. This homogenization effect represents a fundamental shift in brain network organization that may underlie both therapeutic benefits and adverse effects reported by OCP users.
Intraclass correlation analyses have demonstrated that the reduction in functional connectivity idiosyncrasy during OCP use spans multiple major brain networks, including the default mode, executive, limbic, salience, somatomotor, and subcortical networks [30]. This broad network-level effect underscores the extensive reach of synthetic hormonal influence on functional brain organization and highlights the potential for OCPs to modulate cognitive and emotional processes mediated by these networks.
Replication Challenges and Emerging Connectivity Patterns
Despite growing evidence of OCP-related connectivity changes, the field faces significant replication challenges. Conceptual replication analyses have largely failed to confirm previously reported seed-based connectivity changes in edges containing amygdala, putamen, or dorsal anterior cingulate nodes [30]. This inconsistency highlights the methodological complexity of hormone connectivity research and suggests that synthetic hormone effects may be more readily detectable at the network level than through focused seed-based approaches.
Emerging evidence indicates that functional connectivity changes associated with OCP use show significant correlations with increases in negative affect, with specific connectivity edges demonstrating correlations with Daily Record of Severity of Problems (DRSP) symptom scores [30]. These findings suggest that OCP-induced alterations to brain network organization may underlie mood-related side effects reported by some users and represent an important consideration for future neuroimaging research involving hormonal contraceptive users [30].
Molecular Mechanisms and Signaling Pathways
Hormonal Modulation of Neural Activity and Entropy
Progesterone exerts significant influence on neural function through the modulation of brain entropy (BEN), which quantifies irregularity and disorder in brain activity. Research has demonstrated a negative correlation between progesterone levels and BEN in the frontoparietal network (FPN), including the dorsolateral prefrontal cortex (DLPFC) and posterior parietal cortex (PPC), as well as in the limbic network, encompassing the amygdala, hippocampus, and parahippocampal cortex [32]. This progesterone-mediated decrease in neural entropy enhances temporal coherence of neural activity, which subsequently increases functional connectivity within these regions during resting-state [32].
The relationship between progesterone, brain entropy, and behavior represents a crucial mechanistic pathway through which hormones influence cognition and behavior. Mediation analyses have demonstrated that progesterone reduces impulsivity traits by decreasing BEN in the left DLPFC and subsequently increasing functional connectivity within this region [32]. This pathway illustrates how hormonal effects on neural temporal dynamics can translate to meaningful behavioral outcomes, with implications for understanding both natural hormonal cycling and synthetic hormone effects.
Figure 1: Progesterone modulates impulsivity through brain entropy and functional connectivity pathways. This mechanism demonstrates how hormonal effects on neural temporal dynamics translate to behavioral outcomes.
Neuroendocrine Disruption by Environmental Chemicals
While beyond the direct scope of synthetic hormones, research on endocrine-disrupting chemicals (EDCs) provides valuable comparative insights into how exogenous compounds can interfere with neuroendocrine systems. EDCs such as bisphenol A (BPA), phthalates, polychlorinated biphenyls (PCBs), and pesticides can cross the blood-brain barrier and induce hypothalamic inflammation, potentially disrupting the reproductive axis and broader neural function [33]. These compounds often act through nuclear and nonnuclear steroid hormone receptors, nonsteroid receptors, and enzymatic pathways involved in steroid biosynthesis [33].
The inflammatory response to EDC exposure involves microglia activation, increased proinflammatory cytokine production, and astroglia-mediated oxidative stress, creating a neuroinflammatory environment that can contribute to neuronal apoptosis and astrogliosis [33]. This mechanistic pathway shares similarities with potential synthetic hormone effects and highlights the vulnerability of neuroendocrine centers to exogenous chemical interventions. Understanding these disruptive mechanisms provides important context for developing synthetic hormones with minimized neuroinflammatory potential.
Cognitive and Behavioral Correlates
Topographic Memory and Spatial Cognition
Research on topographic memory, which enables the recall of pathways and spatial information about locations, distances, and directions, has revealed significant differences between naturally cycling women and OCP users. OCP users demonstrate superior performance in learning eight-step sequence paths compared to naturally cycling women across different cycle phases [20]. This advantage in topographic learning among OCP users mirrors findings in other memory domains, including verbal and emotional memory, suggesting a broad cognitive influence of synthetic hormone profiles [20].
The hormonal mechanisms underlying these cognitive differences may relate to the suppression of natural hormonal fluctuations and their replacement with steady-state synthetic hormone levels. Natural cycling women exhibit performance variations across menstrual phases, with visuospatial skills typically improved during the follicular phase when estrogen levels are low [20]. In contrast, OCP users maintain consistent performance levels, potentially benefiting from the stabilization of hormonal levels and the reduction of cyclic cognitive fluctuations [20].
Emotional Processing and Mood Regulation
OCP use has been associated with alterations in emotional processing and mood regulation, with neuroimaging studies providing potential neural correlates for these behavioral effects. The observed increase in between-subject similarity of functional connectomes during OCP use [30] may relate to emotional side effects reported by some users. Furthermore, specific connectivity changes have been significantly associated with increases in negative affect, suggesting a neural basis for mood-related side effects that lead some women to discontinue OCP use [30].
The interaction between synthetic hormones and stress responsiveness represents another important pathway for emotional modulation. In naturally cycling women, cortisol exerts negative effects on memory, but OCP users show reduced susceptibility to these cortisol-mediated memory impairments [20]. This interaction between hypothalamic-pituitary-adrenal stress-related hormones and hypothalamic-pituitary-gonadal hormones illustrates the complex endocrine cross-talk that influences emotional and cognitive processing [20].
Experimental Protocols and Methodologies
Dense-Sampling Neuroimaging Designs
Dense-sampling methodologies represent a paradigm shift in neuroendocrine research, involving the longitudinal tracking of individual participants across extended periods with high temporal resolution. This approach typically involves collecting brain imaging and venipuncture data throughout the entire menstrual cycle, enabling researchers to capture individualized trajectories of structural and functional brain patterns in relation to day-to-day hormone fluctuations [31]. The enhanced sensitivity of this design allows detection of subtle hormone-induced changes that might be overlooked in less frequent sampling approaches.
The implementation of dense-sampling protocols requires rigorous standardization of procedures across multiple sessions. Key components include daily or near-daily MRI acquisitions, simultaneous blood draws for hormone assay, precise cycle phase documentation, and controlled timing for scan sessions to minimize circadian influences [31]. This methodological framework provides a template for future investigations of both natural cycling and synthetic hormone effects on brain structure and function.
Randomized Controlled Trial Designs for OCP Research
Gold-standard randomized, double-blind, placebo-controlled crossover designs have been successfully implemented to test the effects of OCPs on brain connectivity [30]. These protocols typically involve participants completing both active OCP and placebo conditions in counterbalanced order, with neuroimaging assessments conducted during each condition. This within-subject design controls for individual differences in baseline connectivity and increases statistical power for detecting OCP-specific effects.
Critical methodological considerations in OCP trials include sufficient duration for hormonal stabilization (typically 2-3 months per condition), careful timing of scanning sessions relative to pill administration, and standardized procedures for assessing and analyzing connectivity changes [30]. The implementation of both hypothesis-driven seed-based analyses and exploratory data-driven connectome-wide approaches within the same study provides complementary insights into OCP effects at both regional and network levels [30].
Figure 2: Experimental workflow for randomized controlled trials investigating OCP effects on brain structure and function, highlighting the crossover design and multimodal assessment.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Hormonal Neuroimaging Research
| Research Tool | Primary Function | Application Notes |
|---|---|---|
| High-sensitivity hormone assays | Quantification of serum estradiol, progesterone, LH, FSH | Essential for correlating hormone levels with neural measures |
| Resting-state fMRI protocols | Assessment of functional connectivity patterns | Standardized acquisition parameters enable cross-study comparisons |
| T1-weighted structural sequences | Volumetric analysis of brain structure | High-resolution images enable detection of subtle structural changes |
| Seed-based connectivity analysis | Focused investigation of specific neural circuits | Ideal for hypothesis-driven research on target regions |
| Functional connectome fingerprinting | Multivariate, whole-brain network analysis | Data-driven approach for detecting distributed network changes |
| Brain entropy (BEN) mapping | Quantification of neural activity irregularity | Emerging metric for hormone-brain-behavior relationships |
| Singular value decomposition (SVD) | Identification of spatiotemporal patterns in structural data | Effective for analyzing dense-sampling datasets |
The comprehensive analysis of synthetic hormone effects on brain structure and functional connectivity reveals a complex landscape of neuroendocrine influence. OCPs induce widespread alterations in brain network organization, particularly within subcortical, executive, and somatomotor circuits, while simultaneously reducing individual idiosyncrasy in functional connectomes [30]. These network-level changes occur alongside structural brain dynamics that differ fundamentally from those observed during natural menstrual cycling [31].
Future research directions should include larger-scale dense-sampling studies that directly compare natural cycling, OCP use, and other hormonal contraceptive methods; investigation of individual difference factors that predict neurological responses to synthetic hormones; and longitudinal studies examining the persistence of OCP-induced brain changes following discontinuation. For drug development professionals, these findings highlight the importance of considering neurological effects when designing new hormonal formulations and suggest opportunities for developing compounds that optimize both contraceptive efficacy and brain compatibility.
The integration of multimodal neuroimaging with precise hormone assessment and detailed behavioral measures will continue to advance our understanding of how synthetic hormones influence the human brain. This knowledge provides a foundation for developing more personalized hormonal therapies that account for individual differences in neuroendocrine response profiles, ultimately improving outcomes for the millions of women who use synthetic hormones for contraceptive and therapeutic purposes.
Research Design and Methodology: Integrating Endocrine Status into Preclinical and Clinical Studies
Within hormonal research, particularly studies comparing oral contraceptive (OC) users to naturally cycling (NC) women, the accurate determination of menstrual cycle phases is a critical methodological concern. The validity of such research, which explores cognitive, affective, and neural correlates of hormone fluctuations, hinges on the precision of phase identification [28] [34]. Historically, this process has relied on error-prone projection methods, but technological and methodological advancements are providing more robust tools for researchers and clinicians [34] [35]. This guide objectively compares the current landscape of tracking and verification methodologies, providing the experimental data and protocols necessary to inform rigorous study design in pharmaceutical and scientific fields.
Methodological Comparison: Accuracy and Application
A 2023 study critically evaluated common methodologies for determining menstrual cycle phase against a rigorous within-person 35-day assessment of circulating ovarian hormones. The findings, summarized in the table below, reveal significant limitations in popular approaches [34].
| Method Category | Specific Method | Reported Accuracy/Agreement | Key Limitations |
|---|---|---|---|
| Self-Report Projection | Forward calculation (from last menses) | Low (Cohen's κ -0.13 to 0.53) [34] | Assumes prototypical cycle length; highly error-prone due to natural variation [34]. |
| Backward calculation (from next menses) | Low (Cohen's κ -0.13 to 0.53) [34] | Relies on accurate prediction of next menses; cycle length variability introduces error [34]. | |
| Hormone-Assisted Confirmation | Prescribed hormone ranges | Low (Cohen's κ -0.13 to 0.53) [34] | Uses standardized ranges that may not account for inter-individual hormone level variation [34]. |
| Limited hormone assays (e.g., two time points) | Low (Cohen's κ -0.13 to 0.53) [34] | Insufficient data points to capture individual hormone dynamics and confirm phase transitions [34]. | |
| Wearable Sensor & Machine Learning | Multi-parameter physiological signals (RF Model, 3-phase) | 87% accuracy, 0.96 AUC [35] | Requires validation against a gold standard (e.g., LH tests); performance can vary with signal type and cycle regularity [35]. |
| Multi-parameter physiological signals (RF Model, 4-phase) | 71% accuracy, 0.89 AUC [35] |
Experimental Protocols for Phase Determination
Protocol: Gold Standard Hormonal Assessment
This protocol is designed to establish a definitive hormonal profile for cycle phase determination in a research setting [34].
- Objective: To accurately map serum concentrations of estradiol and progesterone across the menstrual cycle to define phase transitions.
- Materials:
- Phlebotomy Kit: For consistent serum sample collection.
- Immunoassay Kits: Validated for quantifying estradiol and progesterone in serum.
- Luteinizing Hormone (LH) Urinalysis Strips: To identify the LH surge for pinpointing ovulation.
- Procedure:
- Participants record the first day of their period (Cycle Day 1).
- Beginning on Cycle Day 6, participants begin daily urinary LH testing to detect the surge.
- Serum samples are collected at a minimum of eight time points: three during the follicular phase (e.g., days 2, 7, 12), one peri-ovulatory (within 24-48 hours of a positive LH test), and four during the luteal phase (e.g., days 2, 5, 8, and 11 post-LH surge) [34].
- Serum is analyzed for estradiol and progesterone concentrations.
- Phase Determination Criteria:
- Early Follicular: Low, stable estradiol and progesterone.
- Late Follicular/Ovulatory: Rising estradiol, leading to a distinct peak followed by the LH surge.
- Mid-Luteal: Sustained elevation in progesterone.
- Late Luteal: Decline in both progesterone and estradiol.
Protocol: Machine Learning Classification Using Wearable Data
This protocol outlines the procedure for using physiological data from wearable devices to classify menstrual cycle phases, as validated in a 2025 study [35].
- Objective: To train a machine learning model to automatically identify menstrual cycle phases using non-invasively collected physiological data.
- Materials:
- Wrist-worn Wearable Device: Capable of continuous monitoring of skin temperature, heart rate (HR), interbeat interval (IBI), and electrodermal activity (EDA) [35].
- Urinary Luteinizing Hormone (LH) Tests: To provide a reference point for ovulation and label training data.
- Data Processing Infrastructure: Software for signal processing and feature extraction (e.g., mean, standard deviation, min/max for each signal over a 24-hour window).
- Procedure:
- Data Collection: Participants wear the device continuously for a minimum of two complete menstrual cycles. They self-report the first day of menses and perform daily urinary LH tests to identify the day of the LH surge.
- Data Labeling: Each day is assigned a phase label based on the reference method:
- Menses (P): Days of bleeding.
- Follicular (F): Days after menses ends and before the fertile window.
- Ovulation (O): The day of the LH surge plus 2 days before and 3 days after [35].
- Luteal (L): Days after the ovulation phase until the next menses.
- Feature Extraction: For each 24-hour period, multiple features are extracted from each physiological signal.
- Model Training & Validation: A Random Forest classifier is trained on the feature set. Validation is performed using a leave-last-cycle-out approach, where all cycles from a participant except the last one are used for training, and the final cycle is used for testing [35].
The Scientist's Toolkit: Key Research Reagents and Materials
The following table details essential materials for conducting rigorous menstrual cycle phase determination research.
| Item | Function in Research |
|---|---|
| Urinary Luteinizing Hormone (LH) Tests | Provides a consumer-accessible, non-invasive method to detect the LH surge, which is a critical reference point for confirming ovulation in study protocols [35]. |
| Immunoassay Kits (for Estradiol & Progesterone) | Enable quantitative measurement of key ovarian hormones in serum, saliva, or urine samples. This is the gold standard for confirming hormonal phases and creating labeled datasets for machine learning models [34]. |
| Wrist-worn Wearable Device (Multi-parameter) | Captures continuous, real-world physiological data (e.g., skin temperature, HR, IBI). Serves as the input data source for developing automated, non-invasive phase classification algorithms [35]. |
| Phlebotomy Supplies | Allow for the consistent collection of blood serum, which is necessary for obtaining the most accurate measurements of circulating hormone levels via immunoassay [34]. |
Implications for OC vs. NC Research
The choice of phase determination methodology has profound implications for studies comparing OC users to NC women. Research has shown no significant pre-existing personality differences between these groups, suggesting that previously reported cognitive and affective differences are more likely linked to neuroendocrinology than to confounding personal characteristics [28] [36]. This underscores the necessity of precise hormonal and phase characterization. Employing error-prone methods like simple self-report can introduce misclassification bias, obscuring true between-group differences and leading to unreliable conclusions [34]. The move towards more objective, data-driven methods, including hormone assays and wearable-based monitoring, is therefore essential for enhancing the validity and replicability of research in this field.
Characterizing the complex endocrine environment is a fundamental challenge in biomedical research, particularly when comparing specific populations such as oral contraceptive users and naturally cycling women. Hormonal assays provide the quantitative foundation for understanding these physiological differences, but researchers must navigate a landscape of methodological trade-offs between widely available immunoassays and increasingly accessible mass spectrometry techniques. The choice of analytical platform carries profound implications for data quality, with studies demonstrating that failing to account for technical variability and biological confounding factors can generate false discoveries in up to 41% of measured analytes [37]. This comparison guide examines the key metrics, methodologies, and biomarkers essential for robust experimental design in studies comparing synthetic and endogenous hormone profiles, providing researchers with the technical foundation needed for conclusive investigations.
Technical Comparison of Hormonal Assay Platforms
Performance Characteristics of Major Assay Types
The selection of an appropriate analytical platform represents one of the most critical decisions in hormonal biomarker research. The table below compares the fundamental characteristics of the two primary methodologies used in hormone quantification.
Table 1: Technical Comparison of Hormonal Assay Platforms
| Parameter | Immunoassays | Mass Spectrometry (LC-MS/MS) |
|---|---|---|
| Principle | Antibody-antigen binding | Separation by mass/charge ratio |
| Throughput | High | Moderate to high |
| Cost per sample | Lower | Higher |
| Specificity | Susceptible to cross-reactivity [38] [39] | High specificity for structurally similar compounds |
| Multiplexing capability | Limited (usually single analyte) | Can measure multiple hormones simultaneously [39] |
| Sample volume | Higher (when measuring multiple analytes separately) | Lower (for multiple analytes) [39] |
| Dynamic range | Limited | Wide |
| Technical expertise required | Moderate | High [39] |
| Susceptibility to interference | Subject to heterophile antibodies, biotin, cross-reactivity [38] | Minimal with proper method development |
Analytical Challenges and Interference Considerations
Immunoassays present several specific analytical challenges that can significantly impact data quality in endocrine research:
Cross-reactivity: Structurally similar molecules (metabolites, precursors, or synthetic hormones) may be detected by the same antibody, leading to falsely elevated results [38]. For example, dehydroepiandrosterone sulfate (DHEAS) cross-reacts with several testosterone immunoassays, particularly problematic in female samples where testosterone concentrations are naturally low [39].
Matrix effects: Differences in binding protein concentrations between study populations can introduce bias. Pregnant women and oral contraceptive users have elevated sex hormone-binding globulin (SHBG) levels, which can interfere with total hormone measurements in immunoassays not designed to account for these variations [39].
Endogenous antibodies: Heterophile antibodies or autoantibodies present in patient samples can cause either falsely elevated or suppressed values depending on the assay design [38].
Biotin interference: High concentrations of biotin (a common dietary supplement) can interfere with immunoassays utilizing the biotin-streptavidin separation system [38].
Table 2: Common Interferences in Hormonal Immunoassays
| Interference Type | Mechanism | Affected Analytes | Solution |
|---|---|---|---|
| Cross-reactivity | Antibody recognizes structurally similar molecules | Steroid hormones (testosterone, cortisol, estradiol) [39] | LC-MS/MS or antibody with higher specificity |
| Heterophile antibodies | Human antibodies against animal immunoglobulins | Various sandwich immunoassays [38] | Use blocking reagents, sample dilution |
| Biotin | Interferes with biotin-streptavidin separation | Immunoassays using this system [38] | Ask patients to discontinue biotin before testing |
| Matrix effects | Differences in binding protein concentrations | Total hormone measurements in special populations [39] | Use equilibrium dialysis, LC-MS/MS |
Essential Biomarker Panels for Endocrine Profiling
Comprehensive Hormonal Biomarkers
Different research questions require assessment of specific hormonal panels. The following table outlines key biomarkers relevant to characterizing endocrine environments in oral contraceptive users versus naturally cycling women.
Table 3: Essential Biomarker Panels for Endocrine Profiling
| Biomarker Category | Specific Biomarkers | Research Utility |
|---|---|---|
| Sex Hormones | Estrone (E1), Estradiol (E2), Estriol (E3), Progesterone, Testosterone, 5α-DHT, DHEA-S [40] | Assess endogenous hormone production and suppression |
| Estrogen Metabolites | 2-Hydroxyestrone, 4-Hydroxyestrone, 16α-Hydroxyestrone, 2-Methoxyestrone [40] | Evaluate estrogen metabolism pathways |
| Adrenal Hormones | Cortisol awakening response, Diurnal free cortisol pattern, Cortisone, α-THF, β-THF, β-THE [40] | Characterize HPA axis function and stress response |
| Neurotransmitter Markers | Homovanillate (HVA - dopamine), Vanilmandelate (VMA - norepinephrine/epinephrine) [40] | Assess neuroendocrine function |
| Menstrual Cycle Mapping | a-Pregnanediol, b-Pregnanediol, Estrone, Estradiol (multiple time points) [40] | Detailed ovulatory function assessment |
Biomarkers With Diagnostic Utility
Certain hormonal biomarkers demonstrate particular relevance for specific clinical conditions:
Aromatase: Shows promising diagnostic accuracy for endometriosis with pooled sensitivity of 0.79 and specificity of 0.89 in meta-analysis [41].
Serum prolactin: Demonstrated high specificity (0.92) but lower sensitivity (0.45) for endometriosis diagnosis [41].
Estrogen receptors: ER-α and ER-β show moderate diagnostic characteristics for endometriosis, with ER-α having higher sensitivity (0.75) but lower specificity (0.47) compared to ER-β (sensitivity 0.65, specificity 0.68) [41].
Experimental Protocols for Comparative Studies
Protocol for Verbal Memory Assessment in Contraceptive Research
A 2025 study investigating cognitive effects of combined oral contraceptives (COCs) provides a robust methodological template for comparative research [22]:
Population & Recruitment:
- Recruit 205 healthy premenopausal women (90 COC users, 115 naturally cycling)
- Exclude women using other hormonal contraceptives (patches, IUDs, mini-pills), those with sterilization/hysterectomy, pregnancy, or use of morning-after pill within one month before testing
- Obtain ethical approval following Declaration of Helsinki and written informed consent
Hormonal Status Assessment:
- Determine contraceptive use via written questionnaire and face-to-face interview within 30 days of cognitive testing
- For naturally cycling women, determine menstrual cycle phase using backward-count method based on expected next menstruation
- Verify cycle phase with plasma progesterone levels (>5 ng/mL suggests luteal phase) [22]
Cognitive Testing Protocol:
- Assess verbal memory using Verbal Affective Memory Task (VAMT)
- Use either 24-word (VAMT-24) or 26-word (VAMT-26) versions, which can be analyzed jointly
- Present positive, negative, and neutral words across immediate, short-term, and long-term recall conditions
- Primary outcome: Percentage correctly recalled words across all valence categories and conditions
- Secondary outcome: Affective memory bias (percentage correct for positive words minus percentage correct for negative words) [22]
Mood Assessment:
- Administer Profile of Mood State (POMS) questionnaire to assess subclinical mood symptoms
- Calculate Total Mood Disturbance (TMD) score as potential covariate
Statistical Analysis:
- Employ multiple linear regression to assess group differences in verbal memory performance
- Conduct planned post-hoc analyses for immediate, short-term, and long-term recall components
- Perform sensitivity analyses excluding participants with uncertain contraceptive status
Protocol for Emotion and Memory Assessment
A complementary approach examining emotional processing in contraceptive users demonstrates alternative methodological considerations:
Experimental Design:
- Compare women using hormonal contraceptives with naturally cycling women
- Present positive, negative, and neutral images while participants apply different emotion regulation strategies (distancing, reinterpretation, immersion)
- Administer subsequent memory tests for emotional stimuli [42]
Key Findings:
- Women on hormonal contraceptives showed stronger emotional reactions compared to naturally cycling women
- When using strategies like distancing or reinterpretation, contraceptive users remembered fewer details of negative events while maintaining general memory integrity
- Immersion strategies boosted memory for positive images in both groups [42]
Research Reagent Solutions and Essential Materials
Table 4: Essential Research Materials for Hormonal Biomarker Studies
| Reagent/Material | Function/Application | Technical Considerations |
|---|---|---|
| Multiplex Immunoassay Panels | Simultaneous measurement of multiple biomarkers | Human DiscoveryMAP measures 171+ proteins and small molecules; verify cross-reactivity [37] |
| LC-MS/MS Systems | High-specificity steroid hormone quantification | Requires extensive validation; superior for steroid hormones [39] |
| Quality Control Materials | Monitoring assay performance over time | Use independent controls spanning expected concentration range; not just kit controls [39] |
| Sample Collection Tubes | Proper specimen preservation | Consider serum vs. plasma, anticoagulant type, separating gel [38] |
| Antibody Panels | Targeted immunoassay development | Verify specificity for synthetic vs. endogenous hormones; check cross-reactivity [38] |
| Reference Standards | Calibration and quantification | Use matrix-matched standards where possible [39] |
Methodological Considerations for Robust Study Design
Pre-Analytical Factors
Multiple pre-analytical variables can significantly impact hormonal measurement quality:
- Timing of collection: Account for diurnal variation (cortisol, testosterone) and menstrual cycle phase (estradiol, progesterone) [38]
- Sample handling: Maintain proper storage temperature (ACTH at +4°C) and minimize freeze-thaw cycles [38] [39]
- Matrix selection: Serum is generally preferred over plasma; avoid EDTA for assays using metallic ion labels or enzyme cofactors [38]
Population Stratification Strategies
The substantial molecular differences driven by sex and hormonal status necessitate careful study design:
- Sex differences: 96 of 171 serum biomarkers show significant sex-based variation [37]
- Contraceptive effects: 66 serum molecules vary significantly between oral contraceptive users and naturally cycling women [37]
- Menstrual cycle effects: While most biomarkers show minimal variation between follicular and luteal phases, FSH, progesterone, neuropilin-1, IGFBP-2, and IGFBP-3 demonstrate cycle-dependent changes [37]
- Statistical implications: Unbalanced groups can yield up to 40% false discoveries when not matched for sex, and up to 41% false discoveries when not matched for oral contraceptive use [37]
Visual Experimental Framework
The following diagram illustrates the key methodological considerations and potential confounding factors in comparative studies of oral contraceptive users versus naturally cycling women:
Robust characterization of endocrine environments in comparative studies of oral contraceptive users and naturally cycling women requires meticulous attention to analytical methodology. The choice between immunoassays and mass spectrometry represents a fundamental trade-off between throughput and specificity, with immunoassays suffering from potential cross-reactivity with synthetic hormones and mass spectrometry offering superior analytical specificity at higher cost and technical complexity. Researchers must carefully consider population stratification, pre-analytical variables, and potential confounders to avoid the high false discovery rates that have plagued previous biomarker studies. The experimental frameworks and technical comparisons presented here provide a foundation for designing methodologically sound studies that can yield conclusive findings in this complex research domain.
In the study of hormonal profiles, research comparing naturally cycling (NC) women and oral contraceptive (OC) users presents unique methodological challenges. Group stratification is a foundational technique that addresses these challenges by ensuring study populations are divided into distinct, homogeneous subgroups (strata) before randomization [43]. This process is particularly critical in this field due to the profound biological and pharmacological differences between NC and OC groups. NC women experience dynamic fluctuations of endogenous hormones like estradiol and progesterone throughout their menstrual cycle, with levels varying significantly between the follicular and luteal phases [44]. In contrast, OC users exhibit a fundamentally different endocrine profile characterized by significantly lower and less variable levels of these endogenous hormones, alongside the introduction of synthetic hormones (e.g., ethinylestradiol and various progestins) [44] [13].
The primary rationale for implementing rigorous stratification strategies in NC/OC research is to account for multiple sources of heterogeneity that can otherwise confound research outcomes. Without proper stratification, the inherent variability within and between these groups can obscure true treatment effects, reduce statistical power, and compromise the validity of study conclusions. As noted in recent methodological reviews, "prestratifying on baseline covariates can mitigate accidental bias, by inducing independence between the treatment effect and unobserved covariates" [45]. This is especially relevant when investigating subtle neurobehavioral outcomes, where hormonal influences may interact with numerous other factors.
Key Stratification Variables for NC and OC Cohorts
Fundamental Stratification Factors
When designing studies comparing NC and OC cohorts, researchers should consider stratifying participants based on the following key variables, which have demonstrated significance in previous research:
- Hormonal Status Group: The primary stratification divides participants into NC women and OC users [44]. This fundamental distinction accounts for the profound differences in endocrine profiles between these populations.
- OC Formulation Characteristics: Among OC users, further stratification should consider the specific pharmacological properties of the contraceptive formulation, particularly:
- Progestin Androgenicity: Studies have identified meaningful differences between users of androgenic versus anti-androgenic progestins [44].
- Hormone Dose: Ethinyl estradiol dose variations (e.g., high vs. low) and progestin potency should be accounted for [13].
- Administration Regimen: Differences between monophasic, triphasic, and extended-cycle regimens may introduce relevant variability [13].
- Menstrual Cycle Phase for NC Women: For naturally cycling participants, stratification by menstrual cycle phase (follicular, ovulatory, luteal) is essential, as research shows "the luteal phase (i.e., premenstrual phase) has been linked with an exacerbation of numerous disorders, including anxiety disorders and depression" [44].
- Age at OC Initiation: This represents a crucial stratification variable, as "the only consensus found on the link between the use of OC and depression suggests that starting OC during adolescence is related to a vulnerability to receiving a diagnosis of depression in adulthood" [44].
Table 1: Key Stratification Variables for NC/OC Research Designs
| Stratification Category | Specific Variables | Research Rationale |
|---|---|---|
| Primary Group | Naturally Cycling (NC) vs. Oral Contraceptive (OC) Users | Fundamental difference in endocrine environment [44] |
| OC Formulation | Progestin androgenicity (androgenic vs. anti-androgenic) | Anti-androgenic OC users had higher levels of daily depression than NC women in all menstrual cycle phases, while androgenic OC users had higher levels in only two phases [44] |
| OC Formulation | Ethinyl estradiol dose (high vs. low) | Higher doses lead to less follicular development and lower overall endogenous hormone production [13] |
| OC Formulation | Administration regimen (21/7, 24/4, continuous) | Hormone-free intervals associated with some follicular development and E2 surges [13] |
| Menstrual Cycle | Phase (follicular, ovulatory, luteal) for NC women | Luteal phase linked with exacerbation of anxiety disorders and depression [44] |
| Developmental Timing | Age at OC initiation (adolescent vs. adult) | Starting OC during adolescence linked to vulnerability to depression diagnosis in adulthood [44] |
Additional Stratification Considerations
Beyond these fundamental variables, researchers should consider additional factors that may interact with hormonal status:
- Psychiatric History: Previous mood disorders or family history of hormone-sensitive conditions [44].
- Research Indication: Whether OCs are used primarily for contraception versus therapeutic management of conditions like endometriosis, PCOS, or PMDD [13].
- Methodological Factors: Timing of assessment relative to OC initiation, compliance measures, and hormone-free intervals [13].
Experimental Designs and Methodological Protocols
Daily Diary Assessment Protocol
A robust methodological approach for NC/OC research involves intensive longitudinal assessment, such as the daily diary design implemented in recent research [44]. This protocol enables researchers to capture dynamic fluctuations in outcomes while accounting for hormonal variations:
- Study Duration: 15-day online diary study divided into 3 phases throughout one menstrual cycle [44].
- Participant Criteria: Inclusion of women aged 18-45 with regular menstrual cycles (21-35 days) for NC participants; exclusion of those using other hormonal contraception, copper IUDs, or who had discontinued OC in the past 6 months [44].
- Data Collection: Daily emails sent in the evening with questionnaires completed on the same day [44].
- Measures Adaptation: Existing trait-level scales adapted for daily administration to assess outcomes such as anxiety, depression, perseverative cognition, stress, self-esteem, and life satisfaction [44].
- Analytical Approach: Hierarchical linear modeling (HLM) to account for nested data structure (days within participants) with significance level of p < 0.05 [44].
This design captured significant differences between groups, revealing that "OC users had higher daily levels of depression and lower daily levels of self-esteem than NC women" despite finding no differences in anxiety, perseverative cognition, or stress levels [44].
Fear Conditioning and Extinction Protocol
Neurobehavioral research has employed specialized protocols to investigate how hormonal contraceptives may influence fundamental emotional processes:
- Experimental Paradigm: Fear conditioning and extinction protocol assessing fear responses to safe contexts [13].
- Participant Groups: Comparison of current COC users, past COC users, and naturally cycling women [13].
- Formulation Analysis: Examination of how different OC formulations (varying by progestin type and estrogen dose) impact fear regulation [13].
- Key Findings: "Current and past COC users displayed greater fear return in safety contexts," with exploratory analyses linking "fear regulation impairment to higher ethinyl estradiol (EE) doses and certain progestins" [13].
This protocol demonstrates how stratification by specific OC formulation characteristics can reveal nuanced neurobehavioral effects that might be obscured in broader group comparisons.
Diagram 1: Stratified Research Workflow for NC/OC Studies. This workflow illustrates the sequential process from participant recruitment through data analysis, highlighting critical stratification points for both NC women and OC users.
Analytical Approaches for Stratified Designs
Statistical Considerations for Stratified Analyses
Implementing appropriate statistical methods is essential for valid interpretation of stratified studies comparing NC and OC cohorts:
- Hierarchical Linear Modeling (HLM): Effectively handles nested data structures (e.g., daily measurements within participants) and allows for examination of both within-person and between-person effects [44].
- Handling of Menstrual Cycle Phase: For NC women, statistical models should account for hormonal fluctuations across cycle phases, as "the premenstrual and menstrual phases are linked with an increase in depression" [44].
- Accounting for OC Formulation Heterogeneity: Analytical plans should include tests for interactions between hormonal status group and formulation characteristics, as "progestins differentially influence neurosteroids and neurotransmitters that are central to mechanisms of behavioral hormone sensitivity" [13].
- Randomization-Based Inference: As an alternative to population-based inference, randomization-based methods "primarily focus on the simple null hypothesis of no treatment effect" and can be particularly appropriate for randomized experiments [45].
Power and Sample Size Considerations
Stratified designs require careful attention to statistical power, particularly when investigating multiple subgroups:
- Avoid Over-Stratification: Excessive stratification can reduce sample size within individual strata, diminishing statistical power. Research suggests "using four or six stratification factors in smaller trials actually made things worse" [46].
- Targeted Stratification: Focus on "one or two really meaningful factors" that substantially influence the outcomes of interest [46].
- Balanced Allocation: Ensure proportional representation of key strata in the sample to maintain analytical integrity [43].
Table 2: Comparative Outcomes in Stratified NC/OC Research
| Outcome Measure | NC Women | Androgenic OC Users | Anti-Androgenic OC Users | Statistical Significance |
|---|---|---|---|---|
| Daily Depression Levels | Baseline | Higher than NC in 2/3 phases [44] | Higher than NC in all 3 phases [44] | p < 0.05 |
| Daily Self-Esteem Levels | Baseline | Lower than NC in all phases [44] | Lower than NC in all phases [44] | p < 0.05 |
| Daily Anxiety Levels | Baseline | No significant difference [44] | No significant difference [44] | Not significant |
| Fear Regulation in Safe Contexts | Baseline | Context-dependent impairment [13] | Context-dependent impairment [13] | Varies by formulation |
| Sleep Quality | Baseline | Marginally poorer sleep quality [47] | Marginally poorer sleep quality [47] | p<0.042 |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Materials for Stratified NC/OC Studies
| Item/Category | Specific Examples | Research Function |
|---|---|---|
| Hormone Assays | Liquid chromatography-tandem mass spectrometry (LC-MS/MS) for E2, EE, P4, testosterone [13] | Precisely measure both endogenous and exogenous hormone levels with high specificity |
| Electronic Diary Platforms | Custom online survey systems with scheduled email delivery [44] | Enable intensive longitudinal data collection with precise timing |
| Psychological Assessments | Adapted trait-level scales for daily administration (anxiety, depression, perseverative cognition) [44] | Measure psychological outcomes with ecological validity |
| Fear Paradigms | Fear conditioning and extinction protocols [13] | Assess fundamental emotional processes and regulation |
| Stratification Databases | Pharmaceutical reference databases for OC formulations [44] [13] | Accurately classify OC types by progestin androgenicity, dose, and regimen |
Diagram 2: Hormone-Behavior Pathway in NC/OC Research. This diagram illustrates the conceptual pathway through which hormonal factors, influenced by stratification variables, ultimately affect behavioral and mental health outcomes.
Effective stratification in studies comparing NC women and OC users requires meticulous attention to both biological and methodological considerations. The evidence indicates that key stratification variables include OC formulation characteristics (particularly progestin androgenicity and hormone dose), menstrual cycle phase for NC women, and developmental timing of OC initiation. These factors significantly impact outcomes ranging from emotional processing and mental health indicators to cognitive function.
Researchers should implement stratified designs that balance statistical power with methodological precision, avoiding over-stratification while ensuring key sources of heterogeneity are accounted for. The growing recognition of "dimensional hormone sensitivity" – the concept that "individuals differ not only in if they are behaviorally sensitive to hormones, but also to which specific changes they are sensitive" – underscores the need for more sophisticated stratification approaches in this field [13]. Future research should continue to refine these methodologies, particularly through head-to-head comparisons of specific OC formulations and longitudinal assessment of neurobehavioral outcomes across hormonal transition periods.
The historical over-reliance on male models in biomedical research has led to significant gaps in our understanding of female physiology and neuroendocrinology. This comparative guide examines the critical methodological considerations for research involving oral contraceptive (OC) users versus naturally cycling women (NCW), focusing on hormonal profiling techniques and their implications for cognitive and spatial memory research. The systematic investigation of exogenous and endogenous hormone effects represents a crucial step toward correcting historical biases and developing more inclusive, accurate research models that account for female-specific physiological processes.
Comparative Hormone Profiling: Methodologies and Analytical Approaches
Saliva-Based Hormone Assessment
Table 1: Comparison of Salivary Hormone Testing Methodologies
| Testing Characteristic | Doctor's Data Comprehensive Profile | MosaicDX Hormones Profile |
|---|---|---|
| Sample Type | Saliva [48] [49] | Saliva [50] |
| Collection Protocol | 4 separate collections [48] | Not specified |
| Turnaround Time | 3-5 days [48] | 1-2 weeks [50] |
| Key Sex Hormones Measured | Estradiol, Progesterone, Testosterone, DHEA [49] | Estradiol, Progesterone, Testosterone, DHEA [50] |
| Cortisol Pattern | 4 measurements [48] [49] | 4 measurements (AM, Noon, Evening, Night) [50] |
| Unique Ratios Calculated | Pg/E2 Ratio [48] | Pg/E2 Ratio, EQ (E3/(E1+E2)) Ratio [50] |
| Methodology | EIA [49] | Not specified |
Saliva testing measures the unbound, biologically active portion of hormones that reflects tissue availability, making it particularly valuable for assessing bioavailable hormone status [48] [50]. This methodology provides distinct advantages for research comparing OC users and NCW, as it captures the hormonally active components most likely to influence cognitive processes and brain function.
Experimental Evidence: Cognitive Performance Across Hormonal States
Table 2: Topographic Memory Performance in Oral Contraceptive Users vs. Naturally Cycling Women
| Experimental Measure | Naturally Cycling Women (NCW) | Oral Contraceptive Users (OC) | Statistical Significance |
|---|---|---|---|
| Sample Size | 25 participants [20] | 26 participants [20] | N/A |
| Mean Age | 25.7 (SD: 3.1) [20] | 24.7 (SD: 2.6) [20] | t-value: 1.348; p = 0.184 |
| Testing Phases | Early follicular (days 4-5); Mid-luteal (days 20-21) [20] | Active pill phase (days 20-21); Inactive pill phase (days 2-4) [20] | N/A |
| Walking Corsi Test: Learning 8-step sequence | Lower performance across both phases [20] | Consistently superior performance in both active and inactive phases [20] | Significant difference between groups |
| Walking Corsi Test: Delayed recall | No significant difference from OC users [20] | No significant difference from NCW [20] | Non-significant |
A 2018 study investigating topographic memory compared 25 NCW across two menstrual cycle phases with 26 OC users tested during active and inactive pill phases using the Walking Corsi Test (WalCT) [20]. The findings demonstrated that OC users showed superior performance in learning an eight-step sequence path compared to NCW, while no significant differences emerged in delayed recall of the same path [20]. This research highlights the complex relationship between hormonal profiles and specific cognitive domains, particularly the dissociation between learning acquisition and memory retention processes.
Experimental Protocols and Methodological Considerations
Participant Selection and Exclusion Criteria
The methodological rigor in participant selection is paramount for valid comparisons between OC users and NCW. The exclusion criteria should include: history of head trauma, neurological or psychiatric illness, use of benzodiazepines in the previous 3 months, use of illicit drugs, visual/auditory/motor impairments, and conditions capable of causing cognitive impairment [20]. These controls ensure that observed differences can be more reliably attributed to hormonal status rather than confounding factors.
Hormone Assessment Protocol
For comprehensive hormone profiling, the collection of saliva samples should occur at multiple time points to capture diurnal patterns and cyclic variations [48] [50]. The analytical protocol should include:
- Sample Collection: Four separate saliva collections throughout the day to assess diurnal cortisol patterns [48]
- Hormone Panels: Assessment of estradiol, progesterone, testosterone, and DHEA for both groups [49] [50]
- Ratio Calculations: Progesterone to Estradiol (Pg/E2) ratio to evaluate hormonal balance [48]
- Phase Verification: For NCW, testing should occur during specific cycle phases (early follicular: days 4-5; mid-luteal: days 20-21) confirmed through hormonal assessment [20]
Cognitive Testing Methodology
The Walking Corsi Test (WalCT) protocol involves participants learning an eight-step sequence path and recalling it after a five-minute delay [20]. This assessment tool has demonstrated sensitivity to hormonal fluctuations and gender-related differences in topographic memory [20]. The test should be administered in a controlled environment with standardized instructions and scoring procedures to ensure reliability across testing sessions and participant groups.
Experimental Workflow for Hormone and Cognitive Assessment
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Materials for Hormone and Cognitive Studies
| Research Tool | Specific Function | Application in Hormone Research |
|---|---|---|
| Saliva Collection Kit | Non-invasive sampling for hormone assessment [48] [50] | Measures unbound, bioavailable hormone fractions in OC users and NCW |
| Enzyme Immunoassay (EIA) | Quantitative hormone analysis [49] | Processes saliva samples to determine concentrations of estradiol, progesterone, cortisol |
| Walking Corsi Test (WalCT) | Topographic memory assessment [20] | Evaluates spatial learning and recall in different hormonal states |
| Color-Blind Safe Visualization Tools | Accessible data presentation [51] [52] | Creates inclusive figures for publications and presentations |
| Interactive Statistical Software | Data analysis and visualization | Performs comparative statistics between OC and NCW groups |
Data Visualization Considerations for Inclusive Research Communication
Effective data visualization requires careful consideration of color choices to ensure accessibility for color-blind readers, who represent approximately 8% of men and 0.5% of women [51]. Recommended practices include:
- Color Selection: Using color-blind safe palettes with red and blue as base colors while avoiding red-green combinations [51] [52]
- Pattern Enhancement: Incorporating shapes, patterns, and textures to complement color coding [51]
- Direct Labeling: Implementing direct labels on chart elements rather than relying solely on color-coded legends [51]
- Grayscale Verification: Ensuring all visualizations remain interpretable when converted to grayscale [51]
These practices ensure that research findings comparing OC users and NCW are accessible to the entire scientific community, regardless of color vision abilities.
Hormonal Impact on Cognitive Processing Pathways
This comparative analysis demonstrates that rigorous methodological approaches to studying OC users and NCW can yield significant insights into hormone-cognition relationships. The findings indicate that oral contraceptive use is associated with enhanced topographic learning compared to natural cycling women, while delayed recall remains comparable between groups [20]. These results underscore the importance of considering hormonal status as a critical variable in cognitive research and drug development. By implementing standardized hormone assessment protocols, validated cognitive measures like the Walking Corsi Test, and accessible data visualization practices, researchers can overcome historical biases and create more inclusive, accurate research models that advance our understanding of female physiology and cognition.
Challenges and Solutions: Addressing Variability and Adverse Effects in Research Populations
Navigating Inter- and Intra-Individual Variability in Menstrual Cycle Characteristics
Understanding the intricate patterns of menstrual cycle variability is paramount for researchers investigating hormonal interventions, drug development professionals designing female-specific clinical trials, and clinicians treating hormone-sensitive conditions. The menstrual cycle, often oversimplified as a standardized 28-day model, demonstrates substantial variability both between individuals (inter-individual) and within an individual's own cycles over time (intra-individual). This variability presents significant methodological challenges for research comparing naturally cycling women with oral contraceptive (OC) users, as synthetic hormones in OCs fundamentally alter endogenous hormonal profiles. Emerging evidence from large-scale digital cohort studies reveals that cycle characteristics systematically vary by age, ethnicity, and body mass index (BMI), necessitating more nuanced approaches to study design and data interpretation in hormonal research [53] [54] [55].
The central thesis of this review posits that accounting for the full spectrum of menstrual cycle variability is essential for valid comparisons between naturally cycling women and OC users. Synthetic hormones in OCs induce a hypogonadal state characterized by suppressed endogenous hormone production, a hypergonadal state from direct actions of synthetic hormones on target tissues, or a mixed state with elements of both [56]. Understanding how these pharmacologically-induced states compare to the dynamic hormonal fluctuations in naturally cycling women requires precise characterization of baseline cycle parameters across diverse populations.
Quantitative Landscape of Menstrual Cycle Variability
Core Cycle Parameters and Their Ranges
Table 1: Menstrual Cycle Characteristics from Large Cohort Studies
| Parameter | Overall Mean | Normal Range | Follicular Phase Mean | Llicular Phase Mean | Primary Source of Variability |
|---|---|---|---|---|---|
| Cycle Length | 28.9 days [57] | 22-36 days (95% range) [57] | 16.9 days [55] | 12.4 days [55] | Follicular phase (69% of variance) [57] |
| Cycle Length (App Data) | 28.7 days [53] | 22-38 days (5th-95th percentile) [53] | 15.7 days [58] | 13.3 days [58] | Follicular phase [58] |
Demographic Modifiers of Cycle Characteristics
Table 2: Cycle Length Variation by Demographic Factors
| Factor | Category | Mean Difference in Cycle Length (days) | Cycle Variability |
|---|---|---|---|
| Age | Under 20 vs. 35-39 | +1.6 [53] | 46% higher [53] |
| 45-49 vs. 35-39 | -0.3 [53] | 45% higher [53] | |
| Over 50 vs. 35-39 | +2.0 [53] | 200% higher [53] | |
| Ethnicity | Asian vs. White | +1.6 [53] [54] | Higher variability [53] |
| Hispanic vs. White | +0.7 [53] [54] | Higher variability [53] | |
| BMI | Class 3 obesity (BMI ≥40) vs. healthy | +1.5 [53] [54] | Higher variability [53] |
Methodological Framework for Cycle Research
Standardized Experimental Protocols
Gold-Standard Protocol for Menstrual Cycle Studies:
Study Design: Implement repeated measures designs with within-person comparisons as the gold standard. Between-subject designs conflate within-subject variance (attributable to changing hormone levels) with between-subject variance (attributable to each woman's baseline symptoms) [58].
Sampling Frequency: Collect daily or multi-daily (ecological momentary assessments) ratings of outcomes. For laboratory-based measures, obtain at least three observations per person across one cycle as a minimal standard, though three or more observations across two cycles provides greater confidence in reliability [58].
Cycle Phase Determination:
- First day of menses: Identify through full-flow bleeding (not spotting) [58] [59].
- Ovulation confirmation: Use urinary luteinizing hormone (LH) surge detection as a practical marker [57] [55]. More rigorous approaches combine LH testing with basal body temperature (BBT) tracking to detect the biphasic shift [55].
- Phase calculation: Follicular phase extends from menses onset through ovulation day; luteal phase spans from day after ovulation through day before next menses [58].
Hormone-Sensitive Populations: Screen for premenstrual dysphoric disorder (PMDD) and premenstrual exacerbation (PME) using prospective daily symptom monitoring (e.g., Carolina Premenstrual Assessment Scoring System) rather than retrospective recall, which demonstrates poor concordance with actual symptoms [58].
Oral Contraceptive Research Protocol
Comparative Framework for OC vs. Naturally Cycling Women:
OC Formulation Documentation: Record specific progestin type, androgenicity, and estrogen dose, as these differentially affect brain structure and function [56].
Duration of Use: Document length of OC use, as neurobiological adaptations may evolve over time [56].
Comparison Timepoints: When comparing OC users to naturally cycling women, measure outcomes in naturally cycling women during both early follicular (low hormone) and mid-luteal (high progesterone) phases to capture the dynamic range of endogenous hormone exposure [56].
Brain Assessment Protocols: For neuroimaging studies, employ resting-state functional connectivity (rsFC) to evaluate intrinsic network connectivity. Existing evidence suggests OC use mimics the connectivity pattern of high-progesterone states in naturally cycling women [56].
Conceptual Framework for OC Effects on Hormonal Status
Theoretical Models of Oral Contraceptive Effects [56]
Research Reagent Solutions for Menstrual Cycle Studies
Table 3: Essential Research Materials and Their Applications
| Reagent/Instrument | Primary Function | Research Application | Considerations |
|---|---|---|---|
| Urinary LH Tests | Detects luteinizing hormone surge | Estimating ovulation day for phase calculation | Practical balance between accuracy and feasibility [57] |
| Basal Body Thermometers | Measures resting body temperature | Detecting biphasic shift confirming ovulation | High-precision digital thermometers (0.01°C resolution) recommended [55] |
| Electronic Fertility Monitors | Integrates multiple fertility indicators | Tracking cycle phases in longitudinal studies | Example: Clearblue Easy Fertility Monitor [57] |
| Salivary Hormone Kits | Measures steroid hormones non-invasively | Frequent hormone sampling without phlebotomy | Particularly useful for progesterone assessment [60] |
| Menstrual Cycle Tracking Apps | Digital symptom and cycle logging | Large-scale epidemiological data collection | Examples: Natural Cycles, Flo App [53] [55] |
| Serum Hormone Assays | Quantifies estradiol, progesterone | Gold-standard hormone assessment | Required for validating other methods [60] |
Hormonal Dynamics Across the Natural Cycle and With OC Use
Analytical Approaches for Cycle Data
Statistical Modeling Recommendations
Advanced Analytical Framework:
Multilevel Modeling: Implement multilevel or random effects models to account for nested data (observations within cycles within individuals). These approaches require at least three observations per person to estimate random effects reliably [58].
Cycle Phase Coding: Convert cycle day to a relative phase indicator (e.g., early follicular, late follicular, periovulatory, early luteal, mid-luteal, late luteal) based on biological markers rather than calendar estimation alone [58].
Visualization Techniques: Create individual-level cycle profiles alongside group-level averages to visualize both within-person and between-person variability [58].
Handling Cycle Variability: For women with highly variable cycles, consider using a rolling window of recent cycle lengths rather than population norms for phase estimation [57].
Data Quality Considerations
Validation and Quality Control Measures:
Ovulation Detection Validation: Compare algorithm-detected ovulation days (from BBT or apps) with urinary LH surge tests in a subset of participants to establish method-specific error rates [55].
Hormone Assay Precision: Use low-coefficient of variation assays for hormone assessment, particularly for progesterone, which shows significant intraindividual variability between cycles [60].
Outlier Identification: Establish pre-registered criteria for excluding physiologically implausible cycles (e.g., <10 days or >90 days) while documenting exclusion decisions [55].
The evidence synthesized in this review underscores the critical importance of accounting for both inter- and intra-individual variability in menstrual cycle characteristics when designing studies comparing naturally cycling women and OC users. The substantial variability in cycle parameters—systematically influenced by age, ethnicity, and BMI—challenges the notion of a "standard" menstrual cycle and necessitates sophisticated methodological approaches.
For researchers and drug development professionals, these findings highlight several key considerations: First, the follicular phase represents the primary source of cycle length variability, suggesting that ovulation confirmation rather than calendar estimation is essential for precise phase determination. Second, oral contraceptives create a distinct endocrine environment that may mimic certain natural cycle phases while suppressing others, requiring careful theoretical framing when comparing these groups. Third, large-scale digital data collection platforms have revealed demographic patterns in cycle characteristics previously obscured in smaller homogeneous samples.
Moving forward, rigorous menstrual cycle research requires standardized protocols that embrace rather than ignore inherent variability, appropriate statistical models that account for nested data structures, and conceptual frameworks that acknowledge the complex neuroendocrine effects of synthetic hormones in OCs. By adopting these evidence-based approaches, researchers can generate more valid and reproducible findings that advance our understanding of female physiology and pharmacology across hormonal contexts.
The investigation into the psychological effects of oral contraceptives (OCs) represents a critical frontier in women's health research, requiring meticulous comparison between pharmacologically-treated and naturally cycling endocrine states. Current research reveals that hormonal status—whether naturally cycling (NC) or using oral contraceptives—significantly influences a spectrum of psychological well-being domains that extend beyond traditional mood assessments to include energy levels, feelings of attractiveness, risk-taking, and agitation [23]. The central challenge in this domain lies in accounting for substantial methodological variability across studies, including differences in OC formulations, duration of use, participant characteristics, and assessment tools. This review systematically synthesizes contemporary empirical evidence to elucidate the differential outcomes in mood, self-esteem, and depression metrics between OC users and naturally cycling women, providing researchers with both quantitative comparisons and methodological frameworks for advancing this field.
The complexity of this comparative analysis stems from what has been termed dimensional hormone sensitivity [13], an emerging framework positing that individuals differ not only in whether they are behaviorally sensitive to hormones but also in which specific hormonal changes trigger their symptoms (e.g., progesterone metabolite surges, estradiol withdrawal). This variability is further compounded by pharmacological heterogeneity across OC formulations, each creating a unique hormonal environment with respect to both exogenous hormones and suppression of endogenous production [13]. The following sections provide a comprehensive analysis of current findings, methodological considerations, and mechanistic insights to guide future research and clinical interpretation.
Quantitative Outcomes: Comparative Data Synthesis
Psychological Well-Being and Depression Metrics
Table 1: Comparative Psychological Outcomes Between Oral Contraceptive Users and Naturally Cycling Women
| Metric | OC Users vs. NC Women | Effect Size/Magnitude | Notes/Specific Conditions |
|---|---|---|---|
| Depression Risk (First 2 Years) | Increased | HR = 1.71, 95% CI: 1.55-1.88 [61] | Highest risk during initial use period |
| Lifetime Depression Risk | Increased | HR = 1.05, 95% CI: 1.01-1.09 [61] | Ever use associated with increased lifetime risk |
| Adolescent Initiation Impact | Increased | HR = 1.18, 95% CI: 1.12-1.25 [61] | Vulnerability period for later depression |
| Daily Depression Levels | Increased | Significant elevation [44] | Anti-androgenic OCs: higher in 3 cycle phases; Androgenic OCs: higher in 2 phases |
| Self-Esteem | Decreased | Significantly lower daily levels [44] | OC users had lower self-esteem across all three menstrual cycle phases |
| Happiness | Decreased | R²m = .004-.019 [23] | Lower overall ratings in OC users |
| Attractiveness Feelings | Decreased | R²m = .004-.019 [23] | Lower overall ratings in OC users |
| Energy Levels | Decreased | R²m = .004-.019 [23] | Lower overall ratings in OC users |
| Risk-Taking | Decreased | R²m = .004-.019 [23] | Lower overall ratings in OC users |
| Agitation Variability | Reduced | Lower day-to-day variability [23] | Supports "emotional blunting" hypothesis |
| Relaxation | Increased | R²m = .005-.01 [23] | OC users reported more relaxation |
| Sexual Desire | Increased | R²m = .005-.01 [23] | OC users reported higher sexual desire |
| Sleep Quality | Increased | R²m = .005-.01 [23] | OC users reported better sleep quality |
Neurocognitive and Fear Processing Metrics
Table 2: Neurocognitive and Fear Processing Outcomes
| Metric | OC Users vs. NC Women | Effect Size/Magnitude | Notes/Specific Conditions |
|---|---|---|---|
| Fear Extinction Recall | Impaired | Higher SCRs during extinction recall [62] | Current OC users showed impaired safety signal learning |
| Neural Correlates of Fear | Altered activation | Correlation with hippocampus, ACC, vmPFC activations [62] | Different neural circuitry engagement during fear processing |
| Cortisol Awakening Response | Blunted | 61% reduction relative to non-users [63] | Altered HPA axis dynamics in OC users |
| Working Memory | No significant difference | Not statistically significant [63] | No detected OC use effects on working memory |
| Implicit Negative Affect | Increased | More depressive-like pattern during active hormone intake [64] | Particularly in users reporting high depressive affect generally |
Experimental Protocols: Methodological Approaches
Prospective Daily Diary Methodology
The 28-day prospective daily study design represents a robust approach for capturing temporal dynamics in psychological well-being [23]. This methodology involves:
- Participant Allocation: 22 naturally cycling participants and 18 OC users reporting daily affective and physical symptoms across 28 days.
- Biospecimen Collection: Daily salivary samples analyzed for estradiol, progesterone, and testosterone levels using liquid chromatography-tandem mass spectrometry for specific hormone measurement [13].
- Psychological Assessment: Daily ratings across multiple well-being domains including mood, agitation, risk-taking, feelings of attractiveness, energy levels, relaxation, sexual desire, and sleep quality.
- Statistical Analysis: Comparison between groups using linear mixed models for averages, Levene's test for day-to-day variability, and network models to examine interrelationships between variables.
- Cycle Phase Analysis: Within naturally cycling participants, assessment of cycle phase effects and time-varying associations between hormones and psychological well-being using both person-centered mean and change scores.
This methodology successfully identified that the impact of sex hormones on psychological well-being varied significantly across cycle phases, with progesterone levels showing the largest effects [23].
Implicit vs. Explicit Affect Assessment
The distinction between implicit and explicit affective measures represents a critical methodological consideration in OC research [64]:
- Study Design: Counterbalanced repeated-measures design with 53 healthy women (mean age 19.9 years) tested during both active hormone intake and the washout week.
- Explicit Measures: Standard psychometric self-report tests including depression scales and mood inventories where participants consciously reflect on and report their emotional state.
- Implicit Measures: Assessment of automatic, unconscious affective processes through:
- Affect Misattribution Procedure: Measuring how participants' unconscious affect influences their judgments of neutral stimuli.
- Emotional Stroop Test: Assessing attentional bias toward emotional stimuli through response latency measures.
- Facial Emotion Processing: Evaluating perceptions and reactions to facial expressions of emotion.
- Key Finding: Disassociation between explicit and implicit measures—while self-report data suggested greater negative symptoms during the 'off' phase, implicit measures revealed a more depressive-like pattern during active hormone intake, particularly among women who generally experienced high depressive affect [64].
Population-Based Cohort Methodology
Large-scale epidemiological approaches provide crucial insights into depression risk associations [61]:
- Data Source: UK Biobank data from 264,557 women with linkage to health records including hospital inpatient data, primary care data, and cancer and death registries.
- Exposure Assessment: Touch-screen questionnaire data on OC use including age at initiation and discontinuation, with categorization of never, past, and current users.
- Outcome Ascertainment: Incidence of depression addressed via interviews, inpatient hospital, or primary care data using standardized diagnostic codes.
- Statistical Analysis: Multivariable Cox regression with OC use as a time-varying exposure to estimate hazard ratios for incident depression.
- Sibling Control Analysis: Examination of 7,354 sibling pairs to control for familial confounding and provide evidence for causal relationships.
- Key Finding: The first 2 years of OC use were associated with a 71% higher rate of depression compared to never users (HR = 1.71), with adolescent users particularly vulnerable to long-term risk [61].
Mechanistic Pathways: Biological Underpinnings of Psychological Effects
The psychological effects of oral contraceptives emerge from complex interactions between exogenous hormones and multiple physiological systems. The following diagram illustrates key mechanistic pathways through which OCs influence mood, fear processing, and stress response:
Diagram Title: Mechanistic Pathways of OC Effects on Psychology
Hormonal Suppression and Emotional Blunting
Oral contraceptives suppress endogenous production of estradiol, progesterone, and testosterone, creating a distinct endocrine environment compared to naturally cycling women [23]. This suppression has been linked to reduced day-to-day variability in psychological states including agitation, risk-taking, attractiveness perceptions, and energy levels [23]. This pattern of diminished emotional variability aligns with the emotional blunting hypothesis as a potential mechanism underlying OC-related side effects. The dimensional hormone sensitivity framework suggests that individual differences in sensitivity to specific hormonal changes—whether to synthetic hormone exposure, endogenous hormone suppression, or particular metabolites—may explain why only a subset of women experience significant psychological effects [13].
Stress Axis Dysregulation
OC users demonstrate significant alterations in hypothalamic-pituitary-adrenal (HPA) axis function, evidenced by a 61% reduction in cortisol awakening response (CAR) compared to non-users [63]. This blunted cortisol dynamics may represent disrupted stress response capacity, potentially contributing to increased vulnerability to depressive disorders. The mechanism may involve both increased cortisol binding globulin (CBG) and direct effects of ovarian suppression on HPA regulation [63]. Given the established relationship between HPA axis dysfunction and depression, this pathway represents a plausible biological mechanism linking OC use with mood disturbances in susceptible individuals.
Fear Processing and Neural Circuitry Alterations
Emerging evidence indicates that OC use affects neural systems underlying fear regulation and emotional memory [62]. Current OC users show impaired fear extinction recall and altered activation in fear regulation networks including the hippocampus, anterior cingulate cortex, and ventromedial prefrontal cortex [62]. Notably, these effects may persist beyond discontinuation, as past OC users in low-estradiol states (early follicular phase) exhibited fear responses similar to current users [62]. The progestin type and estrogen dose appear to moderate these effects, with exploratory analyses linking higher ethinyl estradiol doses and specific progestins to greater fear regulation impairment [13].
Research Toolkit: Essential Methodological Components
Table 3: Research Reagent Solutions for OC Psychological Effects Research
| Reagent/Instrument | Function/Application | Specific Examples from Literature |
|---|---|---|
| Salivary Hormone Assays | Measurement of estradiol, progesterone, testosterone, and synthetic hormones | Liquid chromatography-tandem mass spectrometry for specific measurement of E2, EE, P4, and testosterone in saliva [13] |
| Daily Diary Measures | Ecological momentary assessment of psychological states | Custom daily ratings of happiness, attractiveness, risk-taking, energy, agitation, relaxation, sexual desire, sleep quality [23] |
| Implicit Affect Tasks | Assessment of automatic, unconscious affective processes | Affect Misattribution Procedure, Emotional Stroop Test, facial emotion processing tasks [64] |
| Fear Conditioning Paradigms | Evaluation of fear learning and extinction processes | Two-day fear protocol (day 1: fear conditioning/extinction learning; day 2: extinction recall/fear renewal) with skin conductance response measurement [62] |
| Cortisol Awakening Response | Assessment of HPA axis dynamics | Home-sampling of saliva at awakening, 30, and 45 minutes post-awakening to characterize CAR [63] |
| Population Registry Data | Large-scale epidemiological analysis | UK Biobank data linkage with hospital inpatient, primary care, and prescription records [61] |
Discussion: Integration and Research Implications
The synthesized evidence indicates that oral contraceptive use is associated with a complex pattern of both potentially adverse and beneficial psychological effects, with significant individual variability moderated by formulation characteristics, timing of use, and personal susceptibility factors.
The differential outcomes between OC users and naturally cycling women extend beyond depression risk to encompass multiple well-being domains. The consistent observation of reduced self-esteem across menstrual cycle phases in OC users [44] warrants particular attention, as this effect persists regardless of progestin androgenicity. Simultaneously, benefits in relaxation, sexual desire, and sleep quality [23] highlight the importance of multidimensional assessment rather than singular focus on depression metrics.
Methodologically, the dissociation between implicit and explicit affect measures [64] underscores the limitation of relying exclusively on self-report instruments. The finding that implicit measures detected more depressive-like patterns during active hormone intake while participants self-reported worse mood during the withdrawal phase suggests complex interactions between conscious awareness of mood states and neurobiological effects of synthetic hormones.
From a neurobiological perspective, the convergence of evidence across stress axis dysregulation [63], fear processing alterations [62], and emotional blunting [23] suggests that OCs enact multifaceted changes in central nervous system function. These effects likely reflect both the direct actions of synthetic hormones on widespread steroid receptors and indirect effects through suppression of endogenous ovarian hormone production.
Research Gaps and Future Directions
Critical gaps remain in understanding the mechanisms underlying individual differences in psychological sensitivity to OCs. Future research should prioritize:
- Longitudinal neuroimaging studies tracking neural changes during OC initiation and discontinuation
- Pharmacological specificity studies directly comparing different progestin types and estrogen doses
- Gene-environment interactions investigating genetic modifiers of psychological sensitivity to hormonal manipulations
- Development of predictive biomarkers to identify women at risk for adverse psychological effects before prescription
The dimensional hormone sensitivity framework [13] offers a promising approach for advancing this field by recognizing that women differ not only in whether they are sensitive to hormonal exposures but also in which specific hormonal triggers elicit symptoms. Adopting this personalized medicine perspective will ultimately enable more precise contraceptive prescribing that maximizes benefits while minimizing adverse psychological effects for individual women.
The investigation into the differential effects of hormonal profiles on fear extinction and emotion regulation represents a critical frontier in neuropsychopharmacology. This guide systematically compares the psychophysiological and neural outcomes between oral contraceptive (OC) users and naturally cycling individuals, framing these differences within the broader research on cognitive emotion regulation strategies like cognitive reappraisal. Evidence indicates that synthetic hormones in OCs can significantly alter the neural circuitry responsible for processing and extinguishing fear, with implications for vulnerability to anxiety-related disorders and the development of targeted therapeutics [62]. This analysis provides a structured comparison of experimental data, methodologies, and underlying neurobiology for researchers and drug development professionals.
Tabular Comparison of Key Findings
The table below synthesizes core experimental findings comparing oral contraceptive users and naturally cycling individuals in the context of fear learning and emotion regulation.
Table 1: Comparative Summary of Fear Extinction and Emotion Regulation Profiles
| Experimental Measure | Oral Contraceptive Users | Naturally Cycling Individuals (Pre-ovulatory) | Key Implications |
|---|---|---|---|
| Fear Extinction Memory Recall | ✓ Impaired recall, evidenced by higher skin conductance responses (SCRs) during recall tests [62] | ✓ Robust recall, evidenced by lower SCRs [62] | Suggests OC use may be a vulnerability factor for inadequate long-term fear inhibition. |
| Associated Neural Activity during Recall | ✓ Correlated with activations in the hippocampus, dorsal-rostral ACC, and vmPFC [62] | ✓ Expected engagement of a vmPFC-hippocampal circuit supportive of extinction memory [65] | Indicates potential compensatory neural mechanisms in OC users to support a compromised extinction trace. |
| Endogenous Estradiol (E2) Level | ✓ Low, suppressed levels due to HPO axis suppression [66] [62] | ✓ High levels during the pre-ovulatory phase [62] | Posits low E2 as a potential mechanism underlying impaired extinction recall. |
| Response to Cognitive Reappraisal | Limited direct studies; cognitive reappraisal is an effective emotion regulation strategy that alters conditioned fear responses [67] | Limited direct studies; cognitive reappraisal reliably attenuates conditioned fear, though effect may vary with cycle phase [67] [65] | Highlights cognitive regulation as a potential intervention strategy across groups. |
Detailed Experimental Protocols
To ensure reproducibility and critical evaluation, this section outlines the standard methodologies used to generate the comparative data.
Fear Conditioning, Extinction, and Recall Protocol
This multi-day paradigm is the gold standard for investigating fear extinction memory [62].
Day 1: Acquisition & Extinction Learning:
- Acquisition: Participants are presented with a neutral conditioned stimulus (CS+, e.g., a specific colored shape) that is repeatedly paired with a mildly aversive unconditioned stimulus (US, e.g., a mild electric shock or loud tone). A second stimulus (CS-) is never paired with the US. Through this associative learning, the CS+ acquires the property to elicit a conditioned fear response (CR), measured by Skin Conductance Response (SCR).
- Extinction Learning: Subsequently, the CS+ is repeatedly presented without the US. This leads to a new learning process where the CS+ is no longer predictive of danger, and the fear response gradually declines. The formation of an extinction memory is tracked via the reduction in SCR.
Day 2: Extinction Recall (Retrieval):
- After a 24-hour delay, participants are re-exposed to the CS+ and CS- in a context similar to the extinction learning context. The successful retrieval of the extinction memory is measured by low SCRs to the CS+. Impaired extinction recall is defined by significantly higher SCRs to the CS+ in one group (e.g., OC users) compared to another (e.g., naturally cycling women) [62].
Cognitive Reappraisal Protocol for Conditioned Fear
Cognitive reappraisal is a cognitive emotion regulation strategy that involves reinterpreting the meaning of a stimulus to alter its emotional impact [67]. In fear conditioning studies:
- Training: Before or during the experiment, participants are trained to use specific cognitive strategies. For instance, they may be instructed to psychologically distance themselves from the CS+ or to reappraise the threat by telling themselves that the stimulus is safe and cannot cause harm.
- Application: During the presentation of the CS+, participants actively employ the rehearsed reappraisal tactics.
- Measurement: The efficacy of reappraisal is quantified by a reduction in the fear response (SCR, amygdala activity) compared to a control condition where participants simply respond naturally to the stimuli. Systematic reviews confirm that this strategy can successfully attenuate conditioned fear responses [67].
Hormonal Assessment and Group Stratification
- Group Stratification: Participants are typically divided into groups based on hormonal status. Common groupings include: 1) Men (as a non-cycling control), 2) Current OC users, 3) Naturally cycling women in the early follicular phase (low endogenous E2), and 4) Naturally cycling women in the pre-ovulatory phase (high endogenous E2) [62]. Some studies also consider past OC use history.
- Hormonal Assays: Hormone levels, particularly estradiol, are confirmed through analysis of fasting blood samples. This objective measure validates group assignment and allows for correlational analyses with behavioral and neural data [66].
Visualizing the Neural Circuitry of Fear Regulation
The following diagram illustrates the key brain regions implicated in fear conditioning, extinction, and their modulation by hormones and cognitive strategies like reappraisal.
Diagram 1: Neurocircuitry of Fear and its Regulation. Key regions include the amygdala (fear expression), ventromedial Prefrontal Cortex (vmPFC; fear inhibition), hippocampus (contextual memory), dorsal Anterior Cingulate Cortex (dACC; conflict monitoring), and dorsolateral PFC (dlPFC; cognitive regulation). Hormonal states, like estradiol, modulate this circuitry.
Visualizing the Experimental Workflow
This diagram outlines the standard sequence and timing of a typical fear extinction recall study involving oral contraceptive users and naturally cycling participants.
Diagram 2: Fear Extinction Recall Experimental Timeline. The protocol spans two days, measuring fear learning, within-session extinction, and long-term extinction memory recall after a delay. HC=Hormonal Contraceptive, NC=Naturally Cycling.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Materials and Methods for Fear Extinction Research
| Item / Reagent | Primary Function in Research Context |
|---|---|
| Skin Conductance Response (SCR) | The primary psychophysiological measure of arousal and fear, recorded via electrodes on the skin. It quantifies the conditioned fear response to the CS+ and CS- [62]. |
| Functional Magnetic Resonance Imaging (fMRI) | Non-invasive neuroimaging technique used to measure brain activity by detecting changes in blood flow. It localizes neural correlates (e.g., in vmPFC, amygdala, hippocampus) of fear extinction and recall [62]. |
| Hormonal Assay Kits | Used for the quantification of serum hormone levels (e.g., estradiol, progesterone) from fasting blood samples. This objectively verifies participant group status (OC user, follicular phase, etc.) [66]. |
| Standardized Fear Conditioning Software | Software platforms (e.g., Presentation, E-Prime) that precisely control stimulus presentation and synchronize with physiological recording equipment, ensuring experimental rigor and reproducibility. |
| Oral Contraceptives (as variable) | Not a reagent, but a key group stratification variable. Studies often specify the type (combined vs. progestin-only) to understand differential effects on hormonal axes and fear circuitry [66] [62]. |
Oral contraceptive pills (OCPs) are a cornerstone of reproductive healthcare, broadly categorized into combined estrogen-progesterone pills (COCs) and progesterone-only pills (POPs), also known as "mini-pills" [1]. The type and generation of the progestin component in these formulations are critical variables that can significantly influence experimental outcomes in endocrine and physiological research. COCs suppress hypothalamic-pituitary-ovarian (HPO) axis function, leading to decreased secretion of gonadotropins and lower concentrations of endogenous sex steroids like estradiol and progesterone [66]. In contrast, low-dose POPs may not consistently suppress HPO axis function, resulting in endogenous sex steroid profiles that more closely mimic those of naturally menstruating individuals [66]. This fundamental pharmacological difference necessitates careful stratification in research design to avoid confounding results and to enable accurate interpretation of data related to female physiology.
The importance of accounting for OC formulation is particularly evident when measuring biomarkers that respond to external stressors. Recent research demonstrates that the exogenous hormones in combined HCs maintain already suppressed HPO axis function during periods of low energy availability, whereas low-dose progestin-only HC users and naturally menstruating females experience significant decreases in concentrations of estradiol and total testosterone under the same conditions [66]. This review provides a comparative analysis of OC formulations, presents experimental data highlighting their differential effects, and offers methodological guidance for researchers and drug development professionals working in this field.
Quantitative Data Comparison: Hormonal and Physiological Metrics
Hormone Profiles Across OC Types and Natural Cycling
Table 1: Hormonal Biomarker Responses to Low Energy Availability (LEA) by Contraceptive Status
| Hormonal Biomarker | Naturally Menstruating Females | Combined HC Users | Low-Dose Progestin-Only HC Users |
|---|---|---|---|
| Estradiol | Significant decrease [66] | Remained stable [66] | Significant decrease [66] |
| Total Testosterone | Significant decrease [66] | Remained stable [66] | Significant decrease [66] |
| IGF-1 | Significant decrease [66] | Significant decrease [66] | Significant decrease [66] |
| Triiodothyronine (T3) | Significant decrease [66] | Significant decrease [66] | Significant decrease [66] |
| Leptin | Significant decrease [66] | Significant decrease [66] | Significant decrease [66] |
| Cortisol | Remained statistically unchanged [66] | Remained statistically unchanged [66] | Remained statistically unchanged [66] |
Cardiovascular Fluctuation Metrics Across the Menstrual Cycle
Table 2: Cardiovascular Amplitude Across Menstrual Cycle by Contraceptive Status
| Cardiovascular Parameter | Naturally Cycling Participants (n=9,968) | Birth Control Pill Users (n=1,661) | Statistical Significance |
|---|---|---|---|
| RHRamp (Amplitude in BPM) | 2.73 BPM (±1.95) [68] | 0.28 BPM (±1.94) [68] | p < 0.001 [68] |
| RMSSDamp (Amplitude in ms) | 4.65 ms (±6.90) [68] | -0.51 ms (±6.70) [68] | p < 0.001 [68] |
| Percentage with Positive RHRamp | 93.6% [68] | Not reported | Not applicable |
| Percentage with Positive RMSSDamp | 80.6% [68] | Not reported | Not applicable |
Experimental Protocols and Methodologies
Protocol 1: Hormonal Biomarker Response to Low Energy Availability
Study Population and Design: A 46-week observational study investigated the influence of HC use on hormonal biomarkers responding to diet and physical training-induced low energy availability. Thirty-six female physique athletes were stratified into three groups: combined HC users (n=11), low-dose progestin-only HC users (n=11), and naturally menstruating females (n=14). All participants prepared for a physique competition, voluntarily restricting energy intake while participating in resistance training to maintain lean mass and aerobic training to increase energy expenditure [66].
Measurement Timepoints: Baseline measurements were taken before commencing the pre-competition period (pre). Follow-up measurements occurred after approximately 23 weeks of dietary restriction and physical training (post), and again after approximately 23 weeks of recovery (recovery) [66].
Biomarker Analysis: Hormones (estradiol, total testosterone, IGF-1, cortisol, triiodothyronine, and leptin) were analyzed from fasting blood samples. Body composition metrics including body mass, fat mass, fat-free mass, and body fat percentage were measured using bioimpedance [66].
Protocol 2: Cardiovascular Fluctuation Across Menstrual Cycle
Study Population and Data Collection: This study developed a novel measure to quantify and investigate cardiovascular fluctuation across the menstrual cycle using wrist-worn biometric data from 11,590 participants and 45,811 menstrual cycles. Participants were stratified into naturally cycling (n=9,968) and birth control pill user (n=1,661) cohorts. Data was collected from global participants, with the majority from the United States (69.9%), Great Britain (8.0%), and Australia (4.2%) [68].
Cardiovascular Amplitude Calculation: The cycle cardiovascular amplitude for resting heart rate (RHR) was defined as the mean value on days 2-8 (7-day mean centered on day 5, the population RHR minimum) subtracted from the mean value of the final 7 days (centered on day 26, the population RHR maximum). RHRamp was defined as each participant's mean cycle cardiovascular amplitude across all eligible cycles. Similarly, for heart rate variability (RMSSD), amplitude was calculated as the mean value on days 2-8 less the mean value from the final 7 days [68].
Statistical Analysis: Population-level generalized additive mixed models (GAMM) revealed a significant relation (p < 0.001) between the day of the menstrual cycle and metric offset from the cycle mean for both RHR and RMSSD in both unadjusted and adjusted models. Differences between cohorts were tested for statistical significance using generalized linear models (GLMs) [68].
Signaling Pathways and Physiological Mechanisms
OC Effects on Endocrine Pathways
Cardiovascular Fluctuation Measurement Workflow
Cardiovascular Amplitude Analysis
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Materials for OC Formulation Studies
| Research Material | Specification/Function | Application Example |
|---|---|---|
| Wrist-Worn Wearable Device | Photoplethysmography (PPG) capabilities for continuous cardiovascular monitoring [68] | Collection of RHR and RMSSD data across menstrual cycles [68] |
| Fasting Blood Collection Tubes | Standardized tubes for hormone stability during collection and transport [66] | Analysis of estradiol, testosterone, IGF-1, cortisol, T3, and leptin [66] |
| Enzyme-Linked Immunosorbent Assay Kits | Validated kits for specific hormone quantification [66] | Measurement of hormonal biomarkers in serum/plasma samples [66] |
| Bioimpedance Analyzer | Multi-frequency analyzer for body composition assessment [66] | Measurement of fat mass, fat-free mass, and body fat percentage [66] |
| Menstrual Cycle Tracking Application | Validated digital platform for cycle day and bleeding documentation [68] | Precise alignment of physiological data with menstrual cycle phase [68] |
| Hormonal Contraceptive Verification | Protocol for verifying OC type, formulation, and adherence [66] | Accurate participant stratification by OC formulation and generation [66] |
The formulation-specific effects of oral contraceptives present both challenges and opportunities for researchers and drug development professionals. The data clearly demonstrate that failing to account for OC type and generation can lead to significant misinterpretation of experimental results, particularly in studies investigating endocrine function, cardiovascular physiology, and metabolic responses to interventions. The differential effects of combined HCs versus progestin-only HCs on hormonal biomarkers during low energy availability, and their profound impact on cardiovascular fluctuations across the menstrual cycle, underscore the necessity of careful participant stratification in research design.
For drug development professionals, these findings highlight the importance of considering OC usage when designing clinical trials, particularly for medications with hormonal interactions or those metabolized through pathways affected by synthetic steroids. Future research should continue to elucidate the formulation-specific effects of different OC generations and develop standardized methodologies for accounting for these variables in both basic science and clinical applications. By optimizing data interpretation through careful consideration of OC type and generation, researchers can advance our understanding of female physiology and improve the development of targeted therapeutics for all women, regardless of contraceptive status.
Managing Attrition and Expectancy Effects in Long-Term Studies of Hormonal Interventions
Long-term studies on hormonal interventions, such as those investigating oral contraceptives (OCs) and menopausal hormone therapy (MHT), are essential for understanding their lasting health effects. However, their validity is threatened by two major methodological challenges: attrition (the loss of participants over time) and expectancy effects (where participants' or researchers' expectations influence outcomes). Successfully managing these challenges is critical for producing reliable data on the complex interplay between synthetic and naturally cycling hormone profiles. This guide compares methodological approaches and provides structured experimental data to aid researchers in optimizing future study designs.
Methodological Approaches for Managing Attrition
Attrition can introduce significant bias, particularly if dropouts are not random but are related to treatment side effects or burden. The following strategies, derived from major studies, are critical for retention.
Evidence-Based Retention Strategies from Major Cohorts
Table 1: Attrition Predictors and Mitigation Strategies in Longitudinal Hormonal Studies
| Predictor of Attrition | Evidence from Studies | Recommended Mitigation Strategy |
|---|---|---|
| Older Age & Worsening Health | In the WHI 80+ cohort, attrition was higher among older, less healthy participants [69]. | Implement tailored retention protocols for older and clinically complex participants, which may include at-home visits, simplified procedures, and dedicated health check-ins. |
| Lower Education & Minority Status | The WHI 80+ cohort found retained participants were "more likely to be white, educated, and healthier at baseline" [69]. | Develop targeted, culturally sensitive communication and build trust with underserved communities from the study's outset. |
| Study Duration and Burden | WHI retention rates dropped during re-enrollment periods (74% and 83%) compared to initial phases (90%) [69]. | Design streamlined follow-up protocols for extension phases. Use mixed-mode data collection (e.g., phone, mail, online) to reduce participant burden. |
| Lack of Ongoing Engagement | N/A (Inferred from standard practice) | Establish a participant advisory board, provide regular newsletters with study updates and findings, and offer modest, timely compensation for time and effort. |
Experimental Protocol for Monitoring and Reducing Attrition
The Women's Health Initiative (WHI) provides a robust model for managing long-term follow-up.
- Study Design: The WHI included both clinical trial and observational study components, following postmenopausal women for over two decades [69].
- Re-enrollment Procedures: The study featured formal re-enrollment campaigns at the end of initial funding periods. This required renewed verbal consent and allowed for the re-recruitment of participants into extension phases [69].
- Data Collection Methods: To maintain contact and collect data with minimal burden, the WHI utilized annual follow-up visits conducted via mail or telephone. These visits collected updated health information and outcome data [70] [69].
- Statistical Analysis of Attrition: Researchers should:
- Compare baseline characteristics of retained participants versus those lost to follow-up using chi-square tests and analyses of variance [69].
- Use Cox proportional hazards models to identify baseline factors (e.g., age, education, health status) that independently predict attrition risk [69].
- Employ statistical techniques like multiple imputation or inverse probability weighting to account for missing data in final analyses.
The diagram below illustrates a proactive protocol for attrition management, integrating lessons from major studies.
Methodological Approaches for Managing Expectancy Effects
Expectancy effects, including placebo effects and observer bias, are a major concern in trials where subjective endpoints (e.g., mood, cognitive symptoms) are common.
Blinding and Placebo Control in Hormonal Intervention Trials
The gold standard for controlling expectancy effects is the double-blind, placebo-controlled randomized trial.
- The KEEPS-Cog Trial: This study investigated the cognitive effects of MHT initiated early in menopause. Participants were randomly assigned to one of three groups: oral conjugated equine estrogens (oCEE), transdermal estradiol (tE2), or placebo pills and patch. Both active treatment groups also received micronized progesterone. The use of matching placebos (pills and patches) was critical for maintaining blinding and isolating the specific biological effects of the hormones from non-specific expectancy effects [71] [72].
- The WHI Hormone Therapy Trials: Similarly, the WHI compared active HT (0.625mg/d CEE with or without MPA) against matching placebos. The blinding was maintained for the duration of the active intervention phase, which averaged 5.4 to 7.1 years [70].
Table 2: Quantitative Cognitive Outcomes from Blinded MHT Trials
| Study & Cohort | Intervention | Global Cognitive Function | Working Memory | Executive Function | Key Finding on Expectancy |
|---|---|---|---|---|---|
| KEEPS (Ages 50-54 at start) [71] [72] | oCEE vs Placebo | No significant difference | No significant difference | No significant difference | No cognitive benefit or harm was found for either MHT formulation versus placebo, demonstrating that active treatment did not produce effects beyond expectancy. |
| KEEPS (Ages 50-54 at start) [71] [72] | tE2 vs Placebo | No significant difference | No significant difference | No significant difference | |
| WHI (Ages 65-79 at start) [70] | CEE-based vs Placebo | -0.081 SD* | -0.070 SD* | -0.054 SD* | The small but significant decrements in older women, detected under blinded conditions, represent true biological effects rather than negative expectations. |
*SD = Standard Deviation units. Negative values indicate a relative decrement compared to placebo.
Experimental Protocol for a Blinded Hormonal Intervention Trial
The KEEPS-Cog trial provides a detailed methodological blueprint.
- Participant Recruitment: Enroll recently postmenopausal women (within 36 months of last menstrual period) who are in good general and cardiovascular health. This homogenizes the sample and reduces confounding [71] [72].
- Randomization and Blinding:
- Participants are randomly assigned to groups (e.g., oCEE, tE2, or placebo).
- Prepare identical-looking active and placebo pills, and active and placebo patches. All participants receive both a pill and a patch to maintain the blind. For example, the tE2 group would use an active patch and a placebo pill, while the oCEE group would use an active pill and a placebo patch. The placebo group receives both a placebo pill and a placebo patch [72].
- The allocation sequence should be concealed from both participants and investigators (double-blind).
- Treatment Administration:
- Active Oral (oCEE): 0.45 mg/day conjugated equine estrogens.
- Active Transdermal (tE2): 50 μg/day 17β-estradiol patch.
- Progestin Component: All participants with a uterus also receive cyclical micronized progesterone (200 mg/day for 12 days/month) to protect the endometrium. This can be implemented in an open-label manner for the active groups, though it presents a partial challenge to blinding [72].
- Placebo: Matching placebo pills and patches.
- Outcome Assessment: Conduct annual cognitive assessments using a standardized battery. To further minimize bias, certify outcome assessors and use automated or computer-based scoring where possible. The KEEPS Continuation study used latent growth models to analyze cognitive trajectories, controlling for covariates [72].
The workflow for implementing and validating blinding is summarized below.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Hormonal Intervention Studies
| Item | Function in Research | Example from Literature |
|---|---|---|
| Oral Conjugated Equine Estrogens (oCEE) | The active intervention in one arm of a trial; used to study the effects of oral estrogen. | Premarin (0.45 mg/d) was used in the KEEPS trial [72]. |
| Transdermal 17β-Estradiol (tE2) | The active intervention in a separate arm; allows study of non-oral estrogen delivery. | Climara patch (50 μg/d) was used in the KEEPS trial [72]. |
| Medroxyprogesterone Acetate (MPA) | A progestin added to estrogen therapy in women with a uterus to prevent endometrial hyperplasia. | Used in the WHI trial (2.5 mg/d) combined with CEE [70]. |
| Micronized Progesterone | A bioidentical progesterone used for endometrial protection, often associated with fewer side effects. | Prometrium (200 mg/d for 12 days/month) was used in the KEEPS trial [72]. |
| Matching Placebos | The critical control for blinding and isolating biological from expectancy effects. | The KEEPS and WHI trials used placebo pills and patches identical to the active interventions [70] [72]. |
| Telephone Interview for Cognitive Status (TICS-m) | A validated telephone-administered test to assess global cognitive function remotely. | Used for annual cognitive assessments in the WHI Memory Study (WHIMSY) to reduce attrition [70]. |
| Enzyme-Linked Immunosorbent Assay (ELISA) Kits | To quantify hormone levels (e.g., estradiol, testosterone, cortisol) and inflammatory biomarkers (e.g., TNF-α, IL-6) from blood or saliva. | Used to measure inflammatory cytokines (TNF-alpha) and cortisol in studies of hormonal contraceptives and stress [73]. |
Empirical Evidence and Comparative Analysis: Validating Neurobiological and Psychological Outcomes
The burgeoning field of neuroendocrinology increasingly focuses on understanding how synthetic hormones modulate brain architecture. This guide provides an objective, data-driven comparison of brain morphometric findings in oral contraceptive (OC) users versus naturally cycling (NC) women, a crucial area for researchers and drug development professionals investigating the central nervous system effects of hormonal interventions. It synthesizes contemporary neuroimaging methodologies and findings to present a clear comparison of structural outcomes, grounded in a broader thesis of understanding how exogenous hormone profiles differ from naturally cycling endocrine states.
Key Volumetric Differences Between OC Users and NC Women
The following table summarizes the primary brain structural differences observed between OC users and NC women, based on current neuroimaging literature. These findings should be interpreted as a summary of emerging trends, as the field requires further large-scale, longitudinal confirmation.
Table 1: Comparative Summary of Brain Volumetric Findings
| Brain Structure/Region | Trend in OC Users vs. NC Women | Consistency Across Studies | Notes and Methodological Considerations |
|---|---|---|---|
| Global Cortical Volume | Mixed/No Clear Direction | Low | Findings are heterogeneous; may depend on OC formulation and duration of use. |
| Hippocampal Volume | ↓ (Reduction) | Medium | One of the more consistently reported, though not universal, findings. |
| Hypothalamic Volume | ↓ (Reduction) | Medium | Linked to the suppression of the endogenous hypothalamic-pituitary-gonadal axis. |
| Lateral Ventricles | ↑ (Enlargement) | Low | Often reported as an inverse correlate of parenchymal volume loss. |
| Prefrontal Cortex | Mixed | Low | Some studies report regional thinning, others find no significant difference. |
| Amygdala | Mixed | Low | Volume findings are inconsistent; functional reactivity may be more affected. |
| Cerebellum | Mixed | Low | Limited number of studies; potential effect requires further investigation. |
Methodological and Demographic Factors
The interpretation of comparative data is profoundly influenced by methodological and participant factors. The table below outlines key variables that must be considered when designing studies or evaluating literature.
Table 2: Critical Methodological and Cohort Factors in OC/NC Research
| Factor | Impact on Volumetric Results | Recommendation for Researchers |
|---|---|---|
| OC Formulation | Androgen index, progestin generation, and estrogen dose can differentially impact brain structure. | Always document and report the specific OC formulation(s) used by the cohort. |
| Duration of Use | Plastic changes may be dose- and time-dependent; acute vs. chronic effects likely differ. | Stratify participants by duration of use (e.g., <1 year, 1-5 years, >5 years). |
| Menstrual Cycle Phase | NC women show natural volumetric fluctuations across the cycle; OC users have suppressed cycles. | Phase-match NC women for scanning or use a longitudinal design to account for intra-individual variance. |
| Age of OC Initiation | The adolescent brain may be more susceptible to hormonal manipulation. | Document age of menarche and age of OC initiation as critical covariates. |
| MRI Segmentation Tool | Different algorithms (e.g., Freesurfer, CAT12, SynthSeg) can yield varying volumetric estimates [74]. | Use the same validated software pipeline for all participants and disclose the tool used. |
Experimental Protocols & Methodologies
Standardized Neuroimaging Data Acquisition Protocol
A robust magnetic resonance imaging (MRI) protocol is fundamental for reliable morphometric analysis. The following represents a standard acquisition protocol suitable for a comparative study.
- Scanner: 3T MRI scanner (e.g., Siemens Skyra, Philips Achieva, GE Discovery).
- Sequence - Structural T1-weighted:
- Sequence Type: 3D Magnetization Prepared Rapid Acquisition Gradient Echo (MPRAGE) or 3D Bravo.
- Rationale: Provides high-resolution anatomical images essential for precise tissue classification and volumetric measurement.
- Typical Parameters: Repetition Time (TR) = ~2300 ms; Echo Time (TE) = ~2.98 ms; Inversion Time (TI) = 900 ms; Flip Angle = 9°; Voxel Size = 1 mm³ isotropic; Matrix Size = 256 x 256.
- Sequence - T2-weighted/FLAIR:
- Rationale: Used to identify and exclude participants with significant neurological abnormalities or white matter hyperintensities that could confound results.
- Quality Control: Implement real-time and post-hoc quality assessment for motion artifact, signal-to-noise ratio, and contrast-to-noise ratio. Exclusion criteria should be defined a priori.
Volumetric Processing and Analysis Workflow
The processing of acquired images follows a multi-stage, automated pipeline. The reliability of these tools, including in challenging conditions, has been demonstrated in recent studies [74].
Figure 1: Automated pipeline for brain volume analysis from structural MRI.
- Preprocessing: Raw T1-weighted images undergo skull-stripping to remove non-brain tissue, bias field correction for intensity inhomogeneity, and spatial normalization to a standard template space (e.g., MNI152).
- Tissue Segmentation: The brain is classified into probabilistic maps of gray matter (GM), white matter (WM), and cerebrospinal fluid (CSF).
- Regional Parcellation: Using a pre-defined brain atlas (e.g., Desikan-Killiany, AAL), the segmented GM and WM are subdivided into specific anatomical regions-of-interest (ROIs). The use of flexible atlases can help maintain anatomical hierarchy across studies [75].
- Volumetric Calculation & Analysis: The volume of each ROI is calculated. For group comparisons (OC vs. NC), analysis of covariance (ANCOVA) is typically employed, with total intracranial volume (ICV), age, and other relevant demographics as nuisance covariates.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Hormonal Neuroimaging Research
| Item | Function/Application in OC/NC Research | Specification Notes |
|---|---|---|
| High-Contrast Phantoms | Quality assurance and calibration of MRI scanners to ensure longitudinal and cross-site measurement stability. | Geometrically defined structures with known volumes, filled with substances mimicking tissue relaxation properties. |
| Automated Segmentation Software | Processing of T1-weighted MRI data to derive volumetric measurements of brain structures. | Examples include: Freesurfer, CAT12 [74], SPM, FSL. Choice affects results; tool must be consistent. |
| Flexible Brain Atlas | Standardized definition of regions-of-interest (ROIs) for volumetric analysis, allowing for combination/division of structures. | Enables consistent ROI definition across labs while accommodating specific research needs (e.g., hippocampal subfields) [75]. |
| Hormonal Assay Kits | Quantification of serum/plasma levels of hormones (e.g., estradiol, progesterone, SHBG) to validate participant groups. | Essential for confirming endocrine status in NC women (cycle phase) and verifying hormone suppression in OC users. |
| Biobanking Supplies | Secure, long-term storage of biological samples (e.g., blood, DNA) for future correlated genetic or biomarker analyses. | Includes barcoded tubes, -80°C freezers, and a linked electronic database for sample management. |
- Hormonal context: Introduction to hormonal profiles and brain connectivity relationships.
- Functional connectivity: Comparative analysis of brain network alterations using tables.
- Cognitive effects: Examination of emotional and cognitive processing differences.
- Methodology: Detailed experimental protocols and measurement techniques.
- Signaling pathways: Visual diagrams of neuroendocrine mechanisms.
- Research tools: Essential reagents and resources for experimental replication.
Resting-State Functional Connectivity: Mimicry of Luteal Phase Patterns in OC Users
The study of resting-state functional connectivity (rs-FC) has emerged as a crucial approach for understanding the intrinsic organization of the human brain and how it is modulated by various factors, including sex hormones. This comparative guide examines the neurobiological effects of oral contraceptives (OCs) in relation to naturally cycling hormonal profiles, focusing specifically on whether OC use creates neural patterns that mimic the luteal phase of the menstrual cycle. OCs, used by over 100 million women worldwide, introduce synthetic hormones that suppress the natural hypothalamic-pituitary-gonadal axis, fundamentally altering the endocrine environment [76] [77]. While the reproductive effects of OCs are well-established, their impact on brain function and connectivity represents a burgeoning area of research with significant implications for women's cognitive health and medication development.
The menstrual cycle in naturally cycling women is characterized by fluctuating levels of estradiol and progesterone, with the luteal phase featuring elevated levels of both hormones compared to the follicular phase. These hormonal variations have demonstrated effects on brain structure, functional connectivity, and cognitive processing [78] [79]. Simultaneously, research has revealed that synthetic hormones in OCs cross the blood-brain barrier and impact hormone-sensitive brain regions, potentially altering emotional regulation, memory processes, and intrinsic brain network dynamics [78] [42]. This guide systematically compares the current experimental evidence regarding rs-FC patterns across these hormonal conditions, providing researchers and pharmaceutical professionals with objective data to inform future investigation and therapeutic development.
Comparative Analysis of Functional Connectivity Patterns
Default Mode and Executive Control Networks
The default mode network (DMN) and executive control network (ECN) represent two critical brain systems that display notable sensitivity to hormonal influences. Research by Petersen et al. demonstrated that both menstrual cycle phase and OC use significantly modulate connectivity within these networks [76] [77]. Their investigation revealed that naturally cycling women in the luteal phase (characterized by high endogenous progesterone and estradiol) showed altered connectivity in the anterior cingulate cortex (ACC) and left middle frontal gyrus compared to women in the follicular phase (low hormone phase). Interestingly, OC users during their active pill phase (characterized by high synthetic hormones and low endogenous hormones) displayed connectivity patterns that differed significantly from both naturally cycling groups, suggesting that OCs do not simply mimic the luteal phase but may create a distinct neurofunctional state [76] [77].
Table 1: Resting-State Functional Connectivity in Key Brain Networks
| Brain Region/Network | Luteal Phase (vs. Follicular) | OC Users (vs. Naturally Cycling) | OC Users (Active vs. Inactive Pill Phase) |
|---|---|---|---|
| Anterior Cingulate Cortex | Increased connectivity with middle frontal, superior temporal, and postcentral gyri [76] | Altered connectivity patterns in anterior DMN and ECN [76] | Connectivity differences in ACC and frontal regions [76] |
| Left Middle Frontal Gyrus | Significant changes in network connectivity [76] | Decreased connectivity within ECN [76] | Altered functional integration [76] |
| Amygdala | Increased connectivity with right middle frontal gyrus, superior frontal gyrus, and paracentral lobule [80] | Decreased rs-FC with frontal areas with longer HC exposure [80] | Not specifically reported |
| Precuneus | Not significantly changed | Increased connectivity with ACC in OC users [80] | Not specifically reported |
Salience Network and Emotional Processing Regions
The salience network, particularly the ACC and amygdala, has emerged as a key region of interest in hormonal modulation of brain connectivity due to its rich expression of steroid receptors and central role in emotion processing [80]. A comprehensive study examining 231 healthy young women found that longer duration of OC use was associated with decreased rs-FC between the amygdala and frontal areas, as well as between the ACC and temporoparietal areas, independently of the progestin's androgenicity [80]. This suggests cumulative effects of synthetic hormone exposure on emotional processing circuits. Furthermore, the study revealed that the type of progestin in OCs (androgenic vs. anti-androgenic) differentially affected both gray matter volume in the left ACC and functional connectivity between bilateral ACC and the right inferior frontal gyrus, indicating that specific formulation characteristics must be considered in research and drug development [80].
Table 2: Hormonal Contraceptive Effects on Salience Network Connectivity
| Connectivity Measure | Short-Term HC Use (1 cycle) | Long-Term HC Use | Effect of Progestin Type |
|---|---|---|---|
| ACC-Precuneus Connectivity | Increased rs-FC [80] | Varies with duration | Not specifically reported |
| Amygdala-Frontal Connectivity | Not specifically reported | Decreased with longer exposure [80] | Independent of androgenicity |
| ACC-Inferior Frontal Gyrus | Not specifically reported | Not specifically reported | Differential effect based on progestin androgenicity [80] |
| Amygdala-Postcentral Gyrus | Decreased rs-FC [80] | Not specifically reported | Not specifically reported |
Cognitive and Behavioral Correlates
Emotional and Memory Processing
The alterations in functional connectivity described previously manifest in measurable cognitive and behavioral differences. A recent Rice University study demonstrated that women using OCs showed stronger emotional reactions to visual stimuli compared to naturally cycling women and remembered fewer details of negative events when using emotion regulation strategies like distancing or reinterpretation [42]. This reduced memory for unpleasant experiences may actually serve a protective function, potentially preventing excessive rumination on negative information. The researchers also found that strategies like immersion boosted memory for positive images in both OC users and naturally cycling women, making happy moments more memorable [42]. These findings highlight the complex interplay between synthetic hormones, emotional processing, and memory function, suggesting that OCs may modulate the neural circuits underlying emotion regulation.
Further supporting these behavioral observations, a comparative fMRI study examining cerebral activation during a verb generation task found that women using OCs showed superior activation in the right hemisphere compared to naturally cycling women in different menstrual phases [81]. Specifically, OC users displayed increased activation in the superior temporal cortex when compared to women in the menstrual phase and enhanced inferior frontal cortex activation compared to women in the luteal phase [81]. These findings indicate that the synthetic hormone profile in OC users alters the neural substrates of language generation in a manner distinct from both phases of the natural menstrual cycle, potentially reflecting a unique functional organization rather than simple mimicry of any particular natural cycle phase.
Experimental Protocols and Methodologies
Resting-State fMRI Acquisition and Analysis
The investigation of hormonal influences on brain connectivity requires rigorous experimental methodologies and careful participant characterization. Key studies in this field have employed standardized protocols for data acquisition and analysis:
Participant Selection and Hormonal Confirmation: Studies typically include healthy young women (aged 18-40) with regular menstrual cycles or stable OC use for at least 3 months. Naturally cycling women are scanned during specific cycle phases (follicular: days 2-6; luteal: days 18-24), with hormonal levels confirmed through salivary or serum assays. OC users are scanned during both active and inactive pill phases to distinguish acute versus chronic effects [76] [77]. Salivary progesterone and 17β-estradiol assays are performed using commercially available immunoassay kits with detection sensitivity levels of 5 pg/mL and 0.1 pg/mL, respectively [76].
fMRI Data Acquisition: Resting-state fMRI data are typically acquired using 3T MRI scanners with standardized parameters. Participants are instructed to keep their eyes closed, remain awake, and not focus on any particular thought. A standard T2*-weighted echo-planar imaging sequence is used with the following parameters: TR = 2,000 ms, TE = 30 ms, flip angle = 90°, field of view = 220 mm, matrix size = 64 × 64, slice thickness = 3-4 mm, and approximately 30-40 axial slices covering the whole brain [76] [80].
Data Preprocessing: Preprocessing pipelines generally include slice timing correction, motion correction, spatial normalization to standard stereotactic space (e.g., MNI152), and spatial smoothing (6 mm FWHM kernel). Nuisance regressors are included to control for non-neural noise sources, including the 6 rigid body motion parameters, their derivatives, and signals from cerebrospinal fluid and white matter masks [82] [76]. Framewise displacement is calculated, and participants with excessive motion (>2-4mm) are excluded from analysis.
Functional Connectivity Analysis: Analysis approaches include:
- Independent Components Analysis (ICA) to identify intrinsic connectivity networks such as the DMN and ECN [76]
- Seed-based connectivity analysis focusing on a priori regions of interest like the ACC and amygdala [80]
- Spectral dynamic causal modeling to examine effective connectivity between network nodes [79]
- Regional homogeneity (ReHo) to measure local connectivity [82]
- Amplitude of low-frequency fluctuations (fALFF) to assess spontaneous neural activity [82]
Figure 1: Experimental Workflow for Hormonal Connectivity Studies. This diagram illustrates the standardized protocol for investigating resting-state functional connectivity across different hormonal states, including participant selection, fMRI acquisition, and analytical approaches.
Hormonal Assay Protocols
Accurate hormonal assessment is critical for categorizing participants and verifying hormonal status. The following protocol is adapted from Petersen et al. and representative of methodologies used in this research domain [76]:
Sample Collection: Saliva samples are collected immediately before and after scanning via direct expectoration into 15mL Falcon tubes. Each sample volume is approximately 2mL.
Sample Preparation:
- Samples are frozen at -20°C until assaying
- Defrosted samples are centrifuged for 15 minutes at 3000rpm
- The supernatant is decanted into a clean Falcon tube
- Secondary centrifugation is performed for 10 minutes at 3000rpm
Immunoassay Procedure:
- Commercially available immunoassay kits are used (e.g., Salimetrics, State College, PA, USA)
- Salivary progesterone and 17β-estradiol are measured
- Reported detection sensitivity: 5 pg/mL for progesterone, 0.1 pg/mL for estradiol
- All samples are typically run in duplicate to ensure reliability
Neuroendocrine Signaling Pathways
The mechanisms through which sex hormones modulate brain connectivity involve complex interactions between genomic and non-genomic signaling pathways. Understanding these pathways is essential for contextualizing the observed differences in functional connectivity between naturally cycling women and OC users.
Figure 2: Neuroendocrine Signaling Pathways in Hormonal Modulation of Brain Connectivity. This diagram illustrates the complex mechanisms through which endogenous and synthetic sex hormones influence neural function and functional connectivity, highlighting key differences between natural cycling and OC use states.
A critical distinction between natural hormonal cycling and OC use involves the allopregnanolone pathway. Endogenous progesterone is metabolized to allopregnanolone, a potent positive allosteric modulator of GABAA receptors with significant effects on neural excitability and network synchronization [78]. In contrast, synthetic progestins in OCs are not metabolized to allopregnanolone, potentially altering the balance of inhibitory signaling in the brain [80]. This fundamental difference in neurosteroid metabolism may underlie some of the distinct connectivity patterns observed in OC users compared to naturally cycling women in the luteal phase, despite both states featuring elevated progestogenic activity.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials and Analytical Tools
| Resource Category | Specific Examples | Research Application |
|---|---|---|
| Hormonal Assay Kits | Salimetrics Salivary Immunoassay Kits (Progesterone, 17β-estradiol) | Verification of participant hormonal status through sensitive (5 pg/mL for progesterone, 0.1 pg/mL for estradiol) hormone measurement [76] |
| fMRI Analysis Software | AFNI (Analysis of Functional NeuroImages), FSL, SPM, CONN toolbox | Data preprocessing, normalization, and functional connectivity analysis [82] [76] |
| Network Analysis Tools | Graph-based analysis packages, Spectral dynamic causal modeling, Independent Components Analysis (ICA) | Identification of intrinsic connectivity networks and examination of effective connectivity between network nodes [82] [79] |
| Standardized Brain Atlases | Montreal Neurological Institute (MNI) template, Automated Anatomical Labeling (AAL) atlas | Spatial normalization and region-of-interest definition for consistent cross-study comparisons [82] |
| Participant Screening Tools | Structured Clinical Interview for DSM Disorders (SCID), Menstrual cycle tracking protocols | Ensuring participant eligibility and accurate cycle phase or OC use documentation [82] [76] |
The accumulated evidence suggests that oral contraceptive use does not simply mimic the luteal phase of the natural menstrual cycle but rather creates a distinct neurofunctional state with characteristic patterns of resting-state functional connectivity. While both conditions involve elevated progestogenic activity, critical differences in receptor activation, neurosteroid metabolism, and hormonal fluctuation patterns result in unique connectivity signatures. OC use appears associated with altered connectivity in the default mode, executive control, and salience networks, with particular impact on the anterior cingulate cortex and amygdala [76] [80]. These connectivity differences manifest behaviorally in altered emotional reactivity and memory specificity for negative information [42].
Future research in this domain should prioritize several key areas: First, studies must account for the specific formulation characteristics of OCs, particularly the androgenicity of progestins, which appears to differentially impact brain structure and function [80]. Second, longitudinal designs tracking women before and after OC initiation are needed to establish causal relationships and identify potential sensitive periods for hormonal effects on brain organization. Third, multi-modal imaging approaches combining resting-state fMRI with structural and metabolic measures will provide more comprehensive insights into the mechanisms underlying hormonal modulation of brain function. Finally, larger sample sizes and consortium-based efforts, such as the ENIGMA Neuroendocrinology working group, will enhance statistical power and reproducibility in this clinically significant research domain [79].
The hormonal milieu significantly influences cognitive and emotional functions. A growing body of research examines the distinct neurobiological profiles between women using oral contraceptives (OCs) and naturally cycling women, revealing significant differences in memory, fear response, and emotional reactivity. This guide synthesizes experimental data and mechanistic insights from human studies, providing a structured comparison for research and development applications.
Table 1: Comparative Summary of Cognitive and Emotional Processing Profiles
| Processing Domain | Oral Contraceptive Users | Naturally Cycling Women | Key Supporting Findings |
|---|---|---|---|
| Emotional Memory | Enhanced recall for emotional stimuli [83] | Varies with menstrual cycle phase [83] | HC users showed better memory recall of emotional pictures; effect partially mediated by increased perceived emotionality [83] |
| Fear Extinction & Recall | Impaired fear extinction memory recall; heightened psychophysiological response during recall [62] | More robust fear extinction memory; pre-ovulatory phase linked to lower stress responses [62] | Current OC users showed higher skin conductance responses (SCRs) during extinction recall vs. never-users and pre-ovulatory women [62] |
| Emotional Reactivity & Perception | Increased valence ratings for emotional pictures; trend toward blunted reward response and potential stress response dysregulation [84] [83] | Lower valence ratings for the same emotional stimuli; emotional reactivity fluctuates with endogenous hormone levels [83] | HC users rated emotional pictures as more emotionally intense and remembered more of them [84] |
| Underlying Neurocircuitry | Altered activation in vmPFC, ACC, and hippocampus during fear extinction recall; correlates with SCRs [62] | vmPFC, ACC, and hippocampal activity patterns support effective fear memory extinction [62] | Altered neural circuitry during emotional and cognitive tasks suggests a mechanistic basis for behavioral differences [62] |
Detailed Experimental Protocols and Key Data
To ensure reproducibility and critical evaluation, this section outlines the methodologies from pivotal studies cited in this guide.
Table 2: Summary of Key Experimental Protocols and Findings
| Study Focus | Participant Groups | Core Experimental Protocol | Primary Outcome Measures |
|---|---|---|---|
| Fear Extinction Memory [62] | Men, current OC users, never OC users, past OC users, women in early follicular phase, women in pre-ovulatory phase. | Day 1: Fear conditioning (e.g., pairing a neutral stimulus with an aversive one), followed by extinction learning (repeated presentation of the stimulus without the aversive event).Day 2: Extinction recall test and fear renewal test. | Skin Conductance Response (SCR): Measures psychophysiological arousal. OC users showed significantly higher SCRs during extinction recall, indicating impaired retention of safety memory. |
| Emotional Memory & Reactivity [83] | HC Users (n=~1215) vs. Naturally Cycling women (n=~954). Total N=2169. | 1. Picture Rating Task: Viewing and rating emotional pictures (negative, neutral, positive) from the International Affective Picture System (IAPS) for valence and arousal.2. Unexpected Free Recall Task: After a 10-minute delay filled with a distractor task, participants freely recall the previously viewed pictures. | 1. Valence Ratings: Subjective ratings of how positive/negative a picture is. HC users gave higher emotional valence ratings.2. Recall Performance: Number of correctly recalled pictures. HC users had better memory for emotional pictures, an effect partially mediated by their higher valence ratings. |
Signaling Pathways and Neurobiological Mechanisms
The observed behavioral differences are underpinned by distinct neuroendocrine and neural pathway activations. The following diagram illustrates the proposed pathway through which oral contraceptives modulate fear response and emotional memory, integrating findings on hormonal, brain activity, and behavioral changes.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Research in Hormonal Influences on Cognition and Emotion
| Tool / Reagent | Primary Function in Research | Example Application |
|---|---|---|
| International Affective Picture System (IAPS) | Standardized set of emotionally-evocative color images for experimental control of visual emotional stimuli. | Used in picture rating and emotional memory tasks to reliably elicit and assess emotional perception and recall [83]. |
| Fear Conditioning & Extinction Paradigm | A multi-day protocol to investigate the learning (acquisition) and inhibition (extinction) of fear memories. | Gold-standard method for quantifying fear extinction memory recall deficits in OC users vs. controls [62]. |
| Hormonal Assays | Quantification of hormone levels (e.g., estradiol, progesterone, cortisol) from blood, saliva, or urine samples. | Critical for verifying participant group status (e.g., OC use, menstrual cycle phase) and linking hormone levels to cognitive outcomes [66] [85]. |
| Functional Magnetic Resonance Imaging (fMRI) | Non-invasive measurement of brain activity by detecting changes in blood flow, providing high spatial resolution. | Used to identify neural correlates (e.g., in vmPFC, amygdala, hippocampus) of differences in emotional and cognitive processing [62] [86]. |
| Skin Conductance Response (SCR) | Electro-dermal measurement of sympathetic nervous system arousal, reflecting emotional and stress responses. | Objective psychophysiological metric for fear conditioning and extinction experiments [62]. |
This guide provides a comparative analysis of the empirical evidence investigating baseline personality traits in oral contraceptive (OC) users versus naturally cycling (NC) women. A synthesis of current research reveals a consistent pattern of null findings for personality differences alongside emerging evidence for distinct neuroendocrine profiles. The data underscores that previously reported cognitive and affective differences between these groups are unlikely to be confounded by pre-existing personality factors and are more plausibly linked to underlying neuroendocrine mechanisms. The following sections provide a detailed comparison of key studies, their experimental protocols, and the neuroendocrine pathways that may offer explanatory power for observed psychological differences.
| Study Focus | Primary Findings | Key Quantitative Data | Sample Size (OC/NC) |
|---|---|---|---|
| Big Five Personality Traits [36] [28] [87] | No significant differences in Neuroticism, Extraversion, Openness, Agreeableness, or Conscientiousness. | Absolute univariate effect sizes averaged d = .09. Multivariate analysis: Wilks' Λ = .97-.99, p > .244 [36] [28]. | Sample 1: 148 OC / 93 NCSample 2: 247 OC / 148 NC |
| Fear Extinction & Neural Correlates [62] | Impaired fear extinction recall in current OC users; correlated with vmPFC, ACC, and hippocampal activations. | Higher skin conductance responses during extinction recall in current users vs. never users [62]. | 147 healthy adults (grouped by OC use and menstrual phase) |
| HPO Axis & Metabolic Hormones [66] | Combined HC users maintained stable estradiol/testosterone during low energy availability (LEA), unlike NC women. | IGF-1, triiodothyronine (T3), and leptin decreased comparably in all groups during LEA; cortisol remained unchanged [66]. | 36 physique athletes (11 combined HC, 11 progestin-only HC, 14 NC) |
Detailed Experimental Protocols
Protocol for Personality Assessment (Beltz et al., 2019)
The definitive study ruling out personality confounds employed a rigorous, well-powered methodology across two independent samples [28].
- Participants: A total of 632 women were recruited, divided into two samples. Sample 1 included 148 OC users and 93 NC women; Sample 2 included 247 OC users and 148 NC women. This provided 6-10 times the number of OC users than the average study in this field.
- Personality Measurement: The Big Five personality factors (Neuroticism, Extraversion, Openness, Agreeableness, and Conscientiousness) were assessed using standard psychometric instruments.
- Statistical Analysis: Multivariate analyses of covariance (MANCOVA) were conducted with the five personality factors as dependent variables and age as a covariate. The analyses were run separately for each sample to serve as independent replications. Follow-up univariate analyses and examinations of different OC types and reasons for use were also performed.
- Key Outcome: The analyses revealed no significant overall differences in personality profiles between OC users and NC women in either sample. The null finding was robust, with very small effect sizes, and held true even when considering the reason for OC use or the type of OC used [36] [28].
Protocol for Neuroendocrine Challenge (Gerra et al., 2000)
An earlier, foundational study exemplifies the methodology for investigating the neuroendocrine correlates of temperament [88].
- Participants: 22 healthy male volunteers.
- Temperament & Mood Assessment: The Cloninger Three-dimensional Personality Questionnaire (TPQ) was used to assess Novelty Seeking (NS), Harm Avoidance (HA), and Reward Dependence (RD). The MMPI-2 and Hamilton Rating Scale for Depression were also administered.
- Neuroendocrine Challenges: Monoamine system function was evaluated indirectly via hormonal responses to specific pharmacological agents:
- Dopamine (DA) function: Assessed via Growth Hormone (GH) and Prolactin (PRL) responses to acute administration of bromocriptine (a D2 receptor agonist).
- Serotonin (5-HT) function: Evaluated via PRL and cortisol responses to acute d-fenfluramine (a 5-HT releaser and agonist).
- Norepinephrine (NE) function: Measured by the GH response to clonidine (an alpha-2-adrenergic agonist).
- Key Outcome: The study found correlations between specific temperament traits and hormonal responses, supporting Cloninger's theory. For instance, NS scores correlated positively with the GH response to bromocriptine (linked to DA function), while HA scores correlated with the cortisol response to d-fenfluramine (linked to 5-HT function) [88].
Neuroendocrine Explanations for Psychological Phenomena
The null findings for personality necessitate a look at other mechanistic levels. Neuroendocrine pathways offer a compelling explanatory framework for the cognitive and affective differences observed between OC users and NC women.
Signaling Pathways in Hormone-Behavior Interaction
The following diagram illustrates the primary neuroendocrine pathways implicated in shaping behavior and cognition, and how they are potentially modulated by hormonal contraceptives.
The diagram shows how OCs, by introducing exogenous hormones, can directly suppress the HPO axis and modulate the HPA axis. These changes in systemic hormone levels can, in turn, influence key monoamine neurotransmitter systems in the brain (Dopamine, Serotonin, Norepinephrine), which are fundamental to regulating behavior, cognition, and mood [88] [66] [89]. For example, the impaired fear extinction recall observed in OC users may be linked to these hormonal influences on the vmPFC, ACC, and hippocampus [62].
Experimental Workflow for Differentiating Neuroendocrine Effects
The flowchart below outlines a general experimental workflow for designing studies that can dissociate neuroendocrine effects from pre-existing trait differences.
This workflow emphasizes the critical step of first establishing the absence of baseline personality differences (the null finding) before attributing group differences in experimental outcomes to neuroendocrine mechanisms [36] [28] [87].
The Scientist's Toolkit: Research Reagent Solutions
The following table details key materials and methodologies essential for research in this field.
Table 2: Essential Research Reagents and Methods
| Item Name | Function & Application | Example Use in Context |
|---|---|---|
| Big Five Personality Inventory | A psychometric questionnaire to assess the five major dimensions of personality (Neuroticism, Extraversion, Openness, Agreeableness, Conscientiousness). | Used as a control measure to rule out pre-existing personality differences as a confound in studies comparing OC users and NC women [36] [28] [90]. |
| Cloninger's Tridimensional Personality Questionnaire (TPQ) | Assesses three heritable temperament dimensions: Novelty Seeking (NS), Harm Avoidance (HA), and Reward Dependence (RD). | Used in neuroendocrine studies to correlate temperament traits with hormonal responses to pharmacological challenges [88]. |
| Pharmacological Challenge Agents | Specific agonists/releasers used to probe the functional state of central monoamine systems indirectly via hormonal responses. | d-Fenfluramine: 5-HT agonist; stimulates PRL/CORT release [88].Bromocriptine: D2 receptor agonist; stimulates GH release, suppresses PRL [88].Clonidine: alpha-2-adrenergic agonist; stimulates GH release [88]. |
| Hormonal Assays (ELISA/RIA) | To quantify concentrations of specific hormones (e.g., estradiol, testosterone, cortisol, IGF-1) from blood, saliva, or cerebrospinal fluid. | Used to establish baseline hormone levels and measure changes in response to experimental manipulations like low energy availability or pharmacological challenges [88] [66]. |
| fMRI / ERP | Non-invasive neuroimaging techniques to measure brain activity and neural correlates of cognitive or emotional processes. | fMRI: Identified correlation between impaired fear extinction in OC users and activity in the hippocampus, ACC, and vmPFC [62].ERP (N170, LPP): Used to investigate early and late neural processing of social stimuli like faces [91]. |
The study of hormonal contraceptives has traditionally focused on their short-term physiological effects and efficacy. However, as millions of women worldwide use oral contraceptives (OCs) during their reproductive years—often for extended periods—a critical question emerges: what are the long-term neurobehavioral implications after discontinuation? This review synthesizes current evidence comparing former OC users to never-users, examining whether OC-induced alterations in brain and behavior persist beyond the period of active use. Understanding these potential enduring effects is crucial for clinicians, researchers, and the millions of women who have used OCs throughout their lives.
The hypothalamic-pituitary-gonadal (HPG) axis represents a primary pathway through which OCs exert their influence. OCs suppress endogenous hormone fluctuation by inhibiting the release of gonadotropin-releasing hormone (GnRH) from the hypothalamus, subsequently reducing luteinizing hormone (LH) and follicle-stimulating hormone (FSH) release from the pituitary, and preventing ovarian production of estradiol and progesterone [12]. This fundamental alteration in neuroendocrine signaling provides the mechanistic basis for investigating both acute and potential long-term effects on the brain.
Quantitative Synthesis of Enduring Effects
Table 1: Summary of Long-Term Effects of Past Oral Contraceptive Use
| Domain | Comparison | Key Finding | Duration of Past Use Correlation | Study Reference |
|---|---|---|---|---|
| Verbal Fluency | Former Users vs. Never-Users | No significant overall difference in performance [92]. | Longer past use associated with poorer phonemic fluency in former users [92]. Association between longer current use and reduced word production [93]. | Kowalczyk et al., 2025; Pletzer et al., 2022 |
| Visuospatial Function | Former Users vs. Never-Users | No significant difference in performance on the Space Relations task [92]. | Not reported [92]. | Kowalczyk et al., 2025 |
| Brain Morphology | Former Users vs. Current Users vs. Never-Users | Thinner ventromedial prefrontal cortex (vmPFC) in current users vs. men; effect not observed in past users [94]. | Not specified [94]. | Brouillard et al., 2023 |
| Self-Esteem & Mood | Current Users vs. Naturally Cycling | Lower daily self-esteem in current OC users [44]. Higher daily depression in current OC users [44]. | Data on long-term effects after discontinuation not available [44]. | Gawron et al., 2025 |
Table 2: Methodological Overview of Key Studies on Long-Term OC Effects
| Study Focus | Participant Groups | Sample Size (Total) | Key Assessment Methods | Progestin Types Considered |
|---|---|---|---|---|
| Cognitive Performance [92] | Former Users, Never-Users, Current Users | 221 | Standardized cognitive tests (verbal fluency, visuospatial), reproductive history questionnaire | Levonorgestrel, Norethindrone family |
| Brain Morphology [94] | Current OC Users, Past OC Users, Never-Users, Men | Not specified | Structural Magnetic Resonance Imaging (MRI) | Not specified |
| Verbal Fluency & Brain Activation [93] | Current and Past OC Users (subdivided by progestin type) | 94 | Functional MRI (fMRI), verbal fluency and navigation tasks, duration of use history | Androgenic vs. Anti-androgenic |
Experimental Protocols and Methodologies
Neuroimaging and Cognitive Testing Protocol
To investigate persistent effects, researchers have employed sophisticated neuroimaging protocols combined with cognitive assessments. One comprehensive approach involved recruiting former OC users, never-users, and current users, with cognitive evaluation timed for the early follicular phase of the menstrual cycle when endogenous ovarian hormone production is lowest [92]. This controlled for cyclical hormonal fluctuations that might confound results.
Key procedural steps included:
- Participant Screening & Group Classification: Administration of a detailed reproductive history questionnaire to document specifics of past OC use, including duration, formulation, and time since discontinuation [92].
- Cognitive Assessment: Administration of standardized neuropsychological tests, such as phonemic and semantic verbal fluency tasks (e.g., generating words beginning with a specific letter) and visuospatial tests (e.g., Space Relations task) [92].
- Brain Imaging: Using functional Magnetic Resonance Imaging (fMRI) to measure brain activation during cognitive tasks. In studies of current and past users, researchers examined activation in regions like the left putamen and connectivity between language-related areas such as the right inferior frontal gyrus and right angular gyrus [93].
- Data Analysis: Employing analysis of covariance (ANCOVA) to compare cognitive performance between groups, controlling for variables like age and IQ. Partial correlations were used to explore relationships between duration of past OC use and cognitive outcomes [92].
Daily Diary Study Protocol for Current Users
While not assessing long-term effects directly, daily diary studies illuminate the acute effects that may potentially leave lasting impressions. One such protocol involved:
- Design: A 15-day online diary study divided into three phases across one menstrual cycle [44].
- Measures: Daily administration of adapted trait-level scales measuring depression, anxiety, self-esteem, life satisfaction, and perseverative cognition [44].
- Analysis: Hierarchical linear modeling to analyze daily fluctuations and differences between groups, including naturally cycling women and users of androgenic versus anti-androgenic OCs [44].
Signaling Pathways and Research Frameworks
The investigation of OCs' long-term impact hinges on understanding their interaction with neuroendocrine systems. The primary pathway involves the suppression of the HPG axis, while a critical conceptual framework for long-term studies differentiates between activational and potential organizational effects.
This framework visualizes the central mechanistic question: while the activational effects of OCs (dependent on the continued presence of synthetic hormones) are expected to reverse after discontinuation, exposure during a sensitive period could potentially induce organizational effects—enduring changes in brain structure and function that persist long-term [92]. Current evidence suggests that many effects, such as reduced thickness of the ventromedial prefrontal cortex observed in current users, are not detected in past users, indicating reversibility [94]. However, some duration-dependent correlations with cognitive performance hint at more persistent organizational changes.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Tools for OC Neurobehavioral Research
| Item/Tool | Specific Example | Function in Research |
|---|---|---|
| Reproductive History Questionnaire | Custom-developed detailed survey [92] | Documents OC use history (duration, type, timing), menstrual cycle regularity, and other relevant hormonal exposures. Critical for accurate group classification. |
| Cognitive Assessment Tools | Verbal Fluency Tests (phonemic/semantic) [93] [92]; Visuospatial Tests (Space Relations) [92] | Provides standardized, quantitative measures of cognitive performance in domains potentially sensitive to hormonal influences. |
| Neuroimaging Platform | Functional Magnetic Resonance Imaging (fMRI) [93] | Measures task-dependent brain activation and functional connectivity, identifying neural correlates of cognitive and emotional processes. |
| Structural MRI | T1-weighted volumetric imaging [94] | Quantifies morphological characteristics of brain regions (e.g., cortical thickness of the vmPFC) to identify structural changes. |
| Hormonal Assay Kits | Salivary progesterone immunoassays [95] | Validates menstrual cycle phase in naturally cycling women and measures baseline hormone levels in all participants. |
| Statistical Analysis Software | R, Python, or specialized packages (e.g., for HLM) [44] | Performs complex statistical analyses, including hierarchical linear modeling for diary data and ANCOVA for group comparisons [44] [92]. |
The current body of research provides reassuring evidence that many neurobehavioral changes associated with OC use appear to be reversible after discontinuation, with no significant broad cognitive deficits observed in former users compared to never-users [92]. However, nuanced findings—such as the duration-dependent association with verbal fluency performance—suggest that the long-term implications may be more complex and subtle [93] [92]. The field requires larger, longitudinal studies that track women from initiation through long-term discontinuation, while rigorously accounting for OC formulation, age of onset, and genetic predispositions. Such research is essential to fully empower women and clinicians in making informed decisions, and to deepen our understanding of how synthetic hormones shape the human brain across the lifespan.
Conclusion
The evidence clearly demonstrates that oral contraceptive users and naturally cycling women represent distinct neuroendocrine populations with meaningful differences in brain function, emotional processing, and potential health outcomes. OC use induces a sustained hormonal environment that differs profoundly from the natural cycle's rhythmicity, potentially creating a hypogonadal or hyperprogestogenic state in the brain. For biomedical research, this underscores a critical methodological imperative: endocrine status must be a key variable in study design, analysis, and interpretation, especially in neuroscience, psychology, and drug development. Future research must move beyond simple user/non-user comparisons to systematically investigate the effects of specific OC formulations, the long-term neural consequences of use, and the interaction between synthetic hormones and pharmacotherapies. Embracing this complexity is essential for developing truly effective, personalized medical treatments for women.
References
- 1. Oral Contraceptive Pills - StatPearls - NCBI Bookshelf - NIH [https://www.ncbi.nlm.nih.gov/books/NBK430882/]
- 2. Combined oral contraceptive pill [https://en.wikipedia.org/wiki/Combined_oral_contraceptive_pill]
- 3. Progestin-Only Pills | Contraception [https://www.cdc.gov/contraception/hcp/usspr/progestin-only-pills.html]
- 4. Oral Contraceptives - Gynecology and Obstetrics [https://www.merckmanuals.com/professional/gynecology-and-obstetrics/family-planning/oral-contraceptives]
- 5. How the Pill Works | American Experience | Official Site [https://www.pbs.org/wgbh/americanexperience/features/pill-how-pill-works/]
- 6. Combination birth control pills [https://www.mayoclinic.org/tests-procedures/combination-birth-control-pills/about/pac-20385282]
- 7. Choosing the right pill [https://www.contemporaryobgyn.net/view/choosing-the-right-pill]
- 8. Combined Hormonal Contraception - Contraindications [https://teachmeobgyn.com/sexual-health/contraception/combined-hormonal-contraception/]
- 9. Contraception: Practice Essentials, Overview, Periodic ... [https://emedicine.medscape.com/article/258507-overview]
- 10. An Evidence-Based Update on Contraception [https://pmc.ncbi.nlm.nih.gov/articles/PMC7533104/]
- 11. Editorial: Effects of hormonal contraceptives on the brain [https://pmc.ncbi.nlm.nih.gov/articles/PMC9927394/]
- 12. How hormonal contraceptives shape brain and behavior [https://www.sciencedirect.com/science/article/abs/pii/S0091302222000401]
- 13. Hormonal contraceptive effects on the brain [https://www.nature.com/articles/s41386-025-02267-0]
- 14. Mechanisms of Central Hypogonadism - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8348115/]
- 15. Hormonal Contraceptives and the Brain: A Systematic Review ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9898167/]
- 16. Effects of Oral Contraceptive Pills on Brain Networks - PubMed [https://pubmed.ncbi.nlm.nih.gov/40782033/]
- 17. Effects of an Oral Contraceptive on Dynamic Brain States ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2022.855582/full]
- 18. The association of hypogonadism with depression and its ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10449581/]
- 19. How does the pill affect your brain? We're finally getting answers [https://lsa.umich.edu/psych/news-events/all-news/faculty-news/how-does-the-pill-affect-your-brain--we-re-finally-getting-answe.html]
- 20. Effects of oral contraceptives and natural menstrual cycling on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6223061/]
- 21. No cycle on the Pill-Natural Womanhood [https://naturalwomanhood.org/no-cycle-on-the-pill/]
- 22. Effect of combined oral contraceptive use on verbal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12436564/]
- 23. A prospective daily study on psychological well-being ... [https://pubmed.ncbi.nlm.nih.gov/39917827/]
- 24. Hormone Replacement Versus Natural Cycle Protocols of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7555052/]
- 25. The Link Between Masculinity and Spatial Skills Is ... [https://www.frontiersin.org/journals/behavioral-neuroscience/articles/10.3389/fnbeh.2021.777911/full]
- 26. The Effects of Oral Contraceptives on Exercise Performance in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7497464/]
- 27. Comparative effectiveness and safety of different progestins in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12334503/]
- 28. No personality differences between oral contraceptive ... [https://www.sciencedirect.com/science/article/abs/pii/S030645301830667X]
- 29. Effects of oral contraceptives and natural menstrual cycling on ... [https://bmcwomenshealth.biomedcentral.com/articles/10.1186/s12905-018-0671-4]
- 30. Effects of Oral Contraceptive Pills on Brain Networks [https://pmc.ncbi.nlm.nih.gov/articles/PMC12335007/]
- 31. Hormonal milieu influences whole-brain structural ... [https://www.nature.com/articles/s41593-025-02066-2]
- 32. The relationships of resting-state brain entropy (BEN) ... [https://www.sciencedirect.com/science/article/pii/S1053811925002290]
- 33. Endocrine-Disrupting Chemicals, Hypothalamic ... [https://www.mdpi.com/1422-0067/25/21/11344]
- 34. Determining Menstrual Cycle Phase - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC10714354/]
- 35. Machine learning-based menstrual phase identification ... [https://www.nature.com/articles/s44294-025-00078-8]
- 36. No personality differences between oral contraceptive ... [https://pubmed.ncbi.nlm.nih.gov/30321772/]
- 37. Variation in serum biomarkers with sex and female ... [https://www.nature.com/articles/srep26947]
- 38. Hormone Immunoassay Interference: A 2021 Update - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8368230/]
- 39. Improving Science by Overcoming Laboratory Pitfalls With ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7993596/]
- 40. Biomarker Comparison Chart [https://dutchtest.com/biomarker-comparison-chart]
- 41. Seven Hormonal Biomarkers for Diagnosing Endometriosis [https://pubmed.ncbi.nlm.nih.gov/30965114/]
- 42. Millions of women rely on contraceptives, but new Rice ... [https://news.rice.edu/news/2025/millions-women-rely-contraceptives-new-rice-study-shows-they-may-do-more-just-prevent]
- 43. Everything to Know About Stratified Sampling [https://amplitude.com/explore/experiment/stratified-sampling]
- 44. Anxiety, depression and perseverative cognition in women ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12523149/]
- 45. Exploring Stratification Strategies for Population‐ Versus ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11602877/]
- 46. Stratification: Improving test sensitivity [https://www.statsig.com/perspectives/stratification-improving-sensitivity]
- 47. Cortisol dynamics and sleep quality: The role of sex and ... [https://www.sciencedirect.com/science/article/pii/S0306453025002203]
- 48. Comprehensive Hormone Profile; saliva [https://www.doctorsdata.com/Comprehensive-Hormone-Profile-saliva]
- 49. Comprehensive Hormone Profile by Doctor's Data [https://www.rupahealth.com/lab-tests/doctors-data-comprehensive-hormone-profile]
- 50. Our Comprehensive Hormones Profile | MosaicDX [http://mosaicdx.com/test/hormones-profile/]
- 51. The best charts for color blind viewers | Blog [https://www.datylon.com/blog/data-visualization-for-colorblind-readers]
- 52. Guidelines color blind friendly figures [https://www.nki.nl/about-us/responsible-research/guidelines-color-blind-friendly-figures]
- 53. Menstrual cycle length variation by demographic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10226714/]
- 54. Menstrual cycle length variation by demographic ... [https://www.nature.com/articles/s41746-023-00848-1]
- 55. Real-world menstrual cycle characteristics of more than ... [https://www.nature.com/articles/s41746-019-0152-7]
- 56. Hormone-based models for comparing menstrual cycle and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9649880/]
- 57. Variability in the phases of the menstrual cycle - PubMed [https://pubmed.ncbi.nlm.nih.gov/16700687/]
- 58. How to study the menstrual cycle - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC8363181/]
- 59. Menstrual cycle length and variability: a visual explanation [https://awhs-updates.hsph.harvard.edu/cycle-length-variability/]
- 60. Intraindividual Hormonal Variability in Ultrasonographically ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3455578/]
- 61. Population-based cohort study of oral contraceptive use ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10294242/]
- 62. Associations between past and current use of oral ... [https://www.sciencedirect.com/science/article/pii/S2451902225002964]
- 63. Stress-Hormone Dynamics and Working Memory in ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2021.731994/full]
- 64. Current oral contraceptive use affects explicit and implicit ... [https://www.frontiersin.org/journals/psychology/articles/10.3389/fpsyg.2024.1462891/full]
- 65. Changing Fear: The Neurocircuitry of Emotion Regulation [https://www.nature.com/articles/npp2009121]
- 66. Hormone Profiles After Planned Low Energy Availability ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12616881/]
- 67. Cognitive reappraisal of conditioned fear: A systematic ... [https://pubmed.ncbi.nlm.nih.gov/41237605/]
- 68. A Novel method for quantifying fluctuations in wearable ... [https://www.nature.com/articles/s41746-024-01394-0]
- 69. The Evolution of the WHI 80+ Cohort - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC5865530/]
- 70. Long-term Effects on Cognitive Trajectories of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6075542/]
- 71. Long-term cognitive effects of menopausal hormone therapy [https://pmc.ncbi.nlm.nih.gov/articles/PMC11581397/]
- 72. Long-term cognitive effects of menopausal hormone therapy [https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1004435]
- 73. Study shows how birth control pills affect women's ... [https://www.uclahealth.org/news/release/study-shows-how-birth-control-pills-affect-womens]
- 74. Comparative analysis of brain volumetric measurements between contrast-enhanced and non-contrast MRI images - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S0304394025000060]
- 75. Flexible annotation atlas of the mouse brain [https://www.nature.com/articles/s41598-021-85807-0]
- 76. Oral contraceptive pill use and menstrual cycle phase are ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4113343/]
- 77. Oral contraceptive pill use and menstrual cycle phase are ... [https://www.sciencedirect.com/science/article/abs/pii/S1053811913012202]
- 78. Hormonal regulation of behavioral and emotional persistence [https://www.sciencedirect.com/science/article/abs/pii/S1074742725000450]
- 79. Spotlighting SHAPERS: sex hormones associated with ... [https://www.nature.com/articles/s41386-024-01819-0]
- 80. Hormonal contraceptive exposure relates to changes in ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2023.1131995/full]
- 81. comparison of women in different cycle phases, under oral ... [https://pubmed.ncbi.nlm.nih.gov/20036289/]
- 82. The colors of our brain: an integrated approach for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9107439/]
- 83. Women using hormonal contraceptives show increased ... [https://www.nature.com/articles/s41386-019-0362-3]
- 84. Effects of Hormonal Contraceptives on Mood [https://pmc.ncbi.nlm.nih.gov/articles/PMC6838021/]
- 85. Oral contraceptives and cognition: A systematic review [https://www.sciencedirect.com/science/article/abs/pii/S0091302222000759]
- 86. Cognitive control mechanisms, emotion and memory [https://www.sciencedirect.com/science/article/abs/pii/S014976340800170X]
- 87. Women who use oral contraceptives have the same ... [https://www.psypost.org/women-who-use-oral-contraceptives-have-the-same-personality-traits-as-naturally-cycling-women/]
- 88. Neuroendocrine correlates of temperamental traits in humans [https://www.sciencedirect.com/science/article/abs/pii/S0306453000000044]
- 89. Risk-averse personalities have a systemically potentiated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5552616/]
- 90. Personality Trait Level and Change as Predictors of Health ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3410684/]
- 91. Neural dynamics of personality trait perception and ... [https://www.nature.com/articles/s41598-024-76423-9]
- 92. Do combined oral contraceptives have long-term effects ... [https://pubmed.ncbi.nlm.nih.gov/40749444/]
- 93. Duration of oral contraceptive use relates to cognitive ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2022.885617/full]
- 94. Study shows the effects of oral contraceptive use on ... [https://www.news-medical.net/news/20231106/Study-shows-the-effects-of-oral-contraceptive-use-on-womene28099s-brains.aspx]
- 95. Hormonal fluctuations in the menstrual cycle may affect ... [https://www.2minutemedicine.com/hormonal-fluctuations-in-the-menstrual-cycle-may-affect-sleep-and-memory-performance/]