AMH Gen II ELISA vs. Elecsys Cobas: A Comprehensive Analytical Comparison for Clinical and Research Applications
This article provides a detailed comparative analysis of two prominent Anti-Müllerian Hormone (AMH) assays: the manual AMH Gen II ELISA and the fully automated Elecsys Cobas AMH.
AMH Gen II ELISA vs. Elecsys Cobas: A Comprehensive Analytical Comparison for Clinical and Research Applications
Abstract
This article provides a detailed comparative analysis of two prominent Anti-Müllerian Hormone (AMH) assays: the manual AMH Gen II ELISA and the fully automated Elecsys Cobas AMH. Aimed at researchers and drug development professionals, it explores the foundational principles, methodological workflows, and analytical performance characteristics of each assay. Key findings from recent studies indicate that the Elecsys method demonstrates superior precision (CV of 2.8-3.3% vs. 5.5-10.3% for Gen II ELISA), a lower limit of quantitation (0.5 pmol/L vs. 3.0 pmol/L), and optimal performance across its measuring range, albeit with a consistent negative bias of approximately 32% compared to the Gen II ELISA. The review synthesizes evidence on assay standardization, troubleshooting common variability issues, and validates performance against clinical endpoints like antral follicle count. It concludes by discussing the implications for clinical diagnostics and the pressing need for international harmonization of AMH measurement.
Understanding AMH and the Evolution of Its Measurement
The Biological Role of AMH as a Key Biomarker of Ovarian Reserve
Anti-Müllerian Hormone (AMH), a dimeric glycoprotein belonging to the transforming growth factor-β (TGF-β) superfamily, has emerged as a pivotal biochemical marker for the quantitative assessment of ovarian reserve [1]. Produced by the granulosa cells of preantral and small antral follicles in the ovaries, AMH serum concentration provides a direct, reproducible reflection of the primordial follicle pool [2] [1]. Unlike other menstrual cycle-dependent hormones, AMH exhibits relatively stable expression throughout the menstrual cycle, enabling reliable clinical measurement at any time [1] [3]. This stability, coupled with its strong correlation with antral follicle count (AFC), has established AMH as a superior marker for predicting ovarian response in assisted reproductive technology, diagnosing conditions like polycystic ovary syndrome (PCOS), and assessing iatrogenic ovarian damage [1] [4].
The evolution of AMH measurement technologies from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated immunoassays has significantly improved the precision and clinical utility of AMH testing [5] [2] [6]. This guide provides a comprehensive, objective comparison between two prominent AMH assay methods: the established AMH Gen II ELISA and the automated Elecsys Cobas AMH assay, presenting experimental data to inform researchers, scientists, and drug development professionals.
Comparative Assay Performance Data
Analytical Performance Characteristics
Table 1: Direct Comparison of Key Analytical Performance Parameters
| Performance Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Assay Principle | Manual ELISA | Fully Automated Immunoassay |
| Measuring Range | 3–70 pmol/L (without dilution) [5] | 0.03-23 ng/mL (approx. 0.21-164.0 pmol/L) [7] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L [5] | 0.21 pmol/L [8] |
| Analytical Variability (CV%) | 5.5-10.3% [5] | 2.8-3.3% [5] |
| Inter-assay Precision | CV 11-14% (internal controls) [5] | CV <5% (internal controls) [5] [7] |
| Sample Volume | Higher volume required [5] | As low as 30 μL [7] |
| Throughput | Manual processing, slower turnaround | Fully automated, results in 18 minutes [7] |
Method Correlation and Clinical Agreement
Table 2: Correlation Studies Between AMH Assay Methods
| Study Focus | Correlation Findings | Clinical Implications |
|---|---|---|
| Direct Method Comparison | Strong correlation (R² = 0.982-0.983) between Access AMH (automated) and Gen II ELISA [2] | High degree of result comparability between automated and ELISA platforms |
| Inter-assay Correlation | Elecsys values approximately 32% lower than ELISA with increased bias at higher concentrations [5] | Assay-specific reference ranges essential for accurate interpretation |
| PCOS Population Correlation | Strong interassay correlations in low (<2.80 ng/mL) and high (>7.04 ng/mL) AMH subgroups (0.62-0.86) [9] | Consistent performance across different patient populations and AMH levels |
| Correlation with Antral Follicle Count | Significant correlation for both Cobas (0.83) and ELISA (0.86) with total AFC [5] | Both methods reliably reflect biological ovarian reserve marker |
Experimental Protocols and Methodologies
Prospective Comparative Study Design
A 2015 prospective observational study directly compared the AMH Gen II ELISA and Elecsys Cobas AMH methods in a clinical setting to evaluate whether the assays achieved optimal analytical performance goals [5].
Population: 23 women with median age 36 years (range 33-38) undergoing laparoscopic sterilization.
Sample Collection: Blood samples were collected preoperatively, then at 1 week, 1 month, 3 months, and 6 months postoperatively. Serum was isolated within 4 hours and stored at -80°C until analysis.
Testing Protocol:
- AMH Gen II ELISA: Performed using standard manual technique with sample pre-mixture in assay buffer. Analyses performed in duplicate with repeat testing if results differed by >15% [5].
- Elecsys Cobas AMH: Analyzed on Cobas 6000 e601 platform using Roche's standard automated protocol [5].
- Ultrasound Assessment: Antral follicle count (AFC) and ovarian volume measurements performed using Voluson E6 or Logiq 9 systems [5].
Statistical Analysis: Spearman's correlation test determined correlation between AMH and AFC. Passing-Bablok and Bias plots analyzed method comparison. Total variation calculated as CVTOTAL = √[(CVWithin-person Biological Variation)² + (CVAnalytical)²] [5].
Performance Validation Protocol
A 2016 study evaluated the Access AMH assay (another automated assay) against the modified Gen II ELISA method, employing rigorous validation protocols applicable to the field [2].
Precision Testing:
- Intra-assay precision: 10 replicates of QC material during same run
- Inter-assay precision: First run of QC material daily for nine consecutive days
- Calculations: CV% = (standard deviation/mean) × 100 [2]
Stability Assessment:
- Temperature stability: Testing samples stored at 4°C for eight days
- Freeze-thaw stability: Seven freeze-thaw cycles with testing after each thaw
- Calculations: % deviation of mean = (mean - expected mean)/expected mean × 100 [2]
Linearity and Detection Limits:
- Linearity: Series dilutions of calibrator material and patient sera using Cusum test
- Limit of Blank (LoB): Mean of blank + 1.645 × SD of blank
- Limit of Detection (LoD): LoB + 1.645 × SD of low-level samples [2]
Visualization of Assay Workflows and Technical Principles
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for AMH Assay Comparison Studies
| Reagent/Material | Function/Application | Technical Specifications |
|---|---|---|
| Elecsys AMH Reagent | Automated immunoassay for AMH measurement on Cobas platforms | Measuring range: 0.03-23 ng/mL; Imprecision: <5% [7] |
| AMH Gen II ELISA Kit | Manual ELISA for AMH measurement | Measuring range: 3-70 pmol/L; Acceptable CV: 11-14% [5] |
| PreciControl AMH | Internal quality control for Elecsys AMH assay | Two-level control (e.g., 4.29 pmol/L and 34.71 pmol/L) [8] |
| Sample Diluent A | Recommended diluent for automated AMH assays | Required for high-concentration sample dilution [2] |
| Human Recombinant AMH | Calibrator material for assay validation | Used for precision, linearity, and recovery studies [2] |
| Li-heparin Plasma/Serum | Primary sample matrices for AMH testing | Approved sample types; stability varies by storage conditions [7] |
Clinical and Research Applications
Predictive Value in Assisted Reproduction
AMH measurement has become indispensable in predicting ovarian response during in vitro fertilization (IVF) treatments. The Elecsys AMH assay demonstrates strong predictive capability for oocyte yield following gonadotropin-releasing hormone (GnRH) antagonist cycles [4]. A retrospective cohort study of 1,248 patients established that AMH concentration alone accounted for 47% (R² = 0.470) of the variation in ovarian response, increasing to 50.9% with the addition of age, body weight, and gonadotropin dose [4]. Specific Elecsys AMH cut-offs were identified for clinical decision-making: 6.4 pmol/L for predicting low response (0-3 oocytes) and 14.2 pmol/L for predicting high response (>15 oocytes) [4].
Assessment of Ovarian Reserve Across Populations
Age-specific reference intervals for the Elecsys AMH assay have been established across diverse populations. A 2022 study of 1,450 healthy Korean women demonstrated consistent age-related decline patterns, with median AMH decreasing from 30.9 pmol/L in women aged 19-24 years to 0.071 pmol/L in women aged 50-54 years [8]. These findings highlight the importance of population-specific and assay-specific reference intervals for accurate clinical interpretation. The mid-95 percentile reference intervals decreased from 7.93-81.21 pmol/L to 0.07-3.86 pmol/L across these age groups [8].
The comprehensive comparison between AMH Gen II ELISA and Elecsys Cobas AMH assays reveals a consistent trend toward superior analytical performance of automated platforms. The Elecsys system demonstrates enhanced precision (CV 2.8-3.3% versus 5.5-10.3%), broader measuring range (0.21-164.0 pmol/L versus 3-70 pmol/L), and lower limit of quantitation (0.21 pmol/L versus 3.0 pmol/L) compared to the manual ELISA method [5] [7].
While strong correlations exist between method results (R² = 0.982-0.983), the observed systematic bias of approximately 32% emphasizes that these assays are not directly interchangeable without proper calibration [5] [2]. The Elecsys Cobas AMH assay achieves optimal performance standards (CVAnalytical < 0.25*CVWithin Biological Variation) throughout its measuring range, while the AMH Gen II ELISA meets this standard only at higher concentrations [5].
For researchers and clinicians, selection between these methodologies should consider specific application requirements. The automated Elecsys system offers advantages in throughput, precision, and standardization for high-volume clinical settings, while manual ELISA platforms may retain utility in resource-limited environments or for specific research applications. Future directions should focus on international standardization efforts to improve result harmonization across platforms and establish universal reference intervals for enhanced clinical utility across diverse populations.
The measurement of Anti-Müllerian Hormone (AMH) has become a cornerstone in reproductive medicine, providing clinicians with a valuable marker for assessing ovarian reserve, predicting response to ovarian stimulation, and diagnosing conditions like polycystic ovary syndrome (PCOS). For years, the manual AMH Gen II ELISA (Beckman Coulter) served as the standard method in clinical laboratories worldwide. However, this manual method has been challenged by the recent introduction of fully automated immunoassays, notably the Elecsys Cobas AMH assay (Roche Diagnostics), promising enhanced precision and workflow efficiency.
This technological shift from manual to automated platforms represents a significant evolution in immunoassay technology, with profound implications for clinical diagnostics and research. This guide objectively compares the performance characteristics of these two distinct methodological approaches, providing researchers and drug development professionals with experimental data to inform their analytical decisions.
Methodological Comparison: Manual ELISA vs. Automated ECLIA
The fundamental difference between these platforms lies in their operational and technological principles. The AMH Gen II assay is a manual enzyme-linked immunosorbent assay (ELISA) requiring extensive hands-on technician time, including manual pipetting, washing, and incubation steps. The Elecsys Cobas AMH assay, in contrast, is a fully automated electrochemiluminescence immunoassay (ECLIA) performed on Cobas analyzer systems (e.g., e601, e801), which integrates all assay steps from sample aspiration to result calculation [5] [10].
Table 1: Core Methodological and Technical Specifications
| Feature | AMH Gen II ELISA (Beckman Coulter) | Elecsys Cobas AMH Assay (Roche) |
|---|---|---|
| Technology Platform | Manual ELISA | Automated Electrochemiluminescence Immunoassay (ECLIA) |
| Assay Workflow | Manual steps: pipetting, washing, incubation | Fully automated on Cobas systems |
| Antibody Targets | Capture: AMHN,229 region; Detection: AMHC region [9] | Capture: AMHN,229 region; Detection: AMHC region [9] |
| Primary Calibrator | Immunotech AMH reference preps [11] | Proprietary Roche calibrators |
| Measuring Range | 3–70 pmol/L (without dilution) [5] | 0.5–160 pmol/L (without dilution) [5] |
| Limit of Quantitation | 3.0 pmol/L [5] | 0.5 pmol/L [5] |
| Sample Throughput | Lower (batch processing) | High (random access) |
| Interassay CV% | 5.2-10.3% (controls); >30% (clinical samples <10 pmol/L) [5] | 2.8-3.3% (controls); ~28% (clinical samples <10 pmol/L) [5] |
Diagram 1: A comparison of the core workflows for the manual ELISA and automated ECLIA methods.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials and Their Functions
| Item | Function in Analysis | Specific Examples / Notes |
|---|---|---|
| AMH Gen II ELISA Kit | Manual quantification of AMH in serum/plasma. | Includes microtiter plate, standards, controls, detection antibodies. Requires manual protocol [5]. |
| Elecsys Cobas AMH Reagents | Automated quantification on Cobas platforms. | Pre-packaged, ready-to-use reagents including AMH biotinylated antibody, AMH ruthenylated antibody [10]. |
| Cobas Analyzer Systems | Fully automated immunoassay analysis. | e601, e801 models; integrated sample handling, dispensing, and detection [5] [10]. |
| Sample Diluents | Dilution of samples exceeding analytical range. | Critical for minimizing matrix effects; different diluents recommended for each assay [2]. |
| Quality Control (QC) Material | Monitoring assay precision and accuracy. | Human recombinant AMH or pooled human serum at multiple concentrations [5] [2]. |
| Calibrators | Defining the standard curve for concentration calculation. | Traceable to different master calibrators, a source of inter-assay bias [11]. |
Experimental Performance Data and Protocols
A direct comparison study by an independent research group provides robust data on the relative performance of these two assays. The following section details the experimental protocol and summarizes the key findings.
Key Experimental Protocol
- Study Design: A prospective observational pilot study.
- Cohort: 23 women with a median age of 36 years, who underwent laparoscopic sterilization.
- Sample Collection: Blood samples were collected preoperatively, as well as 1 week and 1, 3, and 6 months postoperatively. This yielded a total of 112 serum samples.
- Sample Handling: Serum was isolated within 4 hours of sampling and stored at -80°C until batch analysis.
- AMH Measurement: All samples were analyzed using both the AMH Gen II ELISA and the Elecsys Cobas AMH methods at the Department of Clinical Biochemistry, Aarhus University Hospital (ISO 15189 accredited).
- AMH Gen II ELISA: A standard manual protocol was used, with samples analyzed in duplicate. The acceptance CV for internal controls was <11–14%.
- Elecsys Cobas AMH: Analysis was performed on a Cobas 6000 e601 platform using Roche's standard automated protocol. The acceptance CV for internal controls was <5%.
- Statistical Analysis: Correlation with antral follicle count (AFC) was determined using Spearman's test. Method comparison was performed using Passing-Bablok regression and Bland-Altman plots [5].
Table 3: Summary of Comparative Analytical Performance Data
| Performance Metric | AMH Gen II ELISA (Beckman Coulter) | Elecsys Cobas AMH Assay (Roche) | Interpretation |
|---|---|---|---|
| Correlation with AFC | 0.86 (at baseline) [5] | 0.83 (at baseline) [5] | Both assays show strong and comparable clinical correlation. |
| Systematic Bias | Reference Method | ~32% lower than ELISA [5] | Consistent negative bias for Elecsys; results not interchangeable. |
| Analytical Variability (CV%) | 5.5% - 10.3% (controls) [5] | 2.8% - 3.3% (controls) [5] | Automated assay demonstrates superior precision. |
| Total Variance (CV%) in Clinical Samples <10 pmol/L | 31.9% [5] | 28.0% [5] | Lower total variance for the automated assay, crucial for low levels. |
| Meets Optimal Performance Goal (CVA<0.25*CVWithin-Biol) | Only at high AMH concentrations [5] | Throughout the measuring range [5] | Automated assay fulfills optimal performance standards more consistently. |
The Elecsys assay demonstrated excellent precision, with a repeatability CV of 1.8% and an intermediate precision CV of 4.4% in a separate multicenter evaluation [10]. This performance is a marked improvement over the manual ELISA, particularly at low AMH concentrations, which is critical for assessing diminished ovarian reserve.
Discussion and Clinical Implications
Standardization and Inter-Assay Correlation
A central challenge in AMH measurement is the lack of assay standardization. Despite using antibodies targeting the same regions (AMHN,229 and AMHC) [9], the Elecsys assay consistently reports values 24–32% lower than the AMH Gen II ELISA [5] [10]. This systematic bias underscores that results from these assays are not directly interchangeable, and clinicians must use assay-specific reference intervals.
Interassay correlation is also level-dependent. A large 2022 study found that while correlations between the Gen II, Elecsys, and other assays are strong over a broad range, they can become only moderate in specific mid-range concentrations (2.80–7.04 ng/mL) [9]. This highlights the need for caution when comparing serial measurements performed on different platforms.
Impact on Clinical Applications and Decision-Making
The superior analytical performance of the Elecsys Cobas AMH assay has tangible clinical benefits:
- Robust Ovarian Reserve Assessment: The combination of low variability and a low limit of quantitation (0.5 pmol/L) makes the automated assay particularly reliable for tracking patients with low ovarian reserve and for monitoring the impact of gonadotoxic treatments like chemotherapy [5] [11].
- Accurate Cut-off Values: The automated assay enables the establishment of robust clinical cut-offs. For example, an AMH value of 1.77 ng/mL measured by the Elecsys assay corresponds to an antral follicle count (AFC) >15 with high sensitivity (88.34%) and specificity (68.29%) [12].
- Diagnostic Consistency in PCOS: The precision of the automated assay at high AMH levels is advantageous for evaluating women with suspected PCOS, where accurate high-end measurement is critical [9] [10].
Diagram 2: The logical relationship between assay characteristics and their downstream impact on clinical application and decision-making.
The evidence demonstrates a clear paradigm shift in AMH immunoassay technology. The Elecsys Cobas AMH assay represents a significant advancement over the manual AMH Gen II ELISA, offering a fully automated workflow, superior analytical precision, a wider measuring range, and a lower limit of quantitation. These features make it highly suitable for the high-throughput, precision-driven demands of modern clinical and research laboratories.
However, the persistent bias between methods reinforces the critical need for assay-specific interpretation and underscores the ongoing challenge of a lack of standardization across the industry. For researchers and drug development professionals, the choice of assay must be guided by the required precision, the specific clinical or research question, and the availability of appropriate reference data. The technological shift to automation ultimately provides a more reliable and efficient tool for advancing women's health.
In the realm of immunoassays, the accurate quantification of biomarkers is foundational to both clinical diagnostics and biomedical research. For years, the Enzyme-Linked Immunosorbent Assay (ELISA) has been the established standard, prized for its specificity and reliability. The emergence of electrochemiluminescence (ECL) technology, exemplified by platforms like the Elecsys system (Roche), presents a modern alternative offering automation and enhanced throughput. Within the specific context of Anti-Müllerian Hormone (AMH) testing—a critical marker for ovarian reserve—this guide provides an objective comparison between the traditional AMH Gen II ELISA and the automated Elecsys Cobas AMH assay. The performance data and experimental methodologies outlined herein are intended to assist researchers, scientists, and drug development professionals in making informed technological choices.
Fundamental Principles and Methodologies
Core Technological Mechanisms
The fundamental distinction between these techniques lies in their detection mechanism.
ELISA (Enzyme-Linked Immunosorbent Assay) is a plate-based technique that relies on an enzyme-mediated color change. The target antigen is captured by an immobilized antibody, and a second enzyme-conjugated detection antibody is then added. The subsequent addition of a substrate produces a colored product, with the color intensity, measurable via absorbance, being proportional to the amount of target analyte [13].
Electrochemiluminescence (ECL) Immunoassays, such as the Elecsys platform, utilize a label that emits light upon electrochemical stimulation. In this process, a ruthenium complex label is excited by an electrical current applied via electrodes in the assay plate, leading to light emission at a specific wavelength [14] [15]. This light is then quantified by a detector.
The diagram below illustrates the core procedural and detection pathways for both methods.
Experimental Protocols for AMH Assay Comparison
The following standard operating procedures are derived from a clinical comparison study that evaluated AMH levels across both platforms [5] [16].
Protocol for AMH Gen II ELISA (Beckman Coulter):
- Sample Preparation: A pre-mixture of clinical samples, calibrators, and controls in a specified assay buffer is prepared.
- Assay Procedure: The technique is a manual process. Samples are analyzed in duplicate, requiring a 100 µl aliquot per well. The total sample volume required for duplicate measurements of AMH is 400 µl.
- Data Analysis: The middle value of the duplicate results is used. If the results from the duplicates differ by more than 15%, the analysis must be repeated. The established limit of quantitation (LOQ) for this assay is 3.0 pmol/L [5] [16].
Protocol for Elecsys Cobas AMH Assay (Roche):
- Sample Preparation: Serum samples are processed and can be loaded directly onto the automated system with minimal pre-treatment.
- Assay Procedure: The assay runs on a Cobas 6000 e601 platform using Roche's standard, fully automated protocol. A smaller sample volume of 25 µl is sufficient for a single test.
- Data Analysis: The system automatically calculates concentrations based on the calibration curve. The LOQ for this assay is significantly lower, at 0.5 pmol/L [5] [16].
Comparative Performance Data
A direct comparison within a clinical series reveals critical differences in assay performance, particularly regarding precision and bias.
Table 1: Key Performance Indicators of AMH Immunoassays
| Performance Characteristic | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Analytical Variability (CV%) [5] [16] | 5.5% - 10.3% | 2.8% - 3.3% |
| Limit of Quantitation (LOQ) [5] [16] | 3.0 pmol/L | 0.5 pmol/L |
| Sample Volume per Test [5] [16] | 100 µl | 25 µl |
| Assay Processing Time [14] | ~18 hours | ~3.5 hours |
| Measured Bias [5] [16] | Reference Method | Approximately 32% lower |
Table 2: Operational and Diagnostic Correlation Data
| Comparison Metric | AMH Gen II ELISA | Elecsys Cobas AMH | Implication |
|---|---|---|---|
| Correlation with Antral Follicle Count (AFC) [5] [16] | r = 0.86 (Baseline) | r = 0.83 (Baseline) | Both assays show strong and comparable correlation with AFC. |
| Samples Below LOQ [5] [16] | 15% | 2% | The ECL assay maximizes data detection, especially at low concentrations. |
| Achievement of Optimal Performance Goal* [5] [16] | Only in high measuring range | Throughout the measuring range | The ECL assay meets stringent precision standards more consistently. |
*Optimal performance defined as CVAnalytical < 0.25 * CVWithin Biological Variation [5] [16].
The Scientist's Toolkit: Essential Research Reagents and Materials
The successful execution and validation of these immunoassays depend on a suite of critical reagents and materials.
Table 3: Key Research Reagent Solutions
| Item | Function in the Assay | Application Notes |
|---|---|---|
| Coated Microplates | Solid phase for immobilizing the capture antibody or streptavidin-biotin complex. | Plates for ECL (e.g., from Meso Scale Discovery) often have integrated electrodes [14]. |
| Calibrators and Controls | Used to generate the standard curve and validate assay performance across runs. | Source and purity of peptides (recombinant human vs. bovine) can cause absolute concentration differences between platforms [14] [17]. |
| Capture and Detection Antibodies | Define the specificity of the assay by binding to unique epitopes on the target analyte. | Antibody affinity and the specific epitopes targeted (e.g., pro-region vs. mature region of AMH) are major sources of inter-assay variation [14] [18]. |
| Detection Label (Enzyme or Ruthenium) | Generates the measurable signal (color or light). | Horseradish peroxidase (HRP) is common in ELISA; Ruthenium complexes are used in ECL [15]. |
| Assay Buffer and Diluents | Maintain pH and ionic strength, and minimize non-specific binding. | Matrix effects (e.g., serum vs. tear fluid) must be validated for different sample types [19]. |
The comparison between conventional ELISA and electrochemiluminescence technology reveals a nuanced landscape. The AMH Gen II ELISA is a well-characterized, manual method that provides reliable data but is accompanied by higher analytical variability, a more limited measuring range, and longer processing times. In contrast, the Elecsys Cobas AMH ECL assay offers a fully automated solution with superior precision, a wider dynamic range, and a significantly lower limit of detection, which is crucial for measuring low analyte levels. The observed consistent bias between the two methods underscores that their results are not directly interchangeable. The choice between them should be guided by specific research needs: ELISA may suffice for standard measurements, whereas ECL technology is better suited for high-throughput, precision-sensitive environments requiring maximum sensitivity.
The Clinical Imperative for Accurate and Standardized AMH Measurement
Anti-Müllerian Hormone (AMH) has emerged as a fundamental biochemical marker in reproductive medicine, providing crucial insights into ovarian reserve and playing an increasingly important role in fertility treatment planning and outcome prediction [5] [20]. As a direct serum marker of functional ovarian reserve, AMH levels assist clinicians in assessing ovarian reserve in conjunction with other clinical and laboratory findings before initiating any fertility therapy [7]. The reliability of AMH measurement, however, is fundamentally dependent on the analytical performance of the immunoassays used, creating an imperative for both accuracy and standardization across testing platforms [5] [18].
The evolution of AMH testing from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated immunoassay systems represents a significant advancement in reproductive endocrinology [20] [6]. This guide provides a comprehensive comparison between two principal methodologies: the established AMH Gen II ELISA manual assay and the automated Elecsys Cobas AMH assay, presenting experimental data to inform researchers, scientists, and drug development professionals about the critical performance characteristics of each system.
Methodological Comparison: Assay Architectures and Workflows
AMH Gen II ELISA Assay
The AMH Gen II ELISA is an enzymatically amplified two-site manual immunoassay that employs a standard manual technique requiring pre-mixture of clinical samples, calibrators, and controls in assay buffer before addition to the microplate [5]. This method utilizes antibodies directed against epitopes in both the mature AMHN,C and proAMH molecules, measuring these two entities in a similar but not identical manner [21]. The protocol was revised in 2013 with an additional pre-mixing step to address complement interference issues that caused falsely low readings in the original version [20] [21]. The manual nature of this assay introduces multiple handling steps that can contribute to analytical variation.
Elecsys Cobas AMH Assay
The Elecsys Cobas AMH assay represents the first fully automated AMH assay cleared by the FDA, designed for use on Roche immunoassay systems including the cobas e 411, e 601, e 602, and e 801 analyzers [7]. This automated system uses conformational epitope antibodies in a sandwich immunoassay format, significantly reducing manual handling and associated variability [18]. The assay requires minimal sample volume (as low as 30 μL) and provides rapid results with an 18-minute run time, enabling same-day reporting for in-house testing [7].
Experimental Workflow Comparison
The diagram below illustrates the key procedural differences between the two assay methodologies:
Analytical Performance: Comparative Experimental Data
Precision and Analytical Variability
Multiple studies have demonstrated significant differences in analytical precision between the manual and automated AMH assays. A prospective observational study comparing the two methods found substantially higher analytical variability with the AMH Gen II ELISA, particularly at lower concentrations [5].
Table 1: Analytical Performance Comparison Between AMH Gen II ELISA and Elecsys Cobas AMH Assays
| Performance Parameter | AMH Gen II ELISA | Elecsys Cobas AMH | Experimental Context |
|---|---|---|---|
| Analytical Variability (CV%) | 5.5-10.3% | 2.8-3.3% | Control samples analyzed on consecutive days over 3 months [5] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L (0.42 ng/mL) | 0.5 pmol/L (0.07 ng/mL) | Lowest concentration quantifiable with CV% <15% [5] |
| Measuring Range | 3-70 pmol/L (0.42-9.8 ng/mL) | 0.5-160 pmol/L (0.07-22.4 ng/mL) | Without dilution [5] [7] |
| Inter-assay Imprecision | CV 5.6% | Total imprecision <5% | Manufacturer reported performance [7] [22] |
| Bias Between Methods | Reference method | Approximately 32% lower values | 112 serum samples from 23 women [5] |
Correlation with Ovarian Reserve Markers
Both assays demonstrate significant correlation with established markers of ovarian reserve, particularly antral follicle count (AFC). In clinical studies, the correlation between AMH values and AFC was comparable between methods, with Spearman correlation coefficients of 0.86 for ELISA AMH vs. AFC and 0.83 for Cobas AMH vs. AFC at baseline measurements [5]. This indicates that despite the systematic bias between methods, both maintain clinical utility for ovarian reserve assessment.
Sample Stability and Pre-analytical Considerations
Pre-analytical factors significantly impact AMH measurement reliability. Studies comparing the reproducibility of AMH measurements across various blood/serum storage conditions have demonstrated superior stability with the automated Access AMH assay (which shares technological similarities with Elecsys) compared to the revised Gen II assay [22].
Table 2: Pre-analytical Sample Stability Under Different Storage Conditions
| Storage Condition | AMH Gen II ELISA Performance | Elecsys/Automated Assay Performance | Study Details |
|---|---|---|---|
| Serum stored at -20°C for 48 hours | Significantly lower than fresh controls | Comparable to fresh controls | 23 samples, paired Wilcoxon test [22] |
| Serum stored at 0-4°C for 48 hours | Significantly lower than fresh controls | Comparable to fresh controls | 22 samples [22] |
| Delayed serum separation (48 hours) | Significantly lower than controls | Varied results | 24 samples [22] |
| Long-term storage (-20°C for 2 years) | Significantly higher than controls | Significantly higher than controls | 74 samples [22] |
Clinical Performance: Predictive Value in Fertility Treatments
Prediction of Oocyte Yield
The fundamental clinical application of AMH measurement in reproductive medicine lies in predicting ovarian response to controlled ovarian stimulation (COS). Both assays demonstrate good correlation with oocyte yield following COS, with the automated Elecsys Cobas assay showing performance characteristics suitable for clinical application [5] [20].
A comprehensive study of 3,296 IVF cycles demonstrated that AMH levels measured by automated assay showed a good correlation with the number of oocytes retrieved following COS (R = 0.655) [20]. Receiver operating characteristic (ROC) analysis revealed that the sensitivity of AMH for predicting ovarian response was comparable with or lower than that of estradiol but higher than that of FSH, confirming its utility as a marker of ovarian reserve [20].
Performance in Special Populations
Recent advancements in AMH assay technology have focused on improving sensitivity for special populations, particularly women with low ovarian reserve. Novel high-specificity assays employing linear-epitope antibodies demonstrate enhanced predictive accuracy for oocyte yield in women with diminished ovarian reserve [18]. While the Elecsys assay uses conformational epitope antibodies, these emerging technologies highlight the ongoing evolution toward more precise AMH measurement [18].
For patients with primary ovarian insufficiency (POI), highly sensitive AMH assays with limits of detection as low as 1.3 pg/mL (versus 20 pg/mL for the Access AMH assay) have shown promise in predicting follicular development where conventional assays lack sufficient sensitivity [23].
Research Reagent Solutions: Essential Materials for AMH Assay Evaluation
Table 3: Key Research Reagents and Materials for AMH Assay Comparative Studies
| Reagent/Material | Function | Specification Considerations |
|---|---|---|
| Calibrators | Establish standard curve for quantification | Traceable to international standards; matrix-matched to patient samples |
| Quality Control Materials | Monitor assay precision and accuracy | Multiple concentration levels (low, medium, high) covering clinical decision points |
| Sample Collection Tubes | Maintain sample integrity | Serum separator gel tubes; consideration of gel barrier integrity during centrifugation |
| Assay Buffers | Provide optimal binding conditions | Must include components to eliminate complement interference in ELISA methods |
| Detection Reagents | Generate measurable signal | Enzymatic (ELISA) vs. electrochemiluminescent (Elecsys) detection systems |
| Wash Solutions | Remove unbound materials | Automated washing systems provide superior consistency versus manual methods |
Standardization Challenges and Future Directions
The lack of harmonization between different AMH assays remains a significant challenge in both clinical practice and research. Current AMH assays are not yet harmonized through programs such as the International Consortium for Harmonization of Clinical Laboratory Results, leading to substantial variation in test results among different assay platforms [8]. Studies have consistently demonstrated that AMH concentrations measured using automated assays tend to be lower than those measured using ELISAs, though the degree of difference varies [8].
Future directions in AMH assay development focus on improving specificity through antibodies targeting distinct molecular isoforms of AMH, potentially enhancing predictive accuracy for ovarian response, particularly in challenging populations such as women with low ovarian reserve [18]. The development of international reference materials and standardized calibration approaches will be crucial for achieving true harmonization across testing platforms.
The transition from manual AMH Gen II ELISA to automated platforms like the Elecsys Cobas AMH assay represents significant progress in analytical performance, with demonstrated advantages in precision, sensitivity, and workflow efficiency. The approximately 32% systematic bias between methods underscores the critical importance of method-specific reference intervals and the imperative for clinical laboratories to maintain consistency in testing platforms.
For researchers and drug development professionals, these methodological differences have substantial implications for study design, data interpretation, and comparison of results across different studies. The enhanced analytical performance of automated systems, particularly improved precision and lower limits of quantitation, supports their adoption for both clinical applications and research investigations requiring the highest level of analytical reliability.
As AMH continues to solidify its role as a cornerstone of ovarian reserve assessment, ongoing efforts toward assay standardization and the development of even more specific detection methods promise to further enhance the clinical utility of this important biomarker in reproductive medicine.
Analytical Procedures and Workflow Comparison
Anti-Müllerian Hormone (AMH) has emerged as a crucial biomarker for assessing ovarian reserve in reproductive medicine. The AMH Gen II ELISA (Enzyme-Linked Immunosorbent Assay) represents a significant methodological advancement in quantifying AMH levels, yet requires thorough understanding of its manual protocol and technical demands. This comprehensive guide examines the AMH Gen II ELISA's manual methodology against emerging automated alternatives, particularly the Elecsys Cobas AMH and Access AMH assays. We provide detailed experimental protocols, performance comparisons, and technical specifications to inform researchers and clinical laboratories about the practical considerations for implementing AMH testing. Data presented herein reveal that while the AMH Gen II ELISA establishes a foundational standard with good correlation to ovarian reserve markers, automated systems demonstrate superior precision, reduced hands-on time, and enhanced performance in critical clinical ranges, particularly for patients with diminished ovarian reserve.
The AMH Gen II ELISA, developed by Beckman Coulter, represents the second generation of AMH immunoassays designed to measure AMH levels in human serum and plasma. This assay was developed using two monoclonal antibodies directed to epitopes in the mature region of the AMH molecule, which provides greater stability against proteolysis compared to the proregion [24]. The AMH Gen II assay was standardized to the earlier Immunotech AMH assay, demonstrating excellent correlation (r = 0.98) with a slope of 1.0 when compared using 120 serum samples across the range of 0-20.4 ng/mL [24].
The clinical significance of AMH measurement has expanded substantially, serving as an ideal marker in reproductive medicine that provides information on ovarian reserve, particularly relevant in family planning, infertility therapy, and menopause onset assessment [25]. AMH has also proven valuable in diagnosing certain pathologies including granulosa cell tumors and polycystic ovary syndrome (PCOS) [25]. Unlike other hormonal markers that fluctuate dramatically throughout the menstrual cycle, AMH maintains relative stability with no major short-term changes, making it uniquely valuable for ovarian reserve assessment without cycle timing constraints [20] [2].
Detailed Manual Protocol of AMH Gen II ELISA
Principle and Antibody Configuration
The AMH Gen II ELISA operates on a two-step sandwich-type enzymatic microplate assay principle. The assay employs a pair of monoclonal antibodies that bind to distinct epitopes in the mature region of the AMH molecule. This strategic targeting of the mature region enhances assay stability as this region is more resistant to proteolysis compared to the proregion, partly due to its multiple cystine residues [24]. The mature region also demonstrates the greatest degree of homology between species, with 108 of the last 112 residues being conserved between rat and human sequences [24].
Step-by-Step Workflow
The manual AMH Gen II ELISA protocol requires precise execution of multiple hands-on steps:
Plate Coating: Microtiter plates are coated with the F2B 12H/E monoclonal antibody, purified by Protein G affinity chromatography, which serves as the capture antibody [24].
Sample Preparation: Clinical samples, calibrators, and controls must be manually prepared using the AMH Gen II Assay Buffer. The protocol employs a pre-mixture of clinical samples, calibrators and controls in assay buffer before application to the plate [5]. This pre-mixing step was specifically introduced to eliminate complement interference issues that plagued the original Gen II assay [20].
Incubation and Reaction: The assay requires 20 μL of sample per test and is completed in less than 3 hours total incubation time [24]. The specific incubation conditions for the Gen II ELISA involve room temperature incubation with precise timing at each step.
Washing and Detection: After incubation with samples and detection antibodies, the plate undergoes multiple washing steps before addition of substrate solution. The enzyme reaction is then stopped, and absorbance measured at 450 nm [25].
Calculation: AMH concentrations are determined by interpolation from a calibrator curve ranging from 0.16 to 22.5 ng/mL [24].
Quality Control Parameters
The manual protocol requires analysis in duplicate, with repeat analysis mandated when duplicate results differ by more than 15% [5]. The limit of acceptance for daily internal controls is CV < 11-14%, and the limit of quantitation is 3.0 pmol/L, with a measuring range of 3-70 pmol/L without dilution [5].
Figure 1: AMH Gen II ELISA Manual Workflow. This diagram illustrates the multi-step manual process required for the AMH Gen II ELISA protocol, highlighting the extensive hands-on time and quality control requirements.
Hands-On Time and Technical Demands
Labor-Intensive Methodology
The manual nature of the AMH Gen II ELISA imposes significant hands-on time requirements and technical demands on laboratory personnel. Unlike automated systems, the Gen II ELISA requires:
- Manual sample pre-processing including the pre-mixing step with assay buffer [5]
- Precise manual pipetting of samples, controls, and reagents into microtiter plates
- Multiple incubation steps requiring precise timing and manual handling
- Repetitive washing steps that must be consistently performed across all wells
- Duplicate measurements with repeat analysis when CV exceeds 15% [5]
The total assay time approximately 3 hours, during which skilled technicians must be actively engaged in the process [24]. This extensive hands-on requirement increases the potential for technical error and inter-operator variability compared to automated systems.
Technical Expertise Requirements
The execution of AMH Gen II ELISA demands substantial technical expertise as the method is characterized as a "standard manual technique" [5]. Skilled technicians are required to perform the analyses, and the laboratory must maintain specific qualifications such as ISO 15189 accreditation standard for clinical laboratories to ensure reliable results [5]. The manual technique inherently introduces greater analytical variation compared to automated methods, with documented analytical variability ranging from 5.5 to 10.3% across the measuring range [5].
Performance Comparison with Automated Alternatives
Precision and Analytical Variability
Direct comparison studies reveal substantial differences in analytical performance between manual and automated AMH assays:
Table 1: Analytical Performance Comparison Across AMH Assay Platforms
| Performance Parameter | AMH Gen II ELISA (Manual) | Elecsys Cobas AMH (Automated) | Access AMH (Automated) |
|---|---|---|---|
| Total Analytical Variability (CV%) | 5.5-10.3% [5] | 2.8-3.3% [5] | 2.4-5.2% [6] |
| Limit of Quantitation | 3.0 pmol/L [5] | 0.5 pmol/L [5] | 0.010 ng/mL [6] |
| Measuring Range | 3-70 pmol/L [5] | 0.5-160 pmol/L [5] | Up to 24 ng/mL [6] |
| Bias vs. Gen II ELISA | Reference | -20% [26] | -16% [26] |
| Sample Throughput | Manual, batch processing | Fully automated, random access | Fully automated, random access |
The data demonstrate that automated systems provide superior precision throughout the measuring range, with the Elecsys Cobas method achieving optimal performance standards (CVAnalytical < 0.25* CVWithin Biological Variation) across all concentrations, while the AMH Gen II only achieved optimal performance at the high end of the measuring range [5].
Correlation with Ovarian Reserve Markers
Both manual and automated AMH assays show strong correlation with antral follicle count (AFC), a key marker of ovarian reserve:
Table 2: Clinical Correlation with Antral Follicle Count (AFC)
| Assay Type | Overall Correlation with AFC (r) | Correlation in Low AFC Patients (r) | Correlation in High AFC Patients (r) |
|---|---|---|---|
| AMH Gen II ELISA | 0.83 [26] | 0.52 [26] | Strong correlation [26] |
| Elecsys Cobas AMH | 0.83 [26] | 0.65 [26] | Strong correlation [26] |
| Access AMH | 0.83 [26] | 0.63 [26] | Strong correlation [26] |
Remarkably, while overall correlation with AFC is identical across platforms (r = 0.83), automated assays demonstrate significantly stronger correlation in patients with reduced antral follicle count (AFC 3-12 follicles), a critical patient population where precise ovarian reserve assessment is most crucial [26].
Method Correlation and Standardization
Correlation studies between the Gen II ELISA and automated platforms reveal consistent biases but strong overall correlations:
- The Access AMH assay shows excellent correlation with the Gen II ELISA (R² = 0.9822-0.9832) with a slope of 0.89-0.92 in regression analysis [2] [6]
- The Elecsys Cobas assay demonstrates approximately 32% lower values compared to the Gen II ELISA, with a tendency of increased bias in the high concentration range [5]
- The Access AMH assay yields values that are 0.711-0.755 times those obtained with the Gen II pre-mix assay [20]
These consistent biases highlight the importance of method-specific reference ranges and caution when comparing results across different assay platforms or transitioning from manual to automated methods.
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Reagents for AMH Gen II ELISA Implementation
| Reagent/Material | Specification | Function in Protocol |
|---|---|---|
| Coated Microtiter Plates | F2B 12H/E monoclonal antibody coated plates [24] | Capture and immobilize AMH molecules |
| AMH Gen II Assay Buffer | Specific buffer for sample pre-mixing [5] | Eliminate complement interference during sample preparation |
| Calibrators | 0.16, 0.4, 1.2, 4.0, 10, and 22.5 ng/mL in bovine calf serum [24] | Establish standard curve for quantification |
| Quality Controls | Two levels of internal controls [5] | Monitor assay performance and precision |
| Detection Antibodies | Monoclonal antibodies to mature AMH region [24] | Bind captured AMH for detection |
| TMB Substrate | Tetramethylbenzidine substrate solution [25] | Enzymatic color development for measurement |
| Wash Buffer | Specific buffer for plate washing [5] | Remove unbound materials between steps |
The AMH Gen II assay components are designed to be ready-to-use (except wash solution which requires preparation), and the assay demonstrates no cross-reactivity to relevant substances [25]. The antibodies used in this assay do not infringe on Beckman Coulter patents as they do not bind to the patented region of AMH [25].
Discussion and Clinical Implications
Evolution from Manual to Automated Platforms
The development of automated AMH assays represents a significant advancement in reproductive endocrinology testing. The transition from manual ELISA to automated immunoassay systems has addressed several limitations inherent in the Gen II method. Automated platforms provide:
- Substantially reduced hands-on time through complete automation of pipetting, incubation, and washing steps
- Improved precision with minimal inter-operator variability
- Enhanced sensitivity particularly in the low range critical for diminished ovarian reserve assessment
- Random access capability enabling single-sample testing without batch requirements
Despite the performance advantages of automated systems, the AMH Gen II ELISA established the foundational standard for AMH measurement, with its antibodies incorporated into the automated platforms from both Roche and Beckman Coulter [2]. The correlation between methods, though strong, demonstrates consistent biases necessitating method-specific reference intervals and careful interpretation when transitioning between platforms.
Practical Considerations for Laboratory Implementation
Laboratories must consider several practical aspects when selecting AMH testing platforms:
- Volume and Workflow: The manual Gen II ELISA suits lower-volume laboratories with batch-testing workflows, while automated systems better serve high-volume settings requiring random access testing
- Technical Expertise: Manual methods require significantly more skilled technologist time and expertise to maintain quality
- Clinical Application: Automated assays demonstrate superior performance in the low AMH range critical for predicting poor ovarian response [26]
- Regulatory Status: While the Gen II ELISA is widely established, newer automated assays carry appropriate regulatory approvals (CE marking, FDA clearance) for clinical use [25]
The predictive value of AMH for clinical pregnancy outcomes, particularly in women of late reproductive age (AUC = 0.62-0.69 in women over 35), underscores the importance of precise and reliable AMH measurement regardless of platform [27].
The AMH Gen II ELISA represents a historically significant methodology that established AMH as a crucial biomarker in reproductive medicine. Its detailed manual protocol, while technically demanding and time-intensive, provided the foundation for current understanding of AMH's role in assessing ovarian reserve. The comprehensive performance data presented herein demonstrates that while the Gen II ELISA shows good correlation with ovarian reserve markers like AFC, newer automated platforms offer substantial advantages in precision, particularly in the clinically critical low range, while significantly reducing hands-on time and technical variability. Laboratories must carefully consider their testing volume, technical capabilities, and clinical needs when selecting between these platforms, acknowledging both the robust heritage of the manual ELISA and the operational advantages of contemporary automated systems. As AMH continues to gain importance in predicting treatment outcomes in assisted reproduction, particularly for women of advanced reproductive age, the evolution from manual to automated methods represents a significant advancement in reproductive endocrine testing.
In the field of clinical reproductive endocrinology, the accurate and reliable measurement of Anti-Müllerian Hormone (AMH) has become indispensable for assessing ovarian reserve. As a glycoprotein produced by granulosa cells of preantral and small antral follicles, AMH serves as a direct quantitative marker for the ovarian follicle pool [18] [28]. The evolution of AMH testing methodologies from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated immunoassays represents a significant advancement in laboratory medicine, addressing the growing clinical demand for precision, efficiency, and standardization.
The Elecsys Cobas AMH assay (Roche Diagnostics) emerged as the first fully automated AMH assay cleared by the FDA, representing a paradigm shift in AMH testing methodology [7]. This review provides a comprehensive technical comparison between the Elecsys Cobas AMH automated assay and the traditional AMH Gen II ELISA method, focusing on analytical performance, operational efficiency, and clinical utility within the context of ovarian reserve assessment.
Technical Comparison: Analytical Performance
Precision and Analytical Variability
A critical prospective observational study directly compared the analytical performance of the Elecsys Cobas AMH assay and the AMH Gen II ELISA method, revealing substantial differences in precision and variability [5]. The researchers established analytical goals based on biological variation, with optimal performance defined as CVAnalytical < 0.25* CVWithin Biological Variation.
Table 1: Analytical Performance Comparison Between AMH Assays
| Performance Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Overall Analytical Variability (CV%) | 5.5 - 10.3% | 2.8 - 3.3% |
| Control 1 (High Level ~40 pmol/L) | 5.2% | 3.3% |
| Control 3 (Low Level ~7 pmol/L) | 10.3% | 2.8% |
| Limit of Quantitation (LOQ) | 3.0 pmol/L | 0.5 pmol/L |
| Measuring Range | 3-70 pmol/L | 0.5-160 pmol/L |
| Samples Unable to be Quantitated | 15% | 2% |
| Achievement of Optimal Performance Goals | Only at high end of measuring range | Throughout entire measuring range |
The data demonstrate the Elecsys Cobas system's superior precision, particularly at lower AMH concentrations commonly encountered in clinical populations with diminished ovarian reserve [5]. The significantly lower limit of quantitation (0.5 pmol/L versus 3.0 pmol/L) extends the clinical utility of the Elecsys assay to patient populations with very low AMH levels, such as those with primary ovarian insufficiency or Turner syndrome [28].
Method Comparison and Correlation
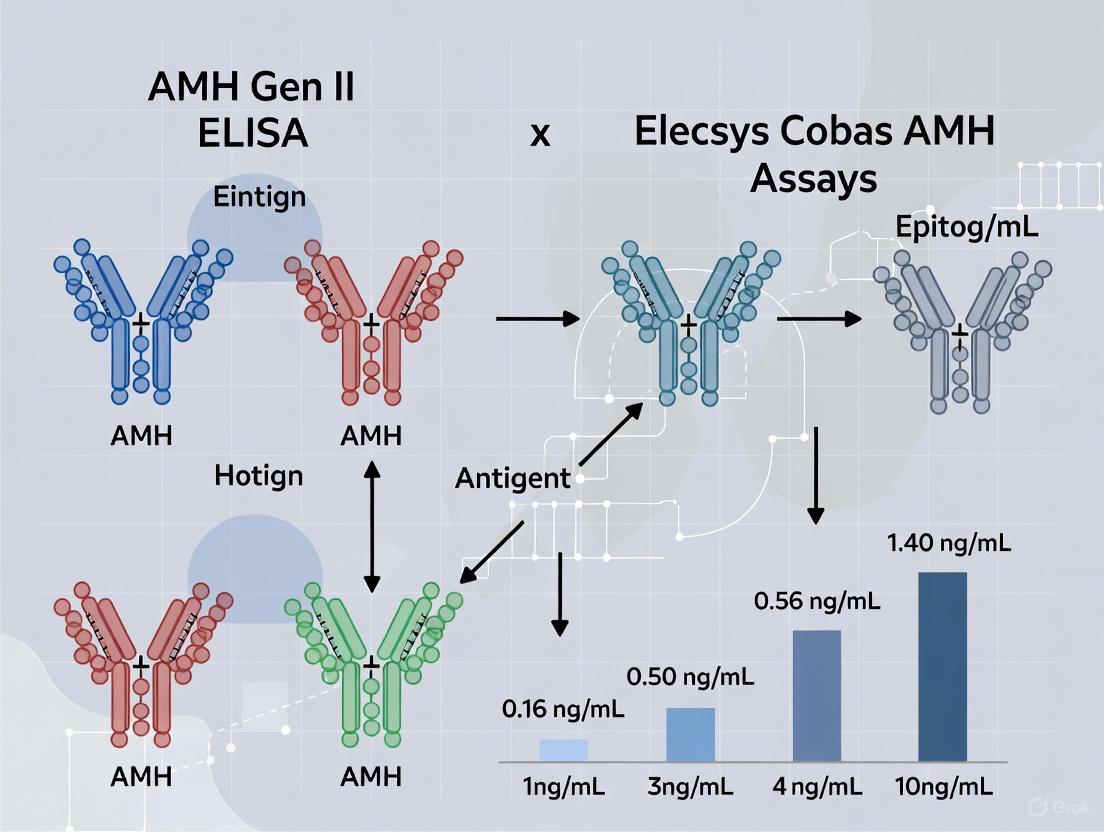
When comparing AMH values between the two methods, researchers observed a strong correlation but with a consistent proportional bias. The Elecsys Cobas AMH assay yielded concentrations approximately 32% lower than those obtained with the AMH Gen II ELISA across the measuring range [5]. This bias exhibited a tendency to increase at higher AMH concentrations, highlighting the importance of method-specific reference intervals and the inability to use results interchangeably between platforms.
The correlation between the two methods was further validated in independent studies developing novel AMH detection technologies. One such study developing a homogeneous light-initiated chemiluminescence assay (LICA) demonstrated excellent correlation with the Elecsys Cobas method (y = 0.9851x + 0.07147, R² = 0.9569) [29].
Operational Characteristics and Throughput
Automation and Workflow Integration
The Elecsys Cobas AMH assay is designed for integration across Roche's immunoassay analyzer portfolio, including the cobas e 411, e 601, e 602, and e 801 systems [7]. This platform approach enables laboratories to match throughput capacity with workload demands while maintaining consistent performance characteristics across instruments.
Table 2: Instrument Specifications for Elecsys Cobas AMH Assay
| Instrument Platform | Throughput | Reagent Positions | Sample Material | Sample Volume |
|---|---|---|---|---|
| cobas e 411 analyzer | Up to 86 tests/hour | 18 | Serum, plasma, urine | As low as 30 μL |
| cobas e 801 analytical unit | Up to 300 tests/hour | 48 | Serum, plasma, urine | As low as 30 μL |
The fully automated nature of the Elecsys Cobas AMH assay eliminates manual processing steps required by the Gen II ELISA method, reducing technologist hands-on time and minimizing potential sources of pre-analytical error [5] [7]. The automated cassette management system on platforms like the cobas e 801 enables continuous operation with onboard reagent stability of up to 112 days (16 weeks), supporting operational efficiency in high-volume laboratory environments [30] [7].
Assay Protocol and Time to Results
The Elecsys Cobas AMH assay protocol is fully automated once samples are loaded onto the instrument. The assay utilizes ElectroChemiLuminescence (ECL) technology for heterogeneous immunoassays, with a total run time of approximately 18 minutes [7]. This represents a significant improvement over the manual ELISA method, which requires multiple incubation, washing, and development steps typically spanning several hours.
The disposable tips and carryover-free pipetting systems incorporated into Cobas analyzers maintain sample integrity throughout the testing process, with integrated clot and bubble detection systems further enhancing result reliability [30] [31].
Experimental Methodology in Comparative Studies
Study Design and Sample Processing
The foundational comparative study between the two methodologies employed a prospective observational design with 23 women undergoing laparoscopic sterilization [5]. Blood samples were collected at multiple time points: preoperatively, one week postoperatively, and at 1, 3, and 6 months postoperatively. This longitudinal design enabled assessment of both analytical and biological variability.
Serum samples were processed within 4 hours of collection through centrifugation and stored at -80°C until analysis [5]. All biochemical measurements were performed at an ISO 15189 accredited clinical biochemistry department by skilled technicians, ensuring standardized processing and minimizing pre-analytical variability.
Measurement Protocols
For the AMH Gen II ELISA, researchers followed the standard application protocol employing a pre-mixture of clinical samples, calibrators, and controls in assay buffer [5]. This manual technique required duplicate analysis with repeat testing if results differed by more than 15%. The acceptance criteria for internal quality control specified CV < 11-14%.
The Elecsys Cobas AMH measurements were performed on Cobas 6000 e601 platforms using Roche's standard automated protocol [5]. The automated system maintained more stringent internal quality control standards with CV < 5%, reflecting the enhanced precision of the automated system.
Statistical Analysis
Researchers employed Spearman's correlation test to evaluate the relationship between AMH levels and antral follicle count (AFC) [5]. Method comparison utilized Passing-Bablok regression and bias plots created using specialized statistical software (Analyse-it). Total variance was calculated as a combination of within-person biological variation and analytical variation using the formula: CVTOTAL = √[(CVWithin-person Biological Variation)² + (CVAnalytical)²].
Technological Basis and Detection Methods
Electrochemiluminescence Technology
The Elecsys Cobas AMH assay utilizes heterogeneous electrochemiluminescence immunoassay (ECLIA) technology, which combines immunochemical specificity with sensitive signal detection [30] [31]. This technology uses ruthenium complex-labeled antibodies that emit light upon electrochemical stimulation, providing a highly stable, reproducible signal with broad dynamic range.
The heterogeneous format includes washing steps to separate bound and free labels, potentially introducing a source of variability, though this is minimized through automated processing [29]. The technology demonstrates superior precision compared to traditional ELISA methods, with imprecision consistently <5% across the measuring range [7].
Emerging Methodological Developments
Recent technological advances have focused on homogeneous immunoassay formats that eliminate washing steps. The light-initiated chemiluminescence assay (LICA) represents one such development, demonstrating high correlation with the Elecsys Cobas method (R² = 0.9569) while potentially simplifying instrumentation requirements [29].
Additionally, novel high-specificity assays targeting distinct molecular isoforms of AMH have emerged, utilizing antibodies directed against linear epitopes on specific regions of the AMH molecule (proAMH, AMHN,C, AMHN, and AMHC) [18]. These assays may provide enhanced biological insights, particularly in specialized clinical populations such as women with low ovarian reserve.
Figure 1: The Elecsys Cobas AMH assay utilizes Electrochemiluminescence (ECL) technology, providing fully automated, high-precision measurement with minimal manual intervention.
Clinical and Research Applications
Ovarian Reserve Assessment
Both the Elecsys Cobas AMH and AMH Gen II ELISA demonstrate strong correlation with antral follicle count (AFC), a sonographic marker of ovarian reserve [5]. In comparative studies, Spearman correlation coefficients with AFC were 0.83 for the Elecsys method and 0.86 for the ELISA method at baseline assessment, confirming the clinical utility of both assays for ovarian reserve evaluation [5].
The enhanced precision of the Elecsys method, particularly at low AMH concentrations, provides clinical advantages in patient populations with diminished ovarian reserve, where accurate quantification in the lower measuring range informs therapeutic decisions and counseling [18].
Specialized Clinical Populations
In patients with Turner syndrome, who frequently experience primary ovarian insufficiency, AMH measurement has emerged as a valuable biomarker for predicting spontaneous puberty and residual ovarian function [28]. The superior sensitivity of the Elecsys assay (LoQ 0.5 pmol/L versus 3.0 pmol/L for ELISA) enables more reliable detection of the very low AMH concentrations typically encountered in this population [5] [28].
For women with low ovarian reserve undergoing fertility treatments, high-specificity AMH assays that target distinct molecular isoforms may enhance prediction of oocyte yield following ovarian stimulation [18]. Research indicates that assays combining AFC with isoform-specific AMH measurements (particularly the AL-196 assay) offer the best predictive value for cumulus-oocyte complexes and mature oocytes (Adjusted R² = 0.474-0.485, p<0.001) [18].
Research Reagent Solutions
Table 3: Essential Research Materials for AMH Assay Comparison Studies
| Reagent/Material | Function/Application | Specification Considerations |
|---|---|---|
| Elecsys Cobas AMH Reagents | Fully automated AMH measurement | 48 reagent positions on cobas e 801; 112-day onboard stability |
| AMH Gen II ELISA Kit | Manual AMH measurement | Requires duplicate analysis; CV acceptance <11-14% |
| Quality Control Materials | Monitoring assay performance | Multiple concentration levels (high, medium, low) |
| Calibrators | Standard curve generation | Traceable to international standards |
| Sample Collection Tubes | Blood specimen collection | Serum or lithium heparin plasma |
| Low-Binding Storage Tubes | Sample preservation at -80°C | Prevents analyte adsorption |
| Automated Immunoassay Analyzer | High-throughput testing | Cobas e 801 (300 tests/hour) or e 411 (86 tests/hour) |
| ELISA Processing Equipment | Manual assay procedure | Incubators, plate washers, readers |
The Elecsys Cobas AMH assay represents a significant advancement in AMH testing methodology, offering enhanced precision, broader measuring range, and fully automated operation compared to the manual AMH Gen II ELISA. The demonstrated analytical superiority of the Elecsys method, with optimal performance throughout the measuring range and substantially improved precision at low AMH concentrations, provides laboratories and clinicians with a more reliable tool for ovarian reserve assessment.
While method-related differences in absolute AMH values preclude direct interchangeability of results, the strong correlation between methods and the Elecsys assay's compliance with optimal performance standards based on biological variation support its adoption in clinical practice. The operational efficiency of the Elecsys system, with rapid turnaround times and continuous loading capabilities, addresses the growing demand for high-throughput AMH testing in diverse clinical settings.
Ongoing developments in AMH assay technology, including homogeneous immunoassay formats and isoform-specific measurements, promise further refinements in analytical specificity and clinical utility, particularly for specialized patient populations with unique diagnostic challenges.
The accurate measurement of Anti-Müllerian Hormone (AMH) is crucial in reproductive medicine for assessing ovarian reserve. The evolution from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated immunoassays has significantly impacted clinical laboratory workflows. This guide provides a objective comparison between two prominent methods: the established AMH Gen II ELISA and the automated Elecsys Cobas AMH assay, focusing on critical operational parameters including run time, sample volume, and onboard stability. Within the broader context of AMH assay research, understanding these practical differences is essential for researchers, scientists, and drug development professionals to optimize laboratory efficiency and data reliability.
Side-by-Side Comparison of Key Assay Parameters
The following table summarizes the core technical specifications of the AMH Gen II ELISA and the Elecsys Cobas AMH assays, highlighting fundamental differences in their operational workflows and performance characteristics [32] [5] [7].
Table 1: Comparative Assay Parameters: AMH Gen II ELISA vs. Elecsys Cobas AMH
| Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Methodology | Manual ELISA [5] [16] | Fully Automated Immunoassay [32] [7] |
| Assay Time | ~2.5 hours [33] | 18 minutes [7] |
| Sample Volume | 20-25 µL [17] [33] | As low as 30 µL [7] |
| Onboard Stability | N/A (manual kit) | Up to 112 days [7] |
| Measuring Range | 0.16–22.5 ng/mL [17] | 0.03–23 ng/mL [7] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L (≈0.21 ng/mL) [32] [5] | 0.5 pmol/L (≈0.035 ng/mL) [32] [5] |
| Precision (CV%) | 5.5% to 10.3% [32] [5] | <5% (2.8% to 3.6%) [32] [5] [7] |
Detailed Experimental Protocols and Performance Evaluation
Methodology for a Comparative Clinical Series
A prospective observational study directly compared these two assays in a clinical setting, providing key experimental data on their performance [32] [5] [16].
- Study Population: The study included 23 women with a median age of 36 years who were seeking laparoscopic sterilization.
- Sample Collection: Blood samples were collected preoperatively, as well as 1 week and 1, 3, and 6 months postoperatively. Serum was isolated within 4 hours and stored at -80°C until analysis.
- Assay Procedures: All biochemical measurements were performed by skilled technicians at an ISO 15189 accredited laboratory.
- AMH Gen II ELISA: A standard manual technique was used. Samples were analyzed in duplicate, with the analysis repeated if results differed by more than 15%. The limit of quantitation (LOQ) was 3.0 pmol/L [5] [16].
- Elecsys Cobas AMH: Analysis was performed on a Cobas 6000 e601 platform using Roche's standard protocol. The LOQ for this method was 0.5 pmol/L [5] [16].
- Statistical Analysis: Spearman’s correlation test was used to determine the correlation between AMH levels and antral follicle count (AFC). Passing-Bablok and bias plots were generated to compare the two methods.
Key Findings from the Comparative Study
The experimental data revealed significant differences in assay performance:
- Correlation and Bias: While a good correlation was found between the two methods, a consistent bias of approximately 32% was observed, with the Elecsys Cobas assay yielding lower values than the AMH Gen II ELISA [32] [5] [16].
- Analytical Precision: The Elecsys Cobas system demonstrated superior precision, with an analytical variability of 2.8–3.3%, compared to 5.5–10.3% for the AMH Gen II ELISA [32] [5].
- Performance at Low Concentrations: The Elecsys Cobas assay achieved optimal performance goals throughout its measuring range. In contrast, the AMH Gen II ELISA met these goals only at the high end of its range. Furthermore, only 2% of measurements fell below the LOQ for the Elecsys assay, compared to 15% for the ELISA [32] [5] [16].
Technical Specifications and Broader Assay Context
Core Assay Characteristics
- AMH Gen II ELISA (Beckman Coulter): This manual assay integrates previous antibody technologies. It requires a pre-mixture of samples, calibrators, and controls in an assay buffer, contributing to its longer hands-on time and higher potential for human error [17] [5].
- Elecsys Cobas AMH (Roche Diagnostics): As the first fully automated AMH assay cleared by the FDA, it is designed for use on Roche's immunoassay platforms. Its key features include minimal manual intervention, high precision, and robust performance across a wide measuring range [7].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagent Solutions for AMH Immunoassays
| Item | Function in the Assay |
|---|---|
| Calibrators | Solutions of known AMH concentration used to generate a standard curve for quantifying AMH in unknown samples. The Elecsys assay uses recombinant human AMH calibrators [7], while the Gen II uses bovine serum AMH calibrators [17]. |
| Coated Microtiter Plates/Magnetic Beads | Solid phase coated with a capture antibody specific to AMH, which binds the antigen from the sample. |
| Detection Antibody | A second antibody, labeled with a reporter molecule (e.g., biotin, ruthenium complex), which binds to the captured AMH to form a "sandwich" complex [17]. |
| Signal Reagent/Substrate | A chemical that reacts with the reporter molecule on the detection antibody to produce a measurable signal (e.g., colorimetric, chemiluminescent) proportional to the AMH concentration [17] [7]. |
| Assay Buffer | A solution used to dilute samples and reagents, which helps to minimize non-specific binding and maintain a consistent reaction environment [17]. |
| Quality Control (QC) Sera | Samples with predetermined AMH levels that are run alongside patient samples to monitor the accuracy and precision of the assay over time [5]. |
Experimental Workflow Visualization
The diagram below illustrates the general workflow for a comparative assay evaluation study, as described in the cited research.
Comparative Analysis of Broader Assay Performance
Detection Limits and Clinical Utility
A critical differentiator between these assays is their performance at low AMH concentrations, which is vital for assessing diminished ovarian reserve or monitoring menopausal transition. The Elecsys Cobas assay has a significantly lower limit of quantitation (0.5 pmol/L) compared to the AMH Gen II ELISA (3.0 pmol/L) [32] [5]. This enhanced sensitivity is corroborated by independent studies comparing multiple platforms, which found that the picoAMH assay (an ultrasensitive manual assay) maximized detection at very low levels, particularly in contrast to the Gen II kit [17]. This suggests that the automated Elecsys assay bridges the gap between older ELISAs and newer, highly sensitive manual tests.
Correlation with Ovarian Follicle Count
Both assays show strong and significant correlation with antral follicle count (AFC), a gold-standard marker for ovarian reserve. In the comparative study, Spearman correlation coefficients with AFC were 0.83 for Cobas AMH and 0.86 for ELISA AMH at baseline, confirming the clinical validity of both methods [5] [16]. Furthermore, a large multicentre study established that an Elecsys AMH cut-off value of 1.77 ng/mL could identify women with an AFC >15 with high sensitivity (88.34%) and specificity (68.29%) [12].
This comparative analysis demonstrates a clear trade-off between manual and automated AMH immunoassays. The AMH Gen II ELISA represents a established manual methodology but is characterized by a longer assay time, higher analytical variability, and poorer performance in the low concentration range. In contrast, the Elecsys Cobas AMH assay offers the operational benefits of full automation, including a rapid 18-minute turnaround, extended onboard reagent stability, and superior precision. Its lower limit of quantitation makes it more reliable for detecting diminished ovarian reserve. For researchers and clinicians, the choice of assay must balance historical data compatibility with the need for workflow efficiency and robust performance across all clinical ranges, particularly where precise low-level quantification is critical.
The accurate measurement of Anti-Müllerian Hormone (AMH) is fundamental to assessing ovarian reserve in reproductive medicine and endocrinology. The analytical performance of an immunoassay, particularly its limit of quantitation (LoQ) and measuring interval, directly determines its clinical utility in detecting both diminished and elevated AMH concentrations. This guide provides a systematic comparison of the analytical ranges of two principal AMH assays: the established manual AMH Gen II ELISA (Enzyme-Linked Immunosorbent Assay) and the fully automated Elecsys Cobas AMH electrochemiluminescence immunoassay. Understanding these analytical parameters is essential for researchers and clinicians in selecting appropriate methodologies for specific clinical or research applications, particularly in fertility assessment and polycystic ovary syndrome (PCOS) diagnosis.
Analytical Performance Comparison
Direct comparative studies reveal significant differences in the analytical performance between the AMH Gen II ELISA and the Elecsys Cobas AMH assays. These differences impact their utility across various clinical scenarios.
Key Metrics and Experimental Findings
A prospective observational study comparing these two methods found that while they demonstrate good correlation, the Elecsys Cobas assay exhibits superior analytical precision, with a notably lower limit of quantitation [5]. The critical analytical parameters are summarized in the table below.
Table 1: Direct Comparison of Key Analytical Range Parameters
| Analytical Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Limit of Quantitation (LoQ) | 3.0 pmol/L (≈0.42 ng/mL) [5] | 0.5 pmol/L (≈0.07 ng/mL) [5] |
| Upper Limit of Measuring Interval | 70 pmol/L (≈9.8 ng/mL) without dilution [5] | 160 pmol/L (≈22.4 ng/mL) without dilution [5] |
| Analytical Variability (CV%) | 5.5% to 10.3% [5] | 2.8% to 3.3% [5] |
| Methodology | Manual ELISA [5] | Fully Automated ECLIA [5] |
Note on Unit Conversion: The conversion between pmol/L and ng/mL is based on the factor 1 ng/mL = 7.14 pmol/L [2].
The data demonstrates a clear advantage for the Elecsys Cobas assay in sensitivity, with a LoQ six times lower than that of the Gen II ELISA. This allows for more reliable quantification in patients with very low ovarian reserve. Furthermore, the wider measuring interval and superior precision of the automated assay reduce the need for sample dilution and re-testing.
Method-Specific Performance Characteristics
Independent evaluations of each assay have corroborated these comparative findings. The AMH Gen II ELISA, as a manual microtiter plate format, is characterized by its larger analytical variation [10]. In contrast, multicenter evaluations of the Elecsys AMH assay confirm its excellent precision, with a coefficient of variation (CV) of 1.8% for repeatability and 4.4% for intermediate precision, affirming its robustness under routine conditions [10]. Other automated assays, such as the Access AMH assay, have also demonstrated strong performance with a LoQ of 0.010 ng/mL (≈0.07 pmol/L) and total imprecision ranging from 2.4% to 5.2% [6].
Experimental Protocols for Performance Evaluation
The determination of an assay's analytical range follows standardized clinical and laboratory guidelines. The following protocols detail the key experiments for establishing the Limit of Quantitation and Measuring Intervals.
Protocol for Determining Limit of Quantitation (LoQ)
The LoQ is the lowest analyte concentration that can be quantitatively determined with acceptable precision (defined as a CV% <15%) [5] [34].
Procedure:
- Sample Preparation: Obtain a series of patient serum samples with low AMH concentrations. Alternatively, prepare a dilution series from a calibrator or a high-concentration patient sample using a appropriate matrix (e.g., assay-specific diluent or low-AMH serum).
- Testing Schedule: Analyze the samples in multiple replicates (e.g., 2-3 replicates) across several batches and over a minimum of 5 days to capture inter-assay variation [34].
- Data Analysis:
- Calculate the mean concentration and CV% for each sample level.
- The LoQ is identified as the lowest concentration level at which the CV% is consistently below the 15% acceptability threshold [5].
Protocol for Verifying Measuring Interval (Linearity)
Assay linearity verifies that results across the reported range are proportional to the true concentration of the analyte in the sample [35].
Procedure:
- Sample Preparation: Create a series of 5-11 sample mixtures using two patient serum pools: one with a high AMH concentration and one with a very low or undetectable concentration. Mix them in varying proportions (e.g., 100:0, 80:20, 60:40, ..., 0:100) [34].
- Sample Analysis: Measure each mixture in duplicate according to the manufacturer's instructions.
- Statistical Analysis:
- Plot the measured AMH values against the expected values (based on the dilution factor).
- Perform linear regression analysis.
- The assay is considered linear across the tested range if the percentage bias for all levels is within pre-defined limits (e.g., ±10%) of the values predicted by the regression model [35].
The following diagram illustrates the logical workflow and decision points in the analytical validation process for a clinical immunoassay.
Diagram 1: Experimental workflow for defining the analytical range of a clinical immunoassay, based on CLSI guidelines.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful execution of the aforementioned protocols requires specific reagents and materials. The following table outlines key solutions used in evaluating AMH immunoassays.
Table 2: Key Research Reagent Solutions for AMH Assay Evaluation
| Reagent/Material | Function in Analysis | Application Example |
|---|---|---|
| Calibrators | Substances with known AMH concentrations used to establish a calibration curve for converting instrument signals into concentration values. | Recombinant human AMH calibrators (e.g., S5 AMH calibrator from Beckman Coulter) are used in serial dilution for linearity testing [2]. |
| Quality Control (QC) Materials | Stable materials with predefined AMH levels (low, medium, high) used to monitor the precision and stability of the assay over time. | QC materials are analyzed repeatedly across different days to determine intra-assay and inter-assay CV% as part of precision evaluation [2]. |
| Sample Diluent | A solution used to dilute high-concentration patient samples that exceed the upper limit of the measuring interval, ensuring readings fall within the linear range. | Sample Diluent A (Beckman Coulter) or other manufacturer-specific diluents are used for pre-diluting samples before re-analysis [2]. |
| Zero Standard / Assay Buffer | A matrix containing no AMH, used for determining the Limit of Blank (LoB) and for preparing dilution series for linearity studies. | A zero-concentration calibrator or a pool of human serum with undetectable AMH is used to determine the LoB and LOD [34] [35]. |
The defining parameters of an assay's analytical range—the Limit of Quantitation and the measuring interval—are critical for its clinical application. The evidence demonstrates a clear performance differential between the AMH Gen II ELISA and the Elecsys Cobas AMH assays. The Elecsys Cobas assay provides a superior analytical range, with a six-fold lower LoQ (0.5 pmol/L vs. 3.0 pmol/L) and a wider measuring interval, all while achieving excellent precision through full automation. This enhanced sensitivity allows for more accurate quantification in patients with diminished ovarian reserve. For research and clinical laboratories, transitioning to fully automated, high-precision platforms like the Elecsys Cobas represents a significant advancement in the reliable and standardized measurement of AMH.
Establishing Age and Population-Specific Reference Intervals for Clinical Interpretation
The clinical measurement of Anti-Müllerian Hormone (AMH) has become integral to ovarian reserve assessment, yet the transition from manual to automated immunoassays presents significant challenges for establishing reliable reference intervals. The AMH Gen II ELISA (Beckman Coulter) has served as the clinical standard for years, but fully automated assays like the Elecsys Cobas AMH (Roche Diagnostics) offer improved analytical performance [5]. This creates an imperative for the establishment of age and population-specific reference intervals that account for both methodological differences and biological variability. Research demonstrates that the reference intervals established for one assay cannot be directly applied to another due to systematic biases and differences in antibody epitope recognition [36] [9]. Furthermore, ethnic variations in AMH levels necessitate population-specific reference intervals, as demonstrated by studies comparing Korean and Caucasian populations [36] [37]. This comparison guide objectively evaluates the performance characteristics of these two predominant AMH assays within the critical context of establishing clinically relevant reference standards.
Comparative Analytical Performance
Key Performance Metrics
Extensive method comparison studies reveal fundamental differences in the analytical performance of the AMH Gen II ELISA and Elecsys Cobas assays, which directly impact their utility in establishing reference intervals.
Table 1: Direct Comparison of AMH Assay Performance Characteristics
| Performance Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Analytical Variability (CV%) | 5.5-10.3% (high to low range) [5] | 2.8-3.3% (across measuring range) [5] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L [5] | 0.5 pmol/L [5] |
| Measuring Range | 3-70 pmol/L (without dilution) [5] | 0.5-160 pmol/L (without dilution) [5] |
| Bias Between Methods | Reference method | Approximately 32% lower values vs. ELISA [5] |
| Assay Format | Manual [5] | Fully automated [5] |
| Optimal Performance Achievement | Only in high end of measuring range [5] | Throughout measuring range [5] |
The Elecsys Cobas system demonstrates superior analytical precision across the entire measuring range, with consistent CV% of 2.8-3.3% compared to the Gen II ELISA's 5.5-10.3%, which shows particularly high variability at lower concentrations [5]. This enhanced precision is critical for establishing reliable reference intervals, especially in the lower ranges relevant to older reproductive ages or diminished ovarian reserve.
The approximately 32% systematic bias between methods underscores the necessity for method-specific reference intervals [5]. This bias demonstrates that values are not interchangeable between platforms, and clinical decision points established for one assay cannot be directly applied to the other.
Biological Variation and Total Assay Performance
The total variation in AMH measurement incorporates both biological and analytical components: CVTOTAL = √[(CVWithin-person Biological Variation)² + (CVAnalytical)²] [5]. The within-person biological variability for AMH ranges from approximately 21-32% [5]. When applying optimal performance goals for biochemical assays (CVAnalytical < 0.25*CVWithin Biological Variation), the Elecsys Cobas method achieves optimal performance throughout the measuring range, while the AMH Gen II only achieves optimal performance at the high end [5].
Experimental Protocols for Method Comparison
Prospective Method Comparison Study Design
A standardized protocol for comparing AMH assays facilitates the generation of reliable data for establishing reference intervals.
Population Selection: A prospective observational study should include healthy women across the reproductive lifespan (typically 20-50 years) with regular menstrual cycles. Exclusion criteria should encompass endocrine diseases, polycystic ovarian syndrome (PCOS), hormonal contraception use, recent pregnancy (<3 months), and suspected malignancy [5]. Sample size calculation should ensure sufficient power across age strata.
Sample Collection and Handling: Blood samples should be collected in serum separation tubes, allowed to clot, and centrifuged within 4 hours of sampling [5]. Serum should be aliquoted and stored at -80°C until analysis to maintain analyte stability [5]. For comprehensive assessment, samples may be collected at multiple time points (e.g., pre-operative, 1 week, 1, 3, and 6 months post-operative) to evaluate biological variation [5].
Laboratory Analysis: All biochemical measurements should be performed in an ISO 15189 accredited laboratory by skilled technicians [5]. For the Gen II ELISA, samples should be analyzed in duplicate following the standard application protocol employing a pre-mixture of clinical samples, calibrators, and controls in assay buffer [5]. The Elecsys Cobas AMH should be analyzed on a Cobas 6000 e601 platform using Roche's standard protocol [5].
Statistical Analysis: Method comparison should utilize Passing-Bablok regression and Bland-Altman plots to assess bias and agreement [5]. Spearman's correlation test should determine the relationship between AMH values and antral follicle count (AFC) [5]. Total variance should be calculated at different concentration levels (<10 pmol/L and >10 pmol/L) to evaluate performance across the measuring range [5].
Reference Interval Establishment Protocol
Establishing age-specific reference intervals requires careful study design and statistical approaches.
Population-Based Sampling: The Korea National Health and Nutrition Examination Survey (KNHANES) model demonstrates the advantage of using nationwide population-based studies to minimize selection bias [36] [37]. Participants should be selected from the general population with careful application of health criteria to identify reference individuals.
Age Stratification: Given that AMH decreases with age, reference intervals should be determined according to narrow age bins [36] [37]. Five-year age groups with sufficient sample size (n>120 per group) provide adequate resolution across reproductive ages [36].
Statistical Analysis: Non-parametric methods should be used to calculate percentiles (e.g., 10th, 50th, 90th) for each age stratum [36]. Polynomial curves can be fitted to the percentile values across age groups to create continuous reference limits [36].
Clinical and Research Applications
Prediction of Ovarian Response
The Elecsys AMH assay demonstrates strong predictive value for ovarian hyper-response in controlled ovarian stimulation. A prespecified AMH cut-off of 15 pmol/L (2.10 ng/mL) predicted hyper-response with a sensitivity of 81.3% and specificity of 64.7% in women undergoing a gonadotrophin-releasing hormone antagonist protocol [38]. The area under the receiver operating characteristic curve was 82.1%, indicating good predictive accuracy [38].
Correlation with Antral Follicle Count
Both assays show strong correlation with antral follicle count (AFC), a sonographic marker of ovarian reserve. At baseline, Cobas AMH versus AFC demonstrated a Spearman correlation coefficient of 0.83, while ELISA AMH versus AFC showed a correlation of 0.86 [5]. This confirms that both assays effectively reflect the functional ovarian follicular pool.
Interchangeability with Existing Reference Ranges
Studies investigating the Access AMH assay (another automated assay from Beckman Coulter) have demonstrated high correlation with the Gen II ELISA method (R² = 0.9822-0.9832), suggesting that reference ranges established using the Gen II ELISA may be interchangeable with certain automated assays [2]. However, this level of correlation is not universal across all automated platforms, emphasizing the need for verification studies.
Table 2: Clinical Performance Characteristics of AMH Assays
| Clinical Application | AMH Gen II ELISA Performance | Elecsys Cobas AMH Performance |
|---|---|---|
| Prediction of Hyper-response | Literature-established cut-offs available | Cut-off 15 pmol/L: 81.3% sensitivity, 64.7% specificity [38] |
| Correlation with AFC | 0.86 Spearman correlation [5] | 0.83 Spearman correlation [5] |
| Cycle Variability | Substantial longitudinal variation reported [39] | Lower analytical variability may better reflect true biological variation [5] |
| PCOS Application | Correlates with follicle count in PCOS patients [9] | Strong interassay correlation in high AMH ranges [9] |
Ethnic Considerations in Reference Intervals
Population-specific studies have revealed important ethnic variations in AMH levels that necessitate tailored reference intervals. Research comparing Korean and Caucasian populations found that the median serum AMH level in the 25-34-year-old Korean population tended to be slightly higher than in the Caucasian population [36] [37]. This highlights the limitation of applying reference intervals derived from one ethnic group to another without validation.
The reference interval established for the Elecsys assay in the Korean population was wider than that mentioned in the manufacturer's package insert, exceeding the limits at both ends of the range [37]. This demonstrates that manufacturer-provided reference intervals may not be optimal for all populations and underscores the importance of establishing locally relevant reference standards.
Research Reagent Solutions
The following table details essential materials and their functions for conducting method comparison studies and establishing reference intervals for AMH assays.
Table 3: Essential Research Reagents and Materials for AMH Method Comparison Studies
| Reagent/Material | Function/Application | Specification Considerations |
|---|---|---|
| AMH Calibrators | Establish standard curve for quantification | Source (recombinant vs. native), value assignment, commutability with patient samples [40] |
| Quality Control Materials | Monitor assay precision and accuracy | Multiple concentration levels (low, medium, high), human serum-based matrix [5] |
| Sample Diluent | Dilution of high-concentration samples | Minimize matrix effects, maintain antibody-epitope binding [2] |
| Antibody Pairs | Capture and detection of AMH isoforms | Epitope specificity (pro vs. mature region recognition) [9] [40] |
| WHO Reference Reagent 16/190 | Harmonization across methods | Assigned value: 489 ng/ampoule; limited commutability [40] |
| Serum Separation Tubes | Sample collection and processing | Gel barrier for efficient serum separation, minimal analyte adsorption [5] |
Molecular Basis of Assay Differences
The fundamental differences between AMH assays stem from the complex molecular biology of AMH isoforms and variable antibody recognition.
AMH is synthesized as a precursor hormone that undergoes proteolytic cleavage to form multiple isoforms with differential biological activity [40]. The most bioactive form is the non-covalent AMHN,C complex, consisting of the pro-region (AMHN) and mature region (AMHC) [40]. Additional cleavage at amino acid 229 generates further fragments including AMHN,229 and AMHM [39].
The AMH Gen II ELISA and Elecsys Cobas assays use identical antibody pairs targeting the AMHN,229 region and the C-terminal region (AMHC) [9]. This consistent epitope recognition contributes to their strong correlation despite differences in assay platform and precision. In contrast, the picoAMH assay (Ansh Labs) uses a different antibody configuration with capture antibody recognizing the C-terminal region and detector antibody recognizing the AMHM region [9].
The variable detection of these different AMH isoforms by different assays contributes to the observed intermethod differences and underscores why reference intervals cannot be directly transferred between methods without proper validation studies.
The establishment of age and population-specific reference intervals for AMH requires careful consideration of assay methodology and population characteristics. The Elecsys Cobas AMH assay demonstrates superior analytical performance with lower variability and wider measuring range compared to the AMH Gen II ELISA, supporting its use for establishing precise reference intervals [5]. The consistent 32% bias between methods necessitates method-specific reference intervals, as values are not directly interchangeable [5]. Furthermore, ethnic variations in AMH levels between Korean and Caucasian populations highlight the importance of population-specific reference standards rather than reliance on manufacturer-provided intervals [36] [37]. Future standardization efforts using the WHO Reference Reagent 16/190 may improve harmonization between methods, though current commutability limitations require continued method-specific validation [40]. For clinical laboratories transitioning between AMH methods, parallel testing and establishment of new reference intervals is essential for accurate patient classification and clinical decision-making.
Addressing Analytical Variability and Performance Goals
In the field of reproductive endocrinology and clinical biochemistry, the precision and reliability of biomarker assays are paramount. Anti-Müllerian Hormone (AMH) has emerged as a crucial biochemical marker for assessing ovarian reserve, making the precision of its measurement a subject of significant clinical importance [5]. This guide provides a detailed, data-driven comparison of the precision of two prominent AMH detection methods: the AMH Gen II ELISA (Enzyme-Linked Immunosorbent Assay) and the Elecsys Cobas AMH automated immunoassay. The Coefficient of Variation (CV%) serves as the key metric for evaluating assay performance, encompassing both intra-assay (within-run) and inter-assay (between-run) precision. Understanding these variances is critical for researchers and clinicians who rely on AMH values for predicting ovarian response, diagnosing conditions like Polycystic Ovary Syndrome (PCOS), and providing fertility counseling [5] [41].
Comparative Assay Performance Data
The table below synthesizes key performance characteristics of the AMH Gen II ELISA and the Elecsys Cobas AMH assays, highlighting critical differences in precision, measuring range, and operational workflow.
Table 1: Performance Comparison of AMH Gen II ELISA and Elecsys Cobas AMH Assays
| Performance Characteristic | AMH Gen II ELISA (Beckman Coulter) | Elecsys Cobas AMH (Roche) |
|---|---|---|
| Intra-Assay CV% | 5.2% - 10.3% [5] | 2.8% - 3.3% [5] |
| Inter-Assay CV% | < 12% (typical for ELISA) [42] [43] | Information not explicitly in search results |
| Analytical Variability | 5.5% - 10.3% [5] | 2.8% - 3.3% [5] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L [5] | 0.5 pmol/L [5] |
| Measuring Range | 3 - 70 pmol/L [5] | 0.5 - 160 pmol/L [5] |
| Assay Principle | Manual ELISA [5] | Automated Immunoassay [5] |
| Bias vs. Other Methods | Reference Method | Approximately 32% lower results vs. Gen II ELISA [5] [44] |
Detailed Analysis of Precision Data
The intra-assay precision, which measures repeatability within a single run, is demonstrably superior for the Elecsys Cobas system. Its CV% remains tightly constrained between 2.8% and 3.3% across different control levels. In contrast, the AMH Gen II ELISA shows a wider and higher CV% range of 5.2% to 10.3%, with the highest imprecision observed at lower concentrations [5]. This trend is confirmed by an independent study which reported an intra-assay CV of ≤ 2.8% for the Elecsys assay and ≤ 5.8% for the AMH Gen II ELISA [44].
For inter-assay precision, which gauges variation between different runs over time, commercial ELISA kits typically specify CV% values of < 10-12% [42] [43]. While specific inter-assay CV% for the Elecsys system was not detailed in the provided results, its low intra-assay CV and fully automated nature are designed to minimize between-run variation.
A key finding from a 2015 prospective study is the consistent bias between the two methods. The Elecsys Cobas assay yields AMH concentrations that are, on average, 32% lower than those obtained from the AMH Gen II ELISA [5]. This systematic difference underscores the critical importance of using assay-specific reference ranges in clinical practice and research, as values are not directly interchangeable.
Experimental Protocols for Precision Validation
Key Experimental Workflow
The following workflow diagram outlines the general procedure for determining the intra-assay and inter-assay CV% for an AMH assay, as derived from the methodologies in the cited studies.
Detailed Methodologies from Cited Studies
The data presented in this guide are derived from rigorously conducted studies. The following details their experimental approaches:
Prospective Observational Study (2015): This study compared the AMH Gen II ELISA and Elecsys Cobas methods using 112 serum samples from 23 women. Samples were analyzed at the Department of Clinical Biochemistry, Aarhus University Hospital, an ISO 15189 accredited laboratory.
- AMH Gen II ELISA Protocol: A standard manual technique was used. Samples were analyzed in duplicate, and the mean value was used. If duplicates differed by more than 15%, the analysis was repeated. The limit of acceptance for internal controls was CV < 11–14% [5].
- Elecsys Cobas Protocol: Analysis was performed on a Cobas 6000 e601 platform using Roche's standard automated protocol. The limit of acceptance for internal controls was a tighter CV < 5% [5].
- Precision Calculation: CV% was calculated as (Standard Deviation / Mean) × 100. The analytical goals for optimal performance were defined as CVAnalytical < 0.25 × CVWithin Biological Variation [5].
Independent Assay Evaluation (2015): This prospective study compared two new automated assays (Elecsys, Access) with two ELISA assays (Gen II, Ansh). It involved 83 patients referred for AMH measurement.
- Precision Measurement: Intra-assay CV% was determined for each method, reinforcing the finding that the Elecsys method had the lowest imprecision (≤ 2.8%) [44].
The Scientist's Toolkit: Key Research Reagent Solutions
The following table lists essential materials and reagents required for performing AMH ELISA, which is relevant for understanding the manual method compared in this guide.
Table 2: Essential Research Reagents for AMH ELISA
| Item | Function/Description | Reference |
|---|---|---|
| Pre-coated Microplate | 96-well plate pre-coated with a capture antibody specific to AMH. Serves as the solid phase for the immunoassay. | [42] [43] |
| Recombinant AMH Standard | A calibrated preparation of AMH used to generate a standard curve for quantifying AMH concentration in unknown samples. | [42] [43] |
| Biotin-Conjugated Detection Antibody | An antibody that binds to a different epitope of AMH, conjugated to biotin. Forms the "sandwich" in a sandwich ELISA. | [43] |
| Streptavidin-HRP (Horseradish Peroxidase) | An enzyme conjugate that binds to biotin. Catalyzes the conversion of the substrate into a colored product. | [45] |
| TMB (Tetramethylbenzidine) Substrate | A chromogenic substrate for HRP. Produces a blue color when oxidized, which is measured spectrophotometrically. | [42] [43] |
| Stop Solution | An acidic solution (e.g., sulfuric acid) that terminates the enzyme-substrate reaction, changing the color from blue to yellow and stabilizing the signal. | [42] [45] |
| Wash Buffer (Concentrate) | A buffered solution, often containing a detergent, used to remove unbound reagents and reduce background signal between incubation steps. | [42] [43] |
The quantitative data on intra-assay and inter-assay CV% provides a clear and objective basis for comparing the precision of AMH assays. The Elecsys Cobas AMH automated immunoassay demonstrates superior analytical precision with significantly lower intra-assay CV% (2.8-3.3%) compared to the manual AMH Gen II ELISA (5.2-10.3%) [5] [44]. This enhanced precision, coupled with a wider measuring range and a lower limit of quantitation, allows the Elecsys assay to achieve optimal performance goals across the entire clinical range [5]. Consequently, for research and clinical applications where high reproducibility and accuracy at low concentrations are critical—such as monitoring ovarian reserve in women with low AMH or making precise diagnostic classifications—the automated Elecsys system holds a distinct advantage. However, researchers must remain acutely aware of the consistent bias between methods and ensure that all interpretations are based on assay-specific reference intervals.
In the field of clinical reproductive medicine, the accurate measurement of Anti-Müllerian Hormone (AMH) is crucial for assessing ovarian reserve, predicting response to controlled ovarian stimulation, and diagnosing conditions such as polycystic ovarian syndrome (PCOS). The evolution of AMH immunoassays has progressed from manual Enzyme-Linked Immunosorbent Assay (ELISA) platforms to fully automated immunoassay systems, each with distinct methodological characteristics and potential sources of analytical error. This comparison guide objectively evaluates the performance of the manual AMH Gen II ELISA (Beckman Coulter) and the automated Elecsys Cobas AMH assay (Roche Diagnostics) within the context of a broader thesis on AMH measurement platforms. Understanding the technical differences and inherent limitations of each system is essential for researchers, scientists, and drug development professionals to appropriately interpret data, design studies, and advance diagnostic applications in reproductive medicine and women's health.
Analytical Principles and Platform Architectures
2.1 Fundamental Immunoassay Designs AMH immunoassays operate on the principle of sandwich-type immunoassays, where two antibodies bind to different epitopes on the AMH molecule. The manual AMH Gen II ELISA utilizes a manual technique with anti-AMH IgG immobilized to microtiter plate wells, a biotinylated antibody conjugate, and streptavidin-enzyme conjugate with tetramethylbenzidine (TMB) chromogen substrate for detection [46]. In contrast, the Elecsys Cobas AMH immunoassay employs a fully automated system using two mammalian monoclonal antibodies conjugated to biotin and ruthenium, with electrochemiluminescence detection on a Cobas 6000 e601 or e801 analyzer [5] [46]. This fundamental difference in detection chemistry and automation level establishes the foundation for variations in analytical performance and potential error profiles between the platforms.
2.2 Molecular Recognition of AMH Isoforms Current commercial AMH immunoassays, including both the Gen II ELISA and Elecsys Cobas, detect both the precursor (proAMH) and mature bioactive (AMHN,C) forms of circulating AMH [47] [11]. The reported AMH values represent a composite of both molecular forms, whose physiological ratio may vary under different clinical conditions. The antibody pairs used in each assay may recognize different epitopes on these AMH isoforms with varying affinities, potentially contributing to measurement bias between platforms [46]. This lack of standardization in epitope recognition and differential detection of AMH isoforms represents a significant source of inter-assay variation that researchers must consider when comparing data across platforms or establishing clinical cut-off values.
Comparative Analytical Performance
3.1 Precision and Analytical Variability A direct comparison study evaluating 112 serum samples from 23 women demonstrated substantial differences in analytical variability between platforms. The Elecsys Cobas method showed significantly lower analytical variability, with coefficients of variation (CV%) ranging from 2.8% to 3.3% across the measuring range. In contrast, the AMH Gen II ELISA demonstrated higher analytical variability, with CV% ranging from 5.2% at higher concentrations (40 pmol/L) to 10.3% at lower concentrations (7 pmol/L) [5]. This pattern of increased imprecision at lower AMH concentrations for the manual ELISA platform is particularly relevant for applications requiring high sensitivity, such as monitoring ovarian function after chemotherapy or in women with severely diminished ovarian reserve.
Table 1: Analytical Performance Comparison of AMH Immunoassays
| Performance Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Assay Format | Manual two-site immunoassay | Fully automated immunoassay |
| Total Analytical Variability (CV%) | 5.5% - 10.3% | 2.8% - 3.3% |
| Limit of Quantification | 3.0 pmol/L | 0.5 pmol/L |
| Measuring Range | 3-70 pmol/L (without dilution) | 0.5-160 pmol/L (without dilution) |
| Samples Without Quantitation | 15% | 2% |
| Bias Between Methods | Reference method | Approximately 32% lower than ELISA |
3.2 Sensitivity and Measuring Range The Elecsys Cobas AMH assay demonstrates superior sensitivity with a limit of quantitation (LoQ) of 0.5 pmol/L compared to 3.0 pmol/L for the AMH Gen II ELISA [5] [32]. This enhanced sensitivity expands the clinical utility of the automated platform for monitoring patients with very low AMH levels, such as women in the late reproductive years, those approaching menopause, or patients recovering from gonadotoxic treatments. The broader measuring range of the Elecsys assay (0.5-160 pmol/L without dilution) compared to the Gen II ELISA (3-70 pmol/L without dilution) also reduces the need for sample dilution, thereby minimizing a potential source of pre-analytical error and improving workflow efficiency in high-volume laboratory settings [5] [46].
Experimental Protocols and Methodological Considerations
4.1 Sample Collection and Handling Protocol For comparative studies of AMH measurement platforms, proper sample collection and handling are critical. The validated protocol requires collection of venous blood samples with subsequent serum separation within 4 hours of sampling [5]. Samples should be aliquoted and stored at -80°C until analysis to maintain AMH stability. Freeze-thaw cycles should be minimized as they may impact AMH integrity. For the manual ELISA platform, samples requiring dilution should follow manufacturer specifications precisely, as improper dilution has been identified as a source of error in the Gen II assay [47]. Both serum and plasma matrices may be used, though serum is generally preferred for most analytes due to potential interference from anticoagulants in immunoassays [48].
4.2 Experimental Comparison Methodology The referenced comparative study employed a prospective observational design with repeated measurements from the same patient cohort [5] [32]. Blood samples were collected preoperatively and at multiple post-operative time points (1 week, 1, 3, and 6 months) from women undergoing laparoscopic sterilization. Each sample was analyzed using both the AMH Gen II ELISA and Elecsys Cobas AMH methods under controlled conditions. Skilled technicians performed all analyses in an ISO 15189 accredited laboratory environment. The assays were validated according to optimal performance goals for biochemical assays, defined as CVAnalytical < 0.25* CVWithin Biological Variation. Method comparison statistics included Passing-Bablok regression, bias plots, and Spearman correlation analysis to assess the relationship between platforms and against reference standards such as antral follicle count (AFC) [5].
5.1 Technical and Operator-Dependent Errors The manual execution of the AMH Gen II ELISA introduces several technical vulnerabilities that can compromise result accuracy. Pipetting inaccuracies represent a primary source of error, as inconsistent volumes during reagent addition, sample transfer, or serial dilution directly impact optical density readings and subsequent concentration calculations [49]. Cross-contamination between wells due to improper pipetting techniques or splashing can further compromise assay specificity. Additionally, the manual technique requires a pre-mixture of clinical samples, calibrators, and controls in assay buffer, introducing another potential variable [5]. These technical limitations of manual processing not only affect assay sensitivity and specificity but also reduce repeatability and increase waste and associated costs in the research setting.
5.2 Timing and Incubation Variability The manual AMH Gen II ELISA is particularly susceptible to timing inconsistencies throughout the multi-step assay procedure. Variations in incubation start and stop times, reagent addition intervals, and even plate washing processes can introduce significant inter-assay variation [49]. Each procedural step—including coating, blocking, washing, incubation with samples and detection antibodies, and substrate reaction—requires precise durations to ensure optimal antigen-antibody binding and enzymatic reactions. In a manual setting, these timing inconsistencies are practically unavoidable and result in variations in binding kinetics and final signal development. Such temporal imprecision directly impacts the reproducibility of results across different experimenters and laboratory environments, challenging the reliability of data for multi-center trials or longitudinal studies.
5.3 Complement Interference and Standardization Issues A particularly significant analytical issue identified with the AMH Gen II ELISA is interference from complement proteins. Studies have attributed the poor reproducibility of the Gen II assay, particularly following sample dilution and under different storage conditions, to assay interference due to binding of serum complement protein C1q [47] [11]. This interference manifests as inconsistent results upon sample dilution and contributes to the higher analytical variability observed with this platform. Additionally, the AMH Gen II ELISA has demonstrated calibration instability and lack of commutability with proposed international reference materials, further complicating standardization efforts across laboratories and comparison with historical data [46] [47].
Diagram 1: Sources and Impacts of Error in Manual ELISA Platforms
Advantages of Automated Immunoassay Systems
6.1 Enhanced Precision Through Automation The Elecsys Cobas AMH assay demonstrates significantly improved precision through full automation of the analytical process. The system eliminates manual pipetting variability by employing automated liquid handling with precisely calibrated volumes [5] [49]. All process steps are controlled by predefined protocols with strictly prescribed incubation parameters and uniform timing across all samples, effectively removing the temporal inconsistencies inherent in manual techniques [49]. This automation extends to the washing and immunostaining procedures, which are executed with machine precision for every sample. The resultant analytical variability of less than 3.3% throughout the measuring range represents a substantial improvement over the manual ELISA platform and meets optimal performance goals for biochemical assays (CVAnalytical < 0.25* CVWithin Biological Variation) across the entire clinical reporting range [5].
6.2 Reduced Vulnerability to Analytical Interference The automated Elecsys Cobas AMH system demonstrates reduced susceptibility to certain forms of analytical interference that affect the manual ELISA platform. While all immunoassays are potentially vulnerable to interference from heterophile antibodies, biotin, or other endogenous factors, the Elecsys assay appears less affected by complement-mediated interference that has been problematic for the Gen II ELISA [48] [47]. The robust assay design, incorporating two mammalian monoclonal antibodies in an automated sandwich format, contributes to this improved reliability. Additionally, the system's integrated quality monitoring includes real-time tracking of assay performance with automatic flagging of anomalous results, providing an additional layer of quality control not available in manual systems [5] [38].
6.3 Workflow Efficiency and Data Integrity Automated AMH platforms offer significant advantages in workflow efficiency and data management for high-volume laboratory settings. The Elecsys system operates on random-access platforms, allowing continuous processing of samples with minimal manual intervention and reduced turnaround times [46] [47]. Integration with Laboratory Information Management Systems (LIMS) enables real-time data transfer without manual transcription, effectively eliminating transcription errors and ensuring data integrity [49]. This automated data handling supports regulatory compliance and quality assurance protocols essential for both clinical diagnostics and drug development research. The combination of operational efficiency and robust data management makes automated platforms particularly suitable for multi-center trials and large-scale epidemiological studies requiring standardized AMH measurement across different sites.
Research Reagent Solutions and Essential Materials
Table 2: Key Research Reagent Solutions for AMH Immunoassay Studies
| Reagent/Material | Function in AMH Immunoassays | Platform Application |
|---|---|---|
| Anti-AMH Monoclonal Antibodies | Recognition of specific AMH epitopes in sandwich assay format | Both platforms (different antibody pairs) |
| Recombinant Human AMH Calibrators | Establishment of calibration curve for AMH quantification | Both platforms (different source materials) |
| TMB Chromogen Substrate | Enzyme-mediated color development for optical detection | AMH Gen II ELISA |
| Ruthenium/Tripropylamine Conjugate | Electrochemiluminescence signal generation | Elecsys Cobas AMH |
| Streptavidin-Biotin Complex | Signal amplification system | Primarily AMH Gen II ELISA |
| Assay Buffer Systems | Optimization of antigen-antibody binding conditions | Both platforms (proprietary formulations) |
| Complement Blocking Reagents | Mitigation of complement interference in serum samples | Primarily for troubleshooting Gen II ELISA |
Standardization Challenges and Future Directions
8.1 Lack of Harmonization Between Assays A fundamental challenge in AMH measurement across all platforms is the lack of standardized calibration. Manufacturers of AMH assays have historically used proprietary calibrators derived from various sources with variably assigned values, leading to significant variation in standard curves between different immunoassays [46] [47]. This standardization gap results in consistent methodological bias, with the Elecsys Cobas AMH assay demonstrating approximately 32% lower values compared to the AMH Gen II ELISA across the measuring range [5] [32]. The World Health Organization has developed an international standard for AMH immunoassays (recombinant AMH preparation code 16/190); however, commutability between this reference material and clinical serum samples has been demonstrated only in some but not all immunoassay methods, limiting its universal application [47] [11]. This lack of harmonization complicates the establishment of universal clinical cut-off values and requires method-specific reference ranges for appropriate interpretation of results.
8.2 Emerging Technologies and Methodological Advances Future directions in AMH measurement technology focus on addressing current limitations through advanced methodological approaches. The development of second-generation reference preparations with wider commutability represents a priority for international standardization efforts [47]. Mass spectrometry-based methods may offer alternative approaches for AMH quantification with potentially improved specificity, though these techniques currently remain primarily in the research domain [48]. Additionally, novel assay designs incorporating engineered antibodies with specific recognition of particular AMH isoforms could provide more clinically relevant measurements by distinguishing between proAMH and the biologically active AMHN,C forms [46] [11]. Such advances would enhance the biological and clinical specificity of AMH measurement beyond the current composite values provided by existing platforms.
The comprehensive comparison between manual AMH Gen II ELISA and automated Elecsys Cobas AMH platforms reveals distinct advantages for automated systems in analytical performance, reproducibility, and operational efficiency. The Elecsys Cobas assay demonstrates superior precision with lower analytical variability (2.8-3.3% vs. 5.5-10.3%), enhanced sensitivity (LoQ: 0.5 pmol/L vs. 3.0 pmol/L), and reduced vulnerability to complement interference compared to the manual ELISA platform. These technical advantages position automated systems as preferable for both clinical applications and research settings requiring high reliability and throughput. Nevertheless, fundamental challenges in assay standardization persist across all platforms, emphasizing the continued need for method-specific reference ranges and careful interpretation of AMH values in both clinical practice and research contexts. Researchers and drug development professionals should consider these analytical characteristics when selecting platforms for AMH measurement, with automated systems offering clear advantages for studies requiring high precision, sensitivity, and operational efficiency.
Strategies for Managing the Systematic Bias Between Assays
The quantification of Anti-Müllerian Hormone (AMH) has become integral to ovarian reserve assessment in clinical and research settings. However, the transition from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated immunoassays has introduced a significant challenge: systematic bias between different measurement platforms. This bias complicates the interpretation of AMH values across different studies and clinical settings, potentially affecting patient management decisions and research conclusions. The core of this challenge lies in the methodological differences between assays, including variations in antibody specificity, calibrator standards, and analytical sensitivity [17]. Understanding the magnitude and consistency of this bias is essential for researchers and clinicians who rely on AMH measurements for assessing ovarian reserve, predicting ovarian response to controlled stimulation, and making diagnostic decisions.
The clinical implications of assay bias are substantial. As AMH measurements increasingly inform critical decisions in reproductive medicine—such as predicting oocyte yield following controlled ovarian stimulation (COS) for in vitro fertilization (IVF) and assessing ovarian reserve in cancer patients—the need for robust strategies to manage between-assay variability becomes paramount [20] [17]. This guide objectively compares the performance of the AMH Gen II ELISA and Elecsys Cobas AMH assays, providing experimental data and methodologies to manage systematic bias effectively.
Quantitative Comparison of AMH Assay Performance
Direct comparison studies reveal consistent and quantifiable differences between the AMH Gen II ELISA and Elecsys Cobas AMH assays. These differences span multiple performance characteristics, including systematic bias, analytical variability, and functional sensitivity.
Table 1: Direct Performance Comparison Between AMH Gen II ELISA and Elecsys Cobas AMH Assays
| Performance Characteristic | AMH Gen II ELISA (Beckman Coulter) | Elecsys Cobas AMH (Roche) | Comparative Data |
|---|---|---|---|
| Systematic Bias | Reference method | Approximately 32% lower values compared to ELISA | Consistent across measuring range; tendency for increased bias at higher concentrations [5] |
| Analytical Variability (CV%) | 5.2-10.3% (controls)22-32% (clinical samples) | 2.8-3.3% (controls)21.3-28% (clinical samples) | Elecsys demonstrates superior precision, particularly at lower concentrations [5] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L | 0.5 pmol/L | Elecsys provides 6-fold better sensitivity [5] |
| Measuring Range | 3-70 pmol/L (without dilution) | 0.5-160 pmol/L (without dilution) | Elecsys offers wider working range [5] |
| Correlation with Antral Follicle Count (AFC) | Spearman r: 0.81-0.86 (vs. AFC) | Spearman r: 0.83 (vs. AFC) | Comparable clinical correlation [5] |
The observed 32% systematic bias between the Elecsys Cobas AMH assay and the AMH Gen II ELISA establishes that these methods are not numerically interchangeable [5]. This bias demonstrates that results from these two platforms cannot be used interchangeably without appropriate correction or interpretation. The Elecsys assay demonstrates superior analytical precision, with a considerably lower coefficient of variation (2.8-3.3%) compared to the Gen II ELISA (5.2-10.3%) across control samples [5]. This enhanced precision is particularly evident at lower AMH concentrations, where the Gen II ELISA shows increased variability.
Table 2: Comparison of Additional Commercial AMH Assays
| Assay | Manufacturer | Systematic Bias | Key Characteristics |
|---|---|---|---|
| Access AMH | Beckman Coulter | 0.711 slope (vs. Gen II pre-mix) | Fully automated; uses identical antibodies to Gen II assays; good correlation with oocyte yield (R=0.655) [20] |
| picoAMH | Ansh Labs | Higher than Gen II | Maximizes detection at very low levels (97% detectability vs. 84% for Gen II) [17] |
| Ultrasensitive AMH | Ansh Labs | Higher than Gen II | Improved detection capabilities [17] |
Beyond the Elecsys and Gen II comparison, other commercially available AMH assays also demonstrate variable performance characteristics. The Beckman Coulter Access AMH assay, another fully automated platform, shows a strong correlation with the Gen II pre-mix assay but reports slightly lower values (regression slope of 0.711) [20]. The Ansh Labs picoAMH and Ultrasensitive assays demonstrate improved detection capabilities at very low AMH concentrations, which is particularly valuable in clinical scenarios such as assessing ovarian reserve in cancer patients or women with diminished ovarian reserve [17].
Experimental Protocols for Assay Comparison
To effectively evaluate and manage systematic bias between AMH assays, researchers should implement standardized experimental protocols. These methodologies enable rigorous comparison and facilitate the development of correction strategies.
Sample Collection and Handling Protocol
Sample Cohort Selection: Prospective studies should include a minimum of 100 serum samples from well-characterized patient populations covering the entire measuring range of both assays (low, medium, and high AMH concentrations) [5] [17]. Participant inclusion should account for relevant clinical characteristics, including age, menstrual cycle phase, and reproductive status.
Sample Processing Standardization: Collect blood samples in serum tubes and process within 4 hours of collection by centrifugation at 2000-3000 × g for 10 minutes. Aliquot serum into cryovials and store immediately at -80°C until analysis [5]. Avoid multiple freeze-thaw cycles (maximum 2 cycles recommended) to maintain AMH stability.
Batch Analysis Approach: Analyze all samples in the same batch for each assay to minimize within-assay variability. Include quality control samples at low, medium, and high concentrations in each run to monitor assay performance [5] [17].
Statistical Methods for Bias Assessment
Correlation Analysis: Perform Spearman's rank correlation to evaluate the relationship between assay results and biological parameters such as antral follicle count (AFC) [5]. Calculate Pearson's correlation coefficient for assay-to-assay comparisons.
Bias Evaluation: Utilize Passing-Bablok regression analysis to determine systematic and proportional biases between methods [5]. This non-parametric method is particularly robust for comparing methods with different measurement principles.
Precision Assessment: Determine within-run and total imprecision by repeatedly testing control materials and patient samples across multiple runs [5]. Express results as coefficients of variation (CV%) with the optimal performance goal being CVAnalytical < 0.25 × CVWithin Biological Variation [5].
Diagram 1: Experimental workflow for assay comparison illustrating the sequential process from study design to bias quantification.
Visualization of Systematic Bias and Analytical Performance
Understanding the nature and magnitude of systematic bias requires effective visualization of comparative data. These visual tools help researchers comprehend both the constant and proportional components of between-assay differences.
Diagram 2: Factors contributing to systematic bias between AMH assays and their effects on clinical interpretation.
Research Reagent Solutions for AMH Assay Comparison
Implementing robust assay comparison studies requires specific research reagents and materials designed to evaluate analytical performance and identify systematic biases.
Table 3: Essential Research Reagents for AMH Assay Comparison Studies
| Reagent/Material | Function in Assay Comparison | Specification Requirements |
|---|---|---|
| Human Serum Panels | Provides biological matrix for comparison studies | Should span clinical measuring range (low, medium, high AMH); well-characterized [5] |
| Quality Control Materials | Monitors assay precision and reproducibility | Multiple concentration levels; stable long-term performance [5] |
| Calibrator Standards | Establishes assay calibration traceability | Human recombinant vs. bovine serum-based; source impacts bias [17] |
| Antibody Pairs | Determines epitope recognition differences | Pro-region vs. mature region specificity [17] |
| Matrix Buffers | Evaluates dilution linearity and parallelism | Synthetic matrix simulating analyte-free human serum [6] |
The selection of appropriate reagents significantly influences the validity of assay comparison studies. Notably, calibrator standards vary substantially between assays, with some utilizing human recombinant AMH while others employ bovine serum AMH, contributing to systematic differences [17]. Similarly, antibody specificity represents a critical factor, as antibodies targeting different regions of the AMH molecule (pro-region versus mature region) demonstrate varying recognition of circulating AMH isoforms [17]. Researchers should carefully document the characteristics of all reagents employed in comparison studies to enable proper interpretation of results.
Strategic Management of Systematic Bias
Establishing Assay-Specific Reference Intervals
The fundamental strategy for managing systematic bias between AMH assays involves establishing and implementing assay-specific reference intervals rather than attempting to apply universal cut-offs across platforms. Research demonstrates that AMH concentrations vary not only by assay method but also by ethnic population, emphasizing the need for population-specific reference intervals for each assay [8].
For the Elecsys Cobas AMH assay, reference intervals established in healthy Korean women demonstrate a clear age-dependent decline, with the mid-95 percentile intervals decreasing from 7.93-81.21 pmol/L in women aged 19-24 years to 0.07-3.86 pmol/L in women aged 50-54 years [8]. Similarly, clinical decision limits for predicting ovarian response differ between assays, with the Elecsys assay demonstrating optimal cut-offs of 6.4 pmol/L for predicting low response and 14.2 pmol/L for predicting high response in GnRH antagonist cycles [4].
Method Validation and Verification Protocols
Implementing comprehensive method validation procedures is essential for laboratories introducing new AMH assays or comparing existing methods. Key validation parameters should include:
Precision Studies: Evaluate within-run, between-run, and total imprecision using clinical samples across the measuring range [5] [50]. Performance goals should follow established standards, with optimal performance defined as CVAnalytical < 0.25 × CVWithin Biological Variation [5].
Linearity and Recovery Assessments: Demonstrate dilutional linearity by serially diluting high-concentration patient samples and assessing recovery against the standard curve [50] [6]. The Elecsys Cobas AMH assay demonstrates linearity up to at least 24 ng/mL [6].
Sample Stability Testing: Evaluate AMH stability under various storage conditions. Studies indicate that AMH samples remain stable at room temperature for up to 48 hours, at 2-8°C for up to 7 days, and at -20°C for up to 15 months [6].
Clinical Correlation and Outcome Studies
Beyond analytical comparisons, establishing clinical correlation is essential for verifying assay performance. Studies demonstrate that both the Elecsys Cobas AMH and Access AMH assays show strong correlation with oocyte yield following controlled ovarian stimulation, with correlation coefficients of 0.74 and 0.655, respectively [4] [20]. These clinical correlations provide validation that despite systematic biases between methods, each assay maintains predictive value for ovarian response when interpreted using assay-specific thresholds.
Managing systematic bias between AMH immunoassays requires a multifaceted approach that acknowledges both analytical differences and clinical implications. The documented 32% bias between the AMH Gen II ELISA and Elecsys Cobas AMH assays necessitates implementation of assay-specific reference intervals and clinical decision limits rather than attempting mathematical conversion between platforms. The superior analytical performance of automated assays, particularly their enhanced precision and lower limits of quantitation, offers significant advantages for clinical practice and research applications.
Researchers and clinicians should prioritize method-specific verification when implementing AMH testing, establishing performance characteristics in their local laboratory setting and against their specific patient population. Future efforts toward international standardization of AMH assays would significantly improve the comparability of research findings and clinical guidelines across platforms and populations. Until such standardization is achieved, the strategies outlined in this guide provide a framework for effectively managing systematic bias between AMH assays in both research and clinical settings.
In the field of reproductive medicine and clinical biochemistry, the measurement of Anti-Müllerian Hormone (AMH) has become a cornerstone for assessing ovarian reserve. The reliability of this biomarker, however, is fundamentally dependent on the analytical performance of the immunoassays used to quantify it. The total variation observed in AMH measurement is a composite of both within-person biological variability and analytical variation introduced by the assay method. According to established principles in clinical biochemistry, the optimal performance standard for an assay is achieved when its analytical coefficient of variation (CVAnalytical) is less than 0.25 times the within-person biological variation (CVWithin Biological Variation). This rigorous framework ensures that the assay itself contributes minimally to the overall variability, thus providing clinicians with reliable data for critical decisions regarding fertility treatment and ovarian response prediction. This guide provides a detailed, evidence-based comparison between two prominent AMH assays—the manual AMH Gen II ELISA and the fully automated Elecsys Cobas AMH assay—evaluating their performance against these exacting analytical goals.
Experimental Protocols and Methodologies
The comparative data presented in this guide are primarily derived from a prospective observational study detailed in multiple sources [32] [5] [16]. The methodology was designed to facilitate a direct head-to-head comparison of the two assays under investigation.
Study Population and Sample Collection
The study enrolled 23 women with a median age of 36 years who were seeking laparoscopic sterilization. Key exclusion criteria encompassed endocrine diseases, oligomenorrhea, polycystic ovarian syndrome (PCOS), and recent hormonal contraceptive use or pregnancy. This selective criteria ensured a cohort representative of women with normal ovarian function for a valid assessment of biological variation. A total of 112 serum samples were collected from each participant at multiple time points: preoperatively, as well as one week, one month, three months, and six months postoperatively. This longitudinal design was crucial for capturing both analytical and biological variability. Serum was separated within four hours of collection and stored at -80°C until batch analysis, preserving sample integrity [5] [16].
Analytical Procedures
All biochemical measurements were performed in an ISO 15189 accredited clinical laboratory by skilled technicians, ensuring high standards of analytical practice [5] [16].
- AMH Gen II ELISA (Beckman Coulter): This manual assay was performed using the standard protocol, which involves a pre-mixture of samples, calibrators, and controls. Samples were analyzed in duplicate, with the analysis repeated if the results differed by more than 15%. The stated limit of quantitation (LOQ) for this assay was 3.0 pmol/L, with a measuring range of 3–70 pmol/L without dilution [5] [16].
- Elecsys Cobas AMH Immunoassay (Roche Diagnostics): This fully automated assay was run on a Cobas 6000 e601 platform according to the manufacturer's standard protocol. It boasts a lower LOQ of 0.5 pmol/L and a broader measuring range of 0.5–160 pmol/L without dilution [5] [16].
Data and Statistical Analysis
The correlation between AMH levels and antral follicle count (AFC) was assessed using Spearman's correlation test. The comparison between the two assays was performed using Passing-Bablok regression and bias plots. The core of the evaluation involved calculating the analytical (CVAnalytical), within-person biological (CVWithin-person), and total (CVTotal) coefficients of variation. These values were then applied to the optimal performance goal: CVAnalytical < 0.25 * CVWithin Biological Variation [32] [5].
Comparative Performance Data
The following tables synthesize the key quantitative findings from the study, providing a clear, direct comparison of the two assays' analytical performance.
Table 1: Key Analytical Performance Characteristics of AMH Gen II ELISA and Elecsys Cobas AMH Assays
| Performance Characteristic | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Assay Format | Manual | Fully Automated [7] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L [5] [16] | 0.5 pmol/L [5] [16] |
| Measuring Range | 3–70 pmol/L [5] [16] | 0.5–160 pmol/L [5] [16] |
| Run Time | Not Specified (Manual) | 18 minutes [7] |
| Sample Volume | Not Specified | As low as 30 µL [7] |
Table 2: Analytical Imprecision and Performance Goal Achievement
| Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Analytical CV (at ~7 pmol/L) | 10.3% [5] [16] | 2.8% [5] [16] |
| Analytical CV (at ~40 pmol/L) | 5.2% [5] [16] | 3.3% [5] [16] |
| Within-Person Biological CV (Concentrations >10 pmol/L) | ~22% [5] [16] | ~22% [5] [16] |
| Optimal Performance Goal (0.25 x CVWithin) | < 5.5% | < 5.5% |
| Meets Optimal Goal? (High Concentration) | Yes (CV 5.2% < 5.5%) | Yes (CV 3.3% < 5.5%) |
| Within-Person Biological CV (Concentrations <10 pmol/L) | ~32% [5] [16] | ~28% [5] [16] |
| Optimal Performance Goal (0.25 x CVWithin) | < 8.0% | < 7.0% |
| Meets Optimal Goal? (Low Concentration) | No (CV 10.3% > 8.0%) | Yes (CV 2.8% < 7.0%) |
Table 3: Method Comparison and Clinical Utility
| Aspect | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Systematic Bias vs. ELISA | Reference | -32% on average [32] [5] |
| Samples Below LOQ in Study | 15% [5] [16] | 2% [5] [16] |
| Correlation with Antral Follicle Count (AFC) | Strong (r = 0.86 at baseline) [5] [16] | Strong (r = 0.83 at baseline) [5] [16] |
Performance Evaluation Workflow
The diagram below visualizes the logical process of evaluating an assay's performance against the optimal goal, leading to the conclusive findings of this study.
Implications for Research and Clinical Practice
The observed performance characteristics have direct and significant implications for both research and clinical settings.
- Assay Selection for Low AMH Concentrations: The superior precision and lower LOQ of the Elecsys Cobas assay make it particularly advantageous for patient populations with diminished ovarian reserve, where accurate quantification of low AMH levels is critical for diagnosis and treatment planning [5] [16]. The AMH Gen II ELISA's high imprecision and failure to meet optimal performance goals at low concentrations could lead to greater uncertainty in clinical decision-making for these patients.
- Impact of Systematic Bias: The consistent negative bias (approximately -32%) of the Elecsys assay relative to the Gen II ELISA underscores the absolute necessity of using method-specific reference intervals [32] [5]. Researchers and clinicians cannot interchange results or use universal clinical decision points between these two assays. This bias is consistent with a large multicenter study which also found Elecsys values to be 24-28% lower than those from a manual ELISA method [10].
- Broader Context of AMH Variability: It is crucial to note that even with an optimal assay, AMH levels exhibit significant biological variability. A 2024 study reported a median inter-cycle variation of 44.3% in consecutive menstrual cycles, which can lead to the reclassification of up to 20% of patients between normal and poor responder categories [51]. This highlights that analytical variation is only one component of the total testing variance and reinforces the need for high-quality assays to minimize their contribution to this overall uncertainty.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents and Materials for AMH Assay Comparison Studies
| Item | Function/Description |
|---|---|
| Elecsys Cobas AMH Assay | A fully automated electrochemiluminescence immunoassay (ECLIA) for the quantitative determination of AMH in human serum and plasma on Cobas e analyzers [7]. |
| AMH Gen II ELISA | A manual two-site immunoassay (ELISA) using antibodies against epitopes in both the pro- and mature regions of AMH, historically used for AMH quantification [46] [5]. |
| Precision Controls | Commercially available control materials at multiple concentration levels (e.g., low, medium, high) used to determine the assay's analytical imprecision (CV%) over time [5] [16]. |
| Calibrators | Manufacturer-provided materials with defined analyte concentrations used to establish the standard curve for converting assay signal (e.g., absorbance, luminescence) into AMH concentration values [46]. |
| Standard Diluent | A protein-based matrix used to serially dilute the calibrators to construct the standard curve and to ensure appropriate sample matrix conditions [46]. |
| Antral Follicle Count (AFC) | A ultrasound-based morphological assessment used as a reference standard to validate the clinical correlation of the AMH measurements [5] [51]. |
The rigorous evaluation of the AMH Gen II ELISA and Elecsys Cobas AMH assays against the optimal performance goal of CVAnalytical < 0.25*CVWithin Biological Variation yields a clear and data-driven conclusion. The Elecsys Cobas AMH assay demonstrates superior analytical performance, meeting the optimal standard across its entire measuring range due to its excellent precision (CV <5%) and low limit of quantitation. In contrast, the AMH Gen II ELISA achieves optimal performance only at high concentrations and fails to meet the required standard at low concentrations, where its imprecision is highest. For researchers and clinicians requiring the most reliable data for low AMH levels and robust, high-throughput automated testing, the Elecsys Cobas AMH assay presents a technically superior alternative. This assessment underscores the critical importance of validating analytical performance against biological variation to ensure the integrity of both clinical diagnostics and research data.
The Critical Role of Sensitive Assays in Special Populations (e.g., POI)
The accurate measurement of Anti-Müllerian Hormone (AMH) has become indispensable in reproductive medicine for assessing ovarian reserve, predicting response to ovarian stimulation, and diagnosing conditions such as polycystic ovarian syndrome (PCOS) and premature ovarian insufficiency (POI) [5] [20]. However, the clinical utility of AMH measurements is highly dependent on the analytical performance of the immunoassays used, particularly regarding their sensitivity, specificity, and precision. This dependency becomes critically important in special populations, such as women with POI or severely diminished ovarian reserve, where AMH levels are exceedingly low and often fall below the detection limits of conventional clinical assays [52] [18]. The development of highly sensitive assays has therefore emerged as a pivotal advancement, enabling clinicians and researchers to detect and quantify these minimal AMH concentrations, thereby facilitating more accurate diagnosis, better prediction of treatment outcomes, and more personalized therapeutic strategies for these challenging clinical cases.
Comparative Analysis of AMH Assay Platforms
Technical Specifications and Performance Characteristics
Table 1: Performance Characteristics of AMH Immunoassays
| Assay Characteristic | AMH Gen II ELISA (Beckman Coulter) | Elecsys Cobas AMH (Roche) | Access AMH (Beckman Coulter) | picoAMH ELISA (Ansh Labs) |
|---|---|---|---|---|
| Platform Type | Manual ELISA [5] | Fully Automated [5] | Fully Automated [20] [6] | Manual ELISA [52] |
| Limit of Quantitation (LOQ) | 3.0 pmol/L [5] | 0.5 pmol/L [5] | 0.01 ng/mL (~0.07 pmol/L) [6] | 1.3 pg/mL (~0.09 pmol/L) [52] |
| Measuring Range | 3–70 pmol/L [5] | 0.5–160 pmol/L [5] | Up to 24 ng/mL [6] | Not specified |
| Analytical Variability (CV%) | 5.5% - 10.3% [5] | 2.8% - 3.3% [5] | 2.4% - 5.2% [6] | Intra-assay: 2.5-5.5%; Inter-assay: 3.7-8.1% [52] |
| Key Clinical Advantage | Historical standard | Optimal performance throughout measuring range [5] | High precision and stability [6] | Superior sensitivity for very low levels [52] |
The data reveals a clear evolution from manual to automated platforms, with significant improvements in analytical precision. The Elecsys Cobas and Access AMH assays demonstrate lower coefficients of variation (CV%) compared to the manual Gen II ELISA, indicating superior reproducibility and reliability [5] [6]. Furthermore, the limit of quantitation has progressively decreased with newer assays, a critical factor for assessing ovarian function in patients with severely diminished ovarian reserve.
Correlation and Bias Between Assay Methods
While different AMH assays generally show good correlation, significant absolute biases exist that prevent direct interchangeability of results. A prospective clinical study directly comparing the AMH Gen II ELISA and the Elecsys Cobas AMH method found a consistent bias of approximately 32%, with Elecsys values being lower than the ELISA values [5] [32]. This bias showed a tendency to increase at higher AMH concentrations [5]. Similarly, the Access AMH assay also yields slightly lower values than the Gen II pre-mix assay, with a regression slope of 0.711 to 0.755 [20]. These discrepancies are attributed to differences in antibody specificity, assay calibration, and the ability to recognize different molecular isoforms of AMH [18]. Consequently, clinical decision-making and interpretation of AMH values must be performed within the context of the specific assay used and its associated reference intervals.
The Critical Application in Premature Ovarian Insufficiency (POI)
The Challenge of Detecting Minimal Ovarian Activity
Premature Ovarian Insufficiency (POI) is characterized by the loss of ovarian function before the age of 40, leading to amenorrhea, elevated gonadotropins, and estrogen deficiency [52] [53]. A hallmark of POI is a severe reduction in the primordial follicle pool, resulting in AMH levels that are typically very low and often undetectable by standard clinical assays [52]. However, some patients with POI may experience sporadic, unpredictable ovarian activity with intermittent follicular development, offering a chance for fertility treatment [52]. Identifying these patients and predicting the likelihood of follicular growth in response to prolonged ovarian stimulation is a significant clinical challenge. Traditional markers like estradiol (E2) and follicle-stimulating hormone (FSH) are of limited value in this context, as E2 can remain undetectable and FSH is influenced by exogenous hormone therapy used in treatment protocols [52]. Ultrasound is only capable of detecting follicles once they reach a size of 2 mm or more, missing earlier stages of development [52]. This creates a pressing need for a more sensitive biomarker that can detect early-stage follicle development and guide treatment decisions.
Superior Predictive Value of Highly Sensitive AMH Assays
The picoAMH ELISA, with its exceptionally low limit of detection (1.3 pg/mL), has demonstrated a superior ability to predict follicular development in POI patients undergoing controlled ovarian stimulation. In a retrospective study of 165 POI patients undergoing 504 long stimulation cycles, AMH levels measured three weeks after stimulation initiation showed outstanding predictive capability for subsequent follicular development detected by ultrasound [52]. The area under the curve (AUC) for this prediction was 0.957, indicating excellent test performance. The study identified an optimal AMH threshold of 2.45 pg/mL for this purpose [52]. Furthermore, a negative correlation was observed between the 3-week AMH level and the time required for follicular detection (R = -0.326), meaning that higher AMH levels predicted a shorter time to observing follicles on ultrasound [52]. This application highlights the transformative role of assay sensitivity: where conventional assays report "undetectable" levels, the picoAMH assay provides a quantifiable value that can directly inform clinical management, such as the decision to extend stimulation in hopes of achieving follicular growth.
Experimental Protocols for Assay Comparison and Validation
Protocol for a Method Comparison Study
The following protocol is derived from a published prospective study comparing the AMH Gen II ELISA and Elecsys Cobas AMH assays [5].
- Sample Collection: Blood samples are collected from study participants (e.g., women of reproductive age) at multiple time points. Serum is separated within 4 hours of sampling and stored at -80°C until analysis to preserve AMH stability [5].
- Sample Analysis: Frozen samples are thawed and analyzed in parallel using both assay platforms. For the manual Gen II ELISA, samples are analyzed in duplicate following the standard application protocol, including a pre-mixture step. If duplicate results differ by more than 15%, the analysis is repeated [5]. The Elecsys Cobas AMH assay is run on a Cobas 6000 e601 platform using the manufacturer's standard protocol for automated analysis [5].
- Data Analysis: The correlation between the two methods is assessed using Spearman's correlation test. Passing-Bablok regression and Bland-Altman bias plots are constructed to evaluate the agreement and systematic bias between the two methods [5]. Analytical goals are validated by comparing the analytical variability (CVAnalytical) of each method to the within-person biological variation, with optimal performance defined as CVAnalytical < 0.25 * CVWithin Biological Variation [5].
Protocol for Assessing Predictive Value in POI Patients
This protocol is based on a retrospective study evaluating the picoAMH ELISA in POI patients [52].
- Patient Population: Women diagnosed with POI according to established criteria (e.g., age <40, amenorrhea >3 months, and elevated FSH levels) who are undergoing prolonged controlled ovarian stimulation are enrolled [52].
- Stimulation and Monitoring: Patients undergo a long GnRH-agonist protocol with gonadotropin stimulation. Standard hormonal monitoring (FSH, LH, E2, P) is performed weekly via automated immunoassay analyzers [52].
- AMH Measurement: A critical component is the measurement of serum AMH levels three weeks (days 18-27) after the initiation of stimulation. This is performed using the highly sensitive picoAMH ELISA to detect minimal hormone concentrations [52].
- Outcome Assessment: The primary outcome is the development of ultrasound-detectable antral follicles (≥ 2 mm) during the stimulation cycle. The correlation between the 3-week AMH level and the occurrence and timing of follicular development is analyzed using receiver operating characteristic (ROC) curves to determine predictive accuracy and optimal threshold values [52].
Advancements in Assay Specificity: Targeting AMH Isoforms
Emerging research focuses on developing high-specificity AMH assays that target distinct linear epitopes on different molecular isoforms of the hormone. Circulating AMH is not a single molecule but a mixture of isoforms, including the non-active proAMH and the biologically active AMHN,C, along with various sub-fragments [18]. Conventional clinical assays use antibodies against conformational epitopes and provide an aggregate measure of these different species, which can lead to platform-specific variation [18]. Novel ELISA assays (e.g., AL-196, AL-124, AL-105, AL-133) utilize monoclonal antibodies directed against specific linear epitopes on the AMH molecule, such as the pro-region (AMHN) or the mature region (AMHC) [18]. In patients with low ovarian reserve, these high-specific assays have shown improved accuracy in predicting oocyte yield following ovarian stimulation compared to conventional automated assays. A prospective study found that a model combining AFC and the AL-196 assay provided the best predictive value for the number of cumulus-oocyte complexes and mature oocytes retrieved [18]. This suggests that knowledge of the relative levels of specific AMH isoforms, rather than just the total concentration, may offer a more refined understanding of follicular dynamics and responsiveness to stimulation, particularly in challenging patient populations.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Research Reagents for Advanced AMH Studies
| Reagent / Assay | Manufacturer | Key Features | Primary Research Application |
|---|---|---|---|
| picoAMH ELISA (AL-124) | Ansh Labs [52] | Ultra-sensitive (LoD: 1.3 pg/mL), detects proAMH & AMHN,C [52] | Detecting very low AMH levels in POI, menopause research [52] |
| PCOCheck AMH ELISA (AL-196) | Ansh Labs [18] | Linear epitope antibody, detects proAMH, AMHN,C & AMHN [18] | Isoform-specific studies, PCOS research, low reserve prediction [18] |
| Total Mature AMH ELISA (AL-133) | Ansh Labs [18] | Detects proAMH, AMHN,C & AMHC [18] | Quantifying total mature AMH, fertility assessment studies |
| Elecsys Cobas AMH Assay | Roche Diagnostics [5] | Fully automated, low LOQ (0.5 pmol/L), high precision (CV: 2.8-3.3%) [5] | High-throughput clinical studies, ovarian response prediction [5] [4] |
| Access AMH Assay | Beckman Coulter [20] [6] | Fully automated, precise (CV: 2.4-5.2%), stable [6] | Correlative studies with oocyte yield, large cohort analysis [20] |
The critical role of sensitive and specific AMH assays in evaluating special populations like women with Premature Ovarian Insufficiency is unequivocal. The progression from manual ELISAs to automated immunoassays and, more recently, to ultra-sensitive and high-specificity assays represents a significant advancement in reproductive endocrinology. These technological improvements have transformed the clinical management of POI patients, enabling the detection of minimal residual ovarian function that was previously unmeasurable and providing a valuable biomarker for predicting the likelihood of follicular development in response to prolonged stimulation. For researchers and clinicians, the selection of an appropriate AMH assay must be guided by the specific clinical or research question, with a clear understanding that performance characteristics, particularly sensitivity at the lower end of the measuring range and specificity for AMH isoforms, are paramount when studying special populations with severely diminished ovarian reserve.
Clinical Concordance and Predictive Value Assessment
This guide provides a comparative analysis of Antral Follicle Count (AFC) and ovarian volume as biomarkers of ovarian reserve, contextualized within broader research on AMH assay methodologies. As researchers and drug development professionals seek the most reliable markers for assessing ovarian function, understanding how these ultrasound-based measures correlate with histological gold standards and predict clinical outcomes is paramount. This objective comparison synthesizes current experimental data to evaluate the performance characteristics, validation evidence, and practical applications of AFC and ovarian volume in both clinical and research settings.
The quantitative assessment of ovarian reserve, representing the remaining pool of primordial follicles, is crucial in fertility research and clinical practice. While the direct histological quantification of primordial follicles constitutes the biological gold standard, this method is invasive and impractical for clinical use [54]. Consequently, the field relies on surrogate markers that can be measured non-invasively. Anti-Müllerian Hormone (AMH), Antral Follicle Count (AFC), and ovarian volume have emerged as the leading modalities for this purpose. Within the context of advancing AMH assay technologies—such as the AMH Gen II ELISA and the Elecsys Cobas AMH assay—it becomes essential to understand how traditional ultrasound markers compare in terms of standardization, correlation with true follicular pools, and predictive value for reproductive outcomes. This guide objectively examines the experimental evidence supporting AFC and ovarian volume, their correlation with established standards, and their role in a comprehensive ovarian reserve assessment strategy.
Biomarker Fundamentals and Measurement Protocols
Antral Follicle Count (AFC)
Definition and Biological Basis: Antral follicles are small, fluid-filled sacs measuring 2-10 mm in diameter that are present in the ovaries during the early follicular phase of the menstrual cycle [55] [56]. These follicles represent the cohort of follicles that have been recruited from the primordial pool and are progressing through the developmental stages. The AFC is the total number of these follicles counted in both ovaries via transvaginal ultrasonography [56]. The physiological premise is that the number of visible antral follicles is directly proportional to the size of the remaining, microscopic primordial follicle pool [56] [54]. This relationship makes AFC a valuable indirect marker of ovarian reserve.
Standardized Measurement Protocol: To ensure consistency and reproducibility, the AFC assessment follows a specific protocol:
- Timing: Measurement is performed on cycle days 2-5 of a spontaneous menstrual cycle [57] [54].
- Equipment: A high-resolution transvaginal ultrasound probe with a frequency of 5-9 MHz is used [54].
- Procedure: The examiner systematically scans each ovary in multiple planes. Follicles between 2 mm and 10 mm in mean diameter are counted. The diameter is typically determined by measuring the mean of two perpendicular dimensions [55] [58].
- Reporting: The counts from the left and right ovaries are summed to produce the total AFC [57].
Ovarian Volume
Definition and Biological Basis: Ovarian volume is a measure of the three-dimensional size of the ovary, calculated from ultrasonographic measurements. The underlying rationale is that ovarian volume decreases as the primordial follicle pool is depleted with age, making it a structural indicator of ovarian reserve [55]. Studies have confirmed that a decrease in ovarian volume is an early sign of follicle depletion and is clinically useful in assessing reproductive status [55].
Standardized Measurement Protocol: The procedure for calculating ovarian volume is geometric:
- Measurement: Each ovary is measured in three perpendicular dimensions (D1, D2, D3) [55] [59].
- Formula: The volume for each ovary is calculated using the formula for an ellipsoid: Volume = (D1 × D2 × D3 × π/6) [55] [59].
- Reporting: The volumes of both ovaries are often added together to report a total basal ovarian volume (BOV) [55].
Correlation with Histological Gold Standards
The most direct validation for any ovarian reserve marker is its correlation with the true, histologically quantified primordial follicle count. A 2025 prospective study provides the most recent and compelling evidence on this relationship.
Table 1: Correlation of AFC and Ovarian Volume with Histological Primordial Follicle Count
| Biomarker | Study Design | Population | Correlation with Primordial Follicle Count (ρ) | P-value | Source |
|---|---|---|---|---|---|
| Antral Follicle Count (AFC) | Prospective cross-sectional | 89 women, aged 35-48 | 0.85 | < 0.001 | [54] |
| Ovarian Volume | (Data not explicitly provided in the study) | ||||
| Serum AMH | Prospective cross-sectional | 89 women, aged 35-48 | 0.75 | < 0.001 | [54] |
This study demonstrated that both AFC and AMH are strongly associated with the histological size of the primordial follicle pool, providing robust histological validation for their use. Notably, AFC showed a very strong positive correlation (ρ=0.85), indicating it is an excellent non-invasive surrogate for the direct assessment of the ovarian reserve [54]. The study concluded that these biomarkers represent reliable, non-invasive markers for reproductive assessment and individualized fertility planning [54].
Predictive Value for Clinical Outcomes in ART
The utility of ovarian reserve markers is ultimately judged by their ability to predict outcomes in assisted reproductive technology (ART). Both AFC and ovarian volume have been extensively studied for this purpose.
Prediction of Ovarian Response
The primary clinical application of AFC is to predict how a patient's ovaries will respond to exogenous gonadotropin stimulation.
Table 2: Predictive Value of AFC for Ovarian Response in IVF
| Total AFC | Classification | Expected Ovarian Response to Stimulation | Typical Clinical Action |
|---|---|---|---|
| < 4 | Extremely Low | Very poor or no response. High cancellation rate. | Consider very high dose stimulation or donor eggs. [56] |
| 4 - 9 | Low | Poor response. Likely to need high doses of FSH. | Use aggressive stimulation protocol. [56] [57] |
| 9 - 21 | Intermediate/Normal | Variable, but usually good response. | Standard stimulation protocol. [56] [57] |
| 22 - 35 | Normal/High | Excellent response to low-dose FSH. | Use low-dose stimulation to prevent over-response. [56] |
| > 35 | Very High | High risk of hyper-response and OHSS. | Use low-dose FSH and consider GnRH agonist trigger. [56] |
A 2007 prospective study randomized to compare tests found that AFC was a superior predictor for hyper-response (ROC-AUC = 0.92), with a sensitivity of 82% and a specificity of 89% [55]. The same study concluded that "AFC performs well as a test for ovarian response being superior or at least similar to complex expensive and time consuming endocrine tests" and is likely the best test for general practice [55]. In a multivariate analysis, the best prediction model for ovarian response included AFC, the Inhibin B-increment, and basal ovarian volume simultaneously [55].
Prediction of IVF Success and Cycle Cancellation
AFC is strongly associated with live birth rates and the risk of cycle cancellation, as shown by clinical data from a fertility center. The association between success rates and female age remains significant, but within each age group, a higher AFC is linked to better outcomes [56].
- Live Birth Rates: For women under 35, live birth rates can be over 40% with normal AFC (>10) but drop significantly to 15-20% with very low AFC (<5) [56].
- Cycle Cancellation: The risk of cycle cancellation is highly dependent on AFC. For women under 35, the cancellation rate is below 10% with an AFC >10, but rises to over 50% with an AFC <5 [56]. This pattern of increasing cancellation with lower AFC holds true across all age groups [56].
Comparative Analysis with Other Biomarkers
AFC vs. Ovarian Volume
While both are ultrasound-derived measures, AFC and ovarian volume offer different strengths. A meta-analysis cited that "ultrasonography as a tool for the prediction of outcome in IVF patients" compared ovarian volume and AFC, and the evidence supports AFC as the more powerful predictor [60]. The 2007 study also placed AFC in a superior position for predicting hyper-response compared to basal ovarian volume [55].
AFC vs. AMH
AMH has become a leading endocrine marker for ovarian reserve. The correlation between AFC and AMH is well-established, with one study finding a Spearman's correlation coefficient of 0.83-0.86 between Cobas AMH and AFC, and 0.81-0.86 between ELISA AMH and AFC [5]. This strong correlation confirms that both markers reflect the same biological reality—the population of small antral follicles.
However, it is critical to note that assay methodology significantly influences AMH values. A 2015 comparison study found that the fully automated Elecsys Cobas AMH assay showed a mean negative bias of approximately 32% compared to the manual AMH Gen II ELISA [5] [32]. Furthermore, the Elecsys Cobas assay demonstrated superior analytical performance with a lower limit of quantitation (0.5 pmol/L vs. 3.0 pmol/L) and significantly better precision throughout the measuring range [5] [32]. Newer automated assays, like the Lumipulse G AMH, continue to be developed, showing good comparability but variable biases against existing methods [61]. This lack of standardization is a key challenge for AMH that does not affect AFC, as the ultrasound measurement is less dependent on the assay platform.
Experimental Workflow and Research Toolkit
Experimental Workflow for Ovarian Reserve Assessment
The following diagram visualizes the standard experimental workflow for validating ovarian reserve biomarkers against the histological gold standard, as performed in contemporary studies.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Ovarian Reserve Biomarker Research
| Item | Function/Application | Example Specifications / Notes |
|---|---|---|
| Transvaginal Ultrasound System | Imaging for AFC and ovarian volume measurement. | High-resolution system with 5-9 MHz endovaginal transducer (e.g., GE Voluson E10) [54]. |
| AMH Immunoassay Kits | Quantitative measurement of serum AMH levels. | Specific assays include AMH Gen II ELISA (Beckman Coulter) and Elecsys Cobas AMH (Roche) [5] [61]. |
| ELISA Microplate Reader | Absorbance reading for manual AMH ELISA. | Required for manual or semi-automated AMH assays. |
| Automated Immunoassay Analyzer | High-throughput, automated AMH quantification. | Platforms include Cobas e601 (Roche) or Lumipulse G600II (Fujirebio) [5] [61]. |
| Follicle-Stimulating Hormone (FSH) Kit | Measurement of basal FSH, a traditional ovarian reserve marker. | Often used as a comparator in validation studies (e.g., Roche Elecsys) [54]. |
| Microtome | Sectioning of paraffin-embedded ovarian tissue for histology. | Produces thin sections (e.g., 5 μm) for staining and analysis [54]. |
| Light Microscope | Visualization and counting of primordial follicles in tissue sections. | Essential for gold standard quantification (e.g., Olympus BX53) [54]. |
The body of evidence firmly establishes Antral Follicle Count (AFC) as a robust and highly reliable biomarker for assessing ovarian reserve. Its very strong correlation with the histological primordial follicle pool (ρ=0.85) validates its biological basis, while its superior performance in predicting ovarian response and IVF outcomes underscores its clinical utility [55] [54]. Ovarian volume, while informative, is generally considered a secondary marker in this context.
Within the broader thesis of AMH assay research, AFC serves as a crucial, method-independent correlate. The documented variability and lack of standardization between different AMH assays, such as the ~32% bias between the Gen II ELISA and Elecsys Cobas methods, highlight a significant challenge in the field [5] [32] [61]. In contrast, AFC provides a standardized, anatomical benchmark against which new and existing AMH assays can be validated and calibrated. For researchers and drug developers, prioritizing AFC in study designs for patient stratification and as a primary endpoint in trials investigating novel fertility therapeutics ensures results are based on a stable and widely accepted metric. Future research should focus on further standardizing AFC protocols across operators and leveraging artificial intelligence to automate counts, thereby enhancing reproducibility and solidifying its role as a cornerstone in ovarian reserve evaluation.
Head-to-Head Performance in Predicting Ovarian Response in IVF
Within the framework of ongoing research on Anti-Müllerian Hormone (AMH) assays, the comparison between the AMH Gen II ELISA and the Elecsys Cobas AMH assay represents a critical point of investigation. AMH has become a cornerstone biomarker for assessing functional ovarian reserve, providing crucial information for predicting ovarian response to stimulation in In Vitro Fertilization (IVF). Its level is strongly correlated with the antral follicle count (AFC) and serves as a reliable predictor of the number of oocytes retrieved following ovarian stimulation. The accurate and precise measurement of AMH is therefore fundamental to individualizing treatment protocols and optimizing outcomes in assisted reproductive technology.
This guide provides an objective, data-driven comparison of two principal methods for measuring AMH: the established manual AMH Gen II ELISA (Beckman Coulter) and the newer, fully automated Elecsys Cobas AMH assay (Roche Diagnostics). For researchers and clinicians, understanding the technical and performance characteristics of these assays is essential for both interpreting individual patient results and for designing robust clinical studies.
Experimental Protocols & Methodologies
To ensure a valid comparison, studies have been conducted using head-to-head methodologies on the same set of patient samples. The following outlines the standard experimental protocols used in these comparative studies.
Sample Collection and Handling
In a typical prospective observational study, blood samples are collected from participants (e.g., women undergoing sterilization or infertility treatment) at multiple time points [5]. The serum is isolated within a few hours of sampling and stored at -80°C until the analysis to preserve the stability of the AMH molecule [5]. All samples are then analyzed in parallel using both the AMH Gen II ELISA and the Elecsys Cobas AMH methods, ensuring a direct comparison under identical pre-analytical conditions.
The AMH Gen II ELISA Protocol
The AMH Gen II ELISA is a manual, two-site immunoassay [5]. The protocol involves a pre-mixture of clinical samples, calibrators, and controls in an assay buffer [5]. This manual technique requires analysts to perform multiple pipetting and washing steps. Samples are typically analyzed in duplicate, and if the duplicate results differ by more than a pre-set percentage (e.g., 15%), the analysis must be repeated, adding to the hands-on time and complexity [5]. The acceptance criteria for internal controls are usually set at a CV of less than 11-14% [5].
The Elecsys Cobas AMH Protocol
The Elecsys Cobas AMH assay is a fully automated electrochemiluminescence immunoassay (ECLIA) designed for use on Roche's Cobas e analyzers (e.g., e 411, e 601, e 801) [7]. The process is entirely automated: the instrument handles all sample dilution, incubation, and measurement steps. The assay time is approximately 18 minutes, allowing for same-day result reporting [7]. The system requires a low sample volume (as low as 30 μL) and has an onboard stability of up to 112 days for reagents [7].
The workflow below illustrates the core procedural differences between the two methods:
Comparative Analytical Performance Data
The core of the assay comparison lies in their analytical performance metrics. The data below, synthesized from independent studies, highlights the critical differences between the two methods.
Table 1: Head-to-Head Analytical Performance Comparison
| Performance Characteristic | AMH Gen II ELISA | Elecsys Cobas AMH | Clinical & Research Implication |
|---|---|---|---|
| Assay Format | Manual immunoassay [5] | Fully automated immunoassay [7] | Throughput & standardization; automated reduces hands-on time and operator-induced variability. |
| Analytical Variability (CV%) | 5.5% - 10.3% [5] | 2.8% - 3.3% [5] | Precision & reliability; lower CV indicates higher reproducibility and more consistent results. |
| Limit of Quantitation (LoQ) | 3.0 pmol/L [5] | 0.5 pmol/L [5] | Sensitivity for low values; crucial for accurately assessing diminished ovarian reserve. |
| Measuring Range | 3–70 pmol/L (without dilution) [5] | 0.5–160 pmol/L (without dilution) [7] | Working range; wider range minimizes need for sample dilution, reducing potential for error. |
| Sample Volume | Not specified in search results | As low as 30 μL [7] | Practicality; lower volume is beneficial for pediatric patients or when sample volume is limited. |
| Correlation with AFC | Spearman r: ~0.81-0.86 [5] | Spearman r: ~0.83 [5] | Clinical utility; both show strong correlation with the ultrasound marker of ovarian reserve. |
| Systematic Bias | Reference method | Approximately 32% lower than Gen II ELISA [5] | Result interpretation; values are not directly interchangeable; method-specific reference ranges are essential. |
Table 2: Performance Against Optimal Analytical Goals
| Concentration Range | Total Variance (CV%) | Achieves Optimal Performance? | ||
|---|---|---|---|---|
| AMH Gen II ELISA | Elecsys Cobas AMH | AMH Gen II ELISA | Elecsys Cobas AMH | |
| Low Range (<10 pmol/L) | 31.9% [5] | 28.0% [5] | No [5] | Yes [5] |
| High Range (>10 pmol/L) | 22.0% [5] | 21.3% [5] | Yes [5] | Yes [5] |
The relationship between different AMH assays and their performance across the measuring range is complex. The following diagram summarizes the key comparative findings and their logical connections:
The Scientist's Toolkit: Key Research Reagents & Materials
For scientists seeking to replicate these comparative studies or implement these assays in a research setting, the following table details essential materials and their functions.
Table 3: Essential Research Reagents and Materials
| Item | Function in AMH Measurement |
|---|---|
| Serum Samples | The biological matrix containing the AMH analyte. Standardized collection and processing (e.g., centrifugation within 4h, storage at -80°C) are critical for pre-analytical stability [5]. |
| AMH Calibrators | Solutions with known concentrations of AMH, used to generate the standard curve for converting instrument signal (e.g., absorbance, luminescence) into a quantitative concentration [5]. |
| Quality Control (QC) Materials | Typically available at multiple levels (e.g., low, medium, high), these are used to monitor the precision and accuracy of the assay run over time [5]. |
| Assay Buffer / Sample Diluent | A solution used to dilute samples, calibrators, and controls to within the assay's analytical measurement range. The choice of diluent can affect recovery and results [2]. |
| Elecsys Cobas Analyzer | The automated immunoassay platform (e.g., cobas e 601) that performs all steps of the Elecsys AMH assay, including pipetting, incubation, and signal detection [7]. |
| Microplate Washer & Reader | Essential instrumentation for the manual ELISA process. The washer removes unbound material, and the reader measures the color intensity developed by the enzyme reaction [5]. |
The direct comparison between the AMH Gen II ELISA and the Elecsys Cobas AMH assays reveals a clear evolution in the measurement of this critical biomarker. While both assays demonstrate a strong correlation with AFC and are useful in predicting ovarian response in IVF, the Elecsys Cobas system demonstrates superior analytical performance. Key advantages include its fully automated format, which reduces hands-on time and operational variability; its lower analytical imprecision across the entire measuring range; and its enhanced sensitivity, allowing for more reliable quantification in patients with very low ovarian reserve.
A critical finding for the research and clinical community is the consistent bias observed between the methods, with the Elecsys assay reporting values approximately 32% lower than the Gen II ELISA [5]. This underscores that AMH values are not interchangeable between different methods. Consequently, the establishment and use of method-specific reference ranges is non-negotiable for the correct interpretation of results in both clinical practice and scientific studies. The move towards automated assays like the Elecsys Cobas AMH represents a step forward in standardizing AMH measurement, ultimately contributing to more reliable and actionable data for managing patients in an IVF setting.
The accurate measurement of Anti-Müllerian Hormone (AMH) has become fundamental in reproductive medicine, particularly for assessing ovarian reserve and diagnosing polycystic ovary syndrome (PCOS). The evolution from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated immunoassays has significantly improved test performance, yet it has also introduced critical challenges in establishing and validating standardized clinical cut-offs. This comparison guide objectively evaluates two prominent AMH detection methods—the AMH Gen II ELISA (Beckman Coulter) and the Elecsys Cobas AMH assay (Roche Diagnostics)—focusing on their analytical performance, clinical validation data, and application in diagnosing polycystic ovarian morphology (PCOM) within the broader PCOS diagnostic framework. For researchers and drug development professionals, understanding these methodological differences is essential for appropriate assay selection, clinical interpretation, and future assay development.
Analytical Performance Comparison
A direct comparison of the technical specifications and performance characteristics of the AMH Gen II ELISA and the Elecsys Cobas AMH assay reveals significant differences that impact their clinical application.
Table 1: Analytical Performance Characteristics of AMH Assays
| Performance Characteristic | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Methodology | Manual ELISA | Automated Immunoassay |
| Limit of Quantitation (LOQ) | 3.0 pmol/L | 0.5 pmol/L |
| Measuring Range | 3–70 pmol/L | 0.5–164.00 pmol/L |
| Analytical Variability (CV%) | 5.5–10.3% | 2.8–3.3% |
| Control 1 (~40 pmol/L) | 5.2% | 3.3% |
| Control 3 (~7 pmol/L) | 10.3% | 2.8% |
| Samples Without Quantitation | 15% | 2% |
| Total Variance (<10 pmol/L) | 31.9% | 28.0% |
Data derived from comparative studies [5] [32] demonstrate that the Elecsys Cobas method exhibits superior analytical precision with significantly lower coefficients of variation (CV%) across the measuring range compared to the AMH Gen II ELISA. The automated Elecsys system also offers a broader functional range and enhanced sensitivity, with a limit of quantitation six times lower than the manual ELISA method (0.5 pmol/L vs. 3.0 pmol/L) [5]. This improved sensitivity at lower concentrations reduces the percentage of samples that cannot be quantified from 15% with the ELISA to just 2% with the Elecsys system [5], providing more clinically actionable results across diverse patient populations.
The total variance observed in clinical samples (combining biological and analytical variability) is notably lower for the Elecsys system, particularly in the critical range below 10 pmol/L (28.0% vs. 31.9%) [5]. When evaluated against optimal performance standards for biochemical assays (CVAnalytical < 0.25*CVWithin Biological Variation), the Elecsys Cobas method achieves optimal performance throughout the entire measuring range, while the AMH Gen II ELISA meets this standard only at higher concentrations [5] [32].
Method Comparison and Bias Assessment
Understanding the correlation and systematic differences between AMH assays is crucial for interpreting results across different platforms and establishing validated clinical cut-offs.
Table 2: Method Comparison and Clinical Correlation
| Comparison Parameter | AMH Gen II ELISA | Elecsys Cobas AMH |
|---|---|---|
| Correlation with AFC | 0.86 (Baseline) | 0.83 (Baseline) |
| Correlation with Ovarian Volume | Significant | Significant |
| Systematic Bias | Reference Method | Approximately 32% lower |
| Within-Person Biological Variability | 21-32% | 21-32% |
A prospective observational study comparing both methods found a good correlation between the AMH Gen II ELISA and Elecsys Cobas assays, but with a consistent systematic bias [5] [32]. The Elecsys method yields concentrations approximately 32% lower than those obtained with the AMH Gen II ELISA across the measuring range [5]. This bias demonstrates a tendency to increase at higher concentrations, highlighting the lack of harmonization between these assays [5].
Both methods maintain strong and statistically significant correlation with antral follicle count (AFC) and ovarian volume as determined by transvaginal ultrasound [5]. The correlation coefficients with total AFC at baseline were 0.86 for the AMH Gen II ELISA and 0.83 for the Elecsys Cobas system [5], confirming the clinical utility of both platforms for ovarian reserve assessment despite their systematic differences.
The total within-person biological variability ranges from approximately 21% to 32% for both methods [5], emphasizing that biological factors contribute significantly to AMH fluctuations independently of analytical considerations. This biological variation underscores the importance of establishing population-specific reference intervals and clinical cut-offs tailored to each assay method.
Clinical Validation of Cut-offs for PCOM Diagnosis
The application of standardized AMH cut-offs for diagnosing polycystic ovarian morphology represents a significant advancement in PCOS diagnostics, particularly with the Elecsys platform.
Table 3: Validated AMH Cut-offs for PCOM Diagnosis
| Assay Platform | Validated PCOM Cut-off | Sensitivity | Specificity | AUC | Study Population |
|---|---|---|---|---|---|
| Elecsys AMH Plus Immunoassay | 3.2 ng/mL (23 pmol/L) | 88.6% (95% CI 85.3-91.3) | 84.6% (95% CI 81.1-87.7) | 93.6% (95% CI 92.2-95.1) | Multicenter validation (455 cases, 500 controls) |
| Elecsys AMH Plus (Ages 25-35) | 3.2 ng/mL (23 pmol/L) | 88.5% | 80.3% | - | Age subgroup analysis |
| Elecsys AMH Plus (Ages 36-45) | 3.2 ng/mL (23 pmol/L) | 77.8% | 90.1% | - | Age subgroup analysis |
The Aphrodite study, a retrospective multicenter case-control investigation, derived and validated an AMH cutoff of 3.2 ng/mL (23 pmol/L) for the Elecsys AMH Plus immunoassay to identify PCOM [62]. This cutoff demonstrated robust performance characteristics with high sensitivity (88.6%) and specificity (84.6%) for PCOM diagnosis, with an area under the receiver-operator characteristic curve of 93.6% in the validation cohort [62]. The consistency of these results across all PCOS phenotypes (A-D according to Rotterdam criteria) supports the utility of this standardized cutoff in diverse clinical presentations of the syndrome.
Age-specific analyses revealed expected variations in test performance, with higher sensitivity (88.5% vs. 77.8%) but lower specificity (80.3% vs. 90.1%) in younger women (25-35 years) compared to older women (36-45 years) using the same 3.2 ng/mL cutoff [62]. This pattern reflects the natural decline of AMH with advancing age and highlights the potential benefit of age-adjusted interpretations in clinical practice.
Ongoing research continues to validate these cut-offs in diverse populations. The prospective HARMONIA study aims to further validate the 3.2 ng/mL AMH cutoff for PCOM determination in an independent population-based cohort [63]. This large, multicenter, noninterventional study has completed enrollment of 1,803 women and will provide additional evidence for the use of the Elecsys AMH Plus immunoassay as an alternative to transvaginal ultrasound for PCOM diagnosis [63].
AMH in PCOS Pathophysiology and Diagnostic Applications
Understanding the biological role of AMH in PCOS pathophysiology provides context for its utility as a diagnostic biomarker and reveals potential therapeutic targets.
The diagram above illustrates the key pathophysiological mechanisms of AMH in PCOS. AMH contributes to PCOS pathogenesis through multiple interconnected pathways. It exacerbates follicular arrest by inhibiting follicular sensitivity to FSH via TGF-β/Smad-mediated aromatase suppression in granulosa cells, thereby reducing estrogen synthesis [64]. Simultaneously, AMH synergizes with hyperandrogenism through an "AMH-LH positive feedback loop" in hypothalamic GnRH neurons, amplifying neuroendocrine dysfunction and increasing LH pulse frequency [64]. These mechanisms collectively promote the accumulation of small antral follicles that characterizes PCOM and contributes to the chronic anovulation typical of PCOS.
Beyond its role in follicular dynamics, research has identified epigenetic modifications in ovarian granulosa cells, such as AMHR2 promoter hypomethylation, that affect gene expression and cellular function in PCOS patients [64]. Environmental factors also modulate AMH expression, with studies showing that exposure to PM2.5 air pollution can reduce AMH levels by 10% for every IQR increase in concentration, with more pronounced effects in younger women [64]. These findings underscore the complex interplay between genetic predisposition, environmental factors, and endocrine pathways in PCOS pathogenesis.
The 2023 International PCOS Guideline has endorsed AMH as a surrogate for PCOM diagnosis via ultrasound, with specific recommendations for ethnicity-specific thresholds due to significant racial disparities [64]. For instance, Japan's 2024 diagnostic criteria (JSOG2024) have recalibrated AMH cutoffs for low-obesity phenotypes, highlighting the necessity of population-specific adaptations [64]. Multiethnic studies reveal striking variations, with Samoan women exhibiting paradoxically high AMH levels independent of BMI, while East Asian women require 15-20% lower thresholds than Europeans to avoid overdiagnosis [64].
Research Reagent Solutions and Essential Materials
For researchers designing studies to evaluate AMH assays or investigate AMH in PCOS pathophysiology, the following key reagents and materials are essential:
Table 4: Essential Research Reagents and Materials
| Reagent/Material | Function/Application | Example Specifications |
|---|---|---|
| AMH Gen II ELISA Kit | Manual quantification of AMH levels | Includes calibrators, controls, assay buffer; LOQ: 3.0 pmol/L [5] |
| Elecsys Cobas AMH Reagent | Automated AMH quantification on Cobas platforms | Measuring interval: 0.07-164.00 pmol/L; LOD: 0.07 pmol/L [8] |
| PreciControl AMH | Quality control for AMH assays | Two-level control (e.g., 4.29 pmol/L and 34.71 pmol/L) [8] |
| Cobas e602 Analyzer | Automated immunoassay testing platform | Compatible with Elecsys AMH assay [8] |
| Anti-AMH Antibodies (Monoclonal) | Enhanced assay specificity and reproducibility | Used in automated platforms for superior performance [65] |
The selection between monoclonal and polyclonal antibody-based assays represents a critical consideration for researchers. Monoclonal antibodies offer higher specificity and reproducibility, leading to better assay performance, while polyclonal antibodies may detect a broader range of AMH isoforms despite their lower specificity [65]. The evolution from manual ELISA methods to automated immunoassays has significantly improved precision, with the Access AMH assay (Beckman Coulter) demonstrating total imprecision of 2.4-5.2% and excellent linearity up to 24 ng/mL [6].
For specialized applications in PCOS research, the Elecsys AMH Plus immunoassay has been specifically validated for PCOM diagnosis with a defined cutoff of 3.2 ng/mL (23 pmol/L) [62]. Ongoing validation studies, such as the HARMONIA protocol, are further establishing the performance characteristics of this assay in diverse populations [63]. When designing clinical studies, researchers should incorporate appropriate reference standards based on population-specific characteristics, as significant ethnic variations in AMH levels have been documented, requiring tailored diagnostic thresholds [8] [64].
The methodological evolution from manual AMH Gen II ELISA to automated Elecsys Cobas AMH assays represents significant advancement in reproductive endocrine diagnostics. The Elecsys system demonstrates superior analytical performance with lower limits of detection, reduced imprecision, and broader measuring ranges. The establishment of a validated AMH cutoff of 3.2 ng/mL (23 pmol/L) for PCOM diagnosis using the Elecsys platform provides clinicians with a standardized, reproducible alternative to ultrasound-based follicle counting, though attention to age-specific and ethnicity-specific variations remains crucial. For researchers and drug development professionals, these comparative data inform both assay selection for clinical studies and future directions for assay harmonization. The ongoing validation of population-specific thresholds and the elucidation of AMH's role in PCOS pathophysiology will continue to enhance the precision and clinical utility of AMH measurement in reproductive medicine.
The accurate measurement of Anti-Müllerian Hormone (AMH) is fundamental to assessing ovarian reserve in reproductive medicine and clinical research. The transition from manual enzyme-linked immunosorbent assays (ELISAs) to fully automated platforms represents a significant advancement in hormonal quantification, yet it introduces critical challenges in assay agreement and calibration. This comparison guide objectively evaluates the performance characteristics of two prominent AMH assays: the established AMH Gen II ELISA (Beckman Coulter) and the automated Elecsys Cobas AMH assay (Roche Diagnostics). For researchers and drug development professionals, understanding the methodological disparities and their impact on result interpretation is essential for ensuring data consistency, facilitating cross-study comparisons, and making informed decisions in both clinical and research settings. This analysis synthesizes experimental data to provide a clear framework for assay selection and result interpretation within the broader context of AMH research.
Methodological Comparison and Analytical Performance
The fundamental differences between the AMH Gen II ELISA and the Elecsys Cobas AMH assay lie in their automation, design, and technical performance characteristics.
Assay Principles and Workflows
The AMH Gen II ELISA is a manual microplate-based assay. The protocol requires technicians to prepare a pre-mixture of clinical samples, calibrators, and controls in an assay buffer before loading them onto the plate. The assay employs a standard manual technique, with samples typically analyzed in duplicate. When duplicate results differ by more than 15%, the analysis must be repeated, adding hands-on time and potential for operator-induced variability [5].
In contrast, the Elecsys Cobas AMH assay is a fully automated electrochemiluminescence immunoassay (ECLIA) performed on Cobas immunoassay analyzers (e.g., the Cobas e601 platform). It uses Roche’s standard protocol, which requires minimal manual intervention. The process is standardized, with the analyzer handling all incubation, washing, and detection steps, thereby reducing operational variability [5] [8].
The diagram below illustrates the core technological differences in the detection methods of the two assays.
Key Performance Parameters
Direct comparative studies reveal significant differences in the analytical performance of the two assays, which are summarized in the table below.
Table 1: Analytical Performance Comparison of AMH Gen II ELISA vs. Elecsys Cobas AMH Assay
| Performance Parameter | AMH Gen II ELISA | Elecsys Cobas AMH | Implications for Research |
|---|---|---|---|
| Analytical Variability (CV%)(Control Level: ~7 pmol/L) | 10.3% [5] | 2.8% [5] | Lower Cobas variability enhances measurement precision and reliability. |
| Limit of Quantitation (LOQ) | 3.0 pmol/L [5] | 0.5 pmol/L [5] | Cobas provides more reliable data for samples with very low AMH levels. |
| Measuring Range(without dilution) | 3–70 pmol/L [5] | 0.5–160 pmol/L [5] [8] | Cobas offers a wider dynamic range, reducing need for sample re-runs. |
| Bias (Relative to ELISA) | Reference | -32% [5] | Direct numerical values are not interchangeable between platforms. |
| Sample Throughput | Manual, lower throughput | Fully automated, high throughput | Cobas is more efficient for processing large sample batches. |
The data show that the Elecsys Cobas assay demonstrates superior precision, with an analytical variability of 2.8-3.3% across control levels, compared to 5.5-10.3% for the AMH Gen II ELISA [5]. This superior precision allows the Elecsys assay to meet optimal performance goals (CVAnalytical < 0.25*CVWithin Biological Variation) across its entire measuring range, whereas the Gen II ELISA only meets this goal at the high end of its range [5]. The dramatically lower LOQ of the Elecsys Cobas assay (0.5 pmol/L vs. 3.0 pmol/L) is particularly critical for studying populations with diminished ovarian reserve, where accurate quantification of low hormone levels is essential [5].
Experimental Data and Correlation Studies
Protocol for Method Comparison Experiments
A standard protocol for directly comparing AMH assays involves a prospective sample collection from a well-defined patient cohort. In one such study [5]:
- Participants: 23 women (median age 36 years) undergoing laparoscopic sterilization.
- Sample Collection: Blood samples were collected preoperatively and at multiple post-operative time points (1 week, 1, 3, and 6 months). Serum was isolated within 4 hours and stored at -80°C until batch analysis.
- Analysis: A total of 112 serum samples were analyzed using both the AMH Gen II ELISA and the Elecsys Cobas AMH methods by skilled technicians in an ISO 15189 accredited laboratory.
- Statistical Analysis: Passing-Bablok regression and Bland-Altman plots were used to assess correlation and systematic bias between the two methods. Spearman's correlation was used to evaluate the relationship between AMH levels and antral follicle count (AFC).
Findings on Assay Correlation and Clinical Validity
Table 2: Correlation and Clinical Agreement from Comparative Studies
| Study Focus | Key Finding | Research Context Interpretation |
|---|---|---|
| Method Correlation | A consistent bias of approximately 32% was observed, with Elecsys values lower than Gen II ELISA values [5]. | A fixed conversion factor cannot be applied; assay-specific reference ranges are mandatory. |
| Correlation with AFC | Both assays showed strong, similar correlation with AFC (e.g., Baseline: Cobas R=0.83, ELISA R=0.86) [5]. | Both methods are biologically valid markers of ovarian follicular status. |
| Inter-assay Correlation | Strong correlations (0.81-0.94) are present over the total AMH range, but this correlation is level-dependent and can be only moderate in mid-ranges [9]. | Correlation is not equivalent to interchangeability, especially for specific subpopulations. |
| Prediction of Oocyte Yield | Automated assays show good correlation with oocyte yield after controlled ovarian stimulation [20]. | Automated platforms are clinically useful for predicting response in IVF cycles. |
The 32% bias observed between the methods underscores that the assays are not numerically equivalent [5]. The Bland-Altman plot from the study indicated a tendency for this bias to increase at higher AMH concentrations [5]. This is a critical consideration when studying patient populations with high AMH levels, such as those with Polycystic Ovary Syndrome (PCOS). A large-scale study of 1,660 PCOS patients confirmed that while strong inter-assay correlations exist across the total range of AMH, the correlation can become only moderate in specific mid-range segments (2.80-7.04 ng/mL) [9].
The Researcher's Toolkit: Essential Reagents and Materials
Successful execution of AMH quantification and interpretation requires an understanding of the key reagents and materials involved. The following table details these critical components.
Table 3: Essential Research Reagent Solutions for AMH Quantification
| Reagent/Material | Function in Assay | Technical Notes for Researchers |
|---|---|---|
| AMH Calibrators | Establishes the standard curve for quantitative analysis. | Not interchangeable between platforms. Calibrator traceability differs between Gen II and Elecsys systems. |
| Capture & Detection Antibodies | Forms the immunocomplex for AMH detection. | Both Gen II and Elecsys use antibodies against the same regions (AMHN,229 and AMHC) [9]. The picoAMH assay (Ansh Labs) uses a different configuration [9]. |
| Quality Control (QC) Sera | Monitors daily assay performance and precision. | Both commercial QC materials and pooled human serum are used. QC rules must be established for each platform independently. |
| Signal Detection Reagents | Generates the measurable signal (colorimetric or ECL). | Stability and lot-to-lot consistency of these reagents directly impact the assay's limit of detection and precision [6]. |
| Sample Collection Tubes | For serum/plasma separation and storage. | Samples are stable at room temperature for up to 48 hours, at 2-8°C for 7 days, and at -20°C for extended periods (up to 15 months) with no significant impact on measured AMH in automated assays [6]. |
The methodological differences between the AMH Gen II ELISA and Elecsys Cobas assays have profound implications for data interpretation and the establishment of clinical and research thresholds.
The Critical Need for Assay-Specific Reference Intervals
The consistent negative bias of the Elecsys assay means that numerical values from different platforms cannot be used interchangeably. Applying reference intervals or clinical decision limits from one assay to results generated by another will lead to misclassification. This is exemplified by the establishment of population-specific reference intervals for the Elecsys platform. For instance, a study of healthy Korean women established the following Elecsys-specific reference intervals [8]:
- Ages 19-24: 7.93 – 81.21 pmol/L
- Ages 35-39: 0.79 – 23.18 pmol/L
- Ages 50-54: 0.07 – 3.86 pmol/L These intervals provide a crucial framework for interpreting Elecsys results but would not be applicable to data generated with the Gen II ELISA. The diagram below outlines the decision-making process for interpreting results based on the assay used.
Implications for Multi-Center and Longitudinal Research
The lack of harmonization between AMH assays presents significant challenges for study design:
- Multi-Center Trials: If different sites use different assay platforms, data pooling becomes problematic. The preferred approach is to use a single, centralized laboratory with one standardized platform.
- Longitudinal Studies: Switching assay platforms mid-study can introduce a systematic shift in the data that is indistinguishable from a true biological change. If a transition is necessary, a method comparison study with a sufficient number of samples should be conducted to understand the bias.
- Meta-Analyses: Researchers combining data from publications must account for the assay type used in each primary study. The observed 32% bias indicates that treating all AMH values as equivalent would severely confound the results.
The comparison between the AMH Gen II ELISA and the Elecsys Cobas AMH assays reveals a clear trajectory toward automated platforms offering superior analytical performance, including enhanced precision, wider measuring ranges, and lower limits of quantitation. However, the absence of harmonization, evidenced by a consistent ~32% bias, means these assays are not numerically interchangeable. For the research community, this underscores a non-negotiable requirement: the use of assay-specific reference intervals and clinical thresholds is critical for valid data interpretation. Future efforts must focus on the international standardization of AMH measurement to ensure that data, regardless of its source, can be reliably compared and aggregated to advance our understanding of ovarian biology and improve patient care outcomes.
The Emergence of Novel and Ultra-Sensitive AMH Detection Platforms
Anti-Müllerian Hormone (AMH) has emerged as a critical biomarker in reproductive medicine, primarily for assessing ovarian reserve, predicting response to ovarian stimulation in IVF, and diagnosing conditions like polycystic ovary syndrome (PCOS) and primary ovarian insufficiency (POI). The accuracy and clinical utility of AMH measurement are fundamentally dependent on the detection platform used. For years, the manual AMH Gen II ELISA was widely considered the standard. However, recent years have witnessed a significant shift with the introduction of fully automated immunoassays, such as the Elecsys Cobas AMH assay, and the development of ultra-sensitive tests designed to measure AMH in challenging patient populations. This evolution addresses key limitations of earlier methods, including analytical variability, limited sensitivity, and inter-assay discrepancies, which can impact critical clinical decisions. This guide provides an objective, data-driven comparison of these emerging platforms against established methods, framing the discussion within the broader research context of AMH Gen II ELISA versus Elecsys Cobas AMH assay studies.
Performance Comparison of Major AMH Assays
Extensive comparative studies have quantified the performance differences between the newer automated platforms and the established manual ELISA method. The data reveal a consistent pattern of improved precision and reliability with automated systems.
Table 1: Analytical Performance Comparison of Key AMH Assays
| Assay | Total Analytical Variability (CV%) | Limit of Quantitation (LOQ) | Measuring Range (without dilution) | Bias vs. Gen II ELISA |
|---|---|---|---|---|
| AMH Gen II ELISA (Manual) | 5.5% - 10.3% [16] | 3.0 pmol/L [16] | 3–70 pmol/L [16] | (Reference) |
| Elecsys Cobas AMH (Automated) | 2.8% - 3.3% [16] | 0.5 pmol/L [16] | 0.5–160 pmol/L [16] | Approximately 32% lower [16] |
| Access AMH (Automated) | 2.4% - 5.2% [6] | 0.010 ng/mL (~0.07 pmol/L) [6] | Up to 24 ng/mL [2] | Highly correlated (R² = 0.98-0.99), minimal bias [2] |
| Lumipulse G AMH (Automated) | Within-lab: 1.6% - 2.5% [66] | Not specified | 0.044 to 22.42 ng/ml (linearity) [66] | ~17.8% bias vs. Elecsys AMH Plus [66] |
A pivotal 2015 study directly comparing the Gen II ELISA and Elecsys Cobas methods found a strong correlation but a consistent negative bias of approximately 32% for the Elecsys assay [16]. This means results from these two methods are not interchangeable, and clinicians must use assay-specific reference ranges. Furthermore, the Elecsys method demonstrated superior precision (CV of 2.8-3.3%) compared to the Gen II ELISA (CV of 5.5-10.3%), particularly at low AMH concentrations [16]. When evaluated against optimal performance goals for biochemical assays, the Elecsys Cobas method met these standards across its entire measuring range, while the Gen II ELISA only achieved optimal performance at the high end of its range [16].
The Beckman Coulter Access AMH assay, another automated platform, also shows excellent correlation with the Gen II ELISA (R² = 0.9822) and demonstrates high precision with a total imprecision of 2.4% to 5.2% [2] [6]. Its low limit of quantitation (0.010 ng/mL) makes it highly sensitive for detecting very low AMH levels [6].
Experimental Protocols for Key Comparative Studies
The performance data presented above are derived from rigorous experimental designs. Understanding these methodologies is crucial for interpreting results and assessing the validity of the comparisons.
Protocol: Direct Comparison of Gen II ELISA vs. Elecsys Cobas
A prospective observational study provided a direct clinical comparison of the two core platforms [16] [5].
- Sample Cohort: 23 women (median age 36) seeking laparoscopic sterilization. Patients with endocrine disease, PCOS, hormonal contraception, or suspected malignancy were excluded.
- Sample Collection & Handling: A total of 112 serum samples were collected pre-operatively and at multiple post-operative time points (1 week, 1, 3, and 6 months). Serum was isolated within 4 hours of sampling and stored at -80°C until batch analysis.
- Assay Methodology:
- AMH Gen II ELISA (Beckman Coulter): A manual technique performed according to the standard protocol. Samples were analyzed in duplicate, with repeats if results differed by more than 15%. The acceptance CV for internal controls was <11–14%.
- Elecsys Cobas AMH (Roche): Analyzed on a Cobas 6000 e601 platform using the manufacturer's standard automated protocol. The acceptance CV for internal controls was <5%.
- Statistical Analysis: Correlation with antral follicle count (AFC) was determined using Spearman's test. Method comparison was performed using Passing-Bablok and Bland-Altman plots.
Protocol: Evaluation of Ultra-Sensitive Assays in POI
A 2025 retrospective study demonstrated the clinical application of a highly sensitive AMH assay in a challenging patient population [23].
- Sample Cohort: 165 patients with Primary Ovarian Insufficiency (POI) undergoing 504 long controlled ovarian stimulation cycles. POI was defined by last menstruation before age 40, serum FSH >25 mIU/mL, and E2 <20 pg/mL.
- Intervention: AMH levels were measured three weeks after stimulation initiation using the pico AMH ELISA (Ansh Labs), an ultra-sensitive assay with a limit of detection (LoD) of 1.3 pg/mL.
- Objective: To determine if these minimal AMH levels could predict subsequent follicular development detected by ultrasound.
- Outcome Analysis: The predictive value of the 3-week AMH level was evaluated using receiver operating characteristic (ROC) curve analysis.
Table 2: Key Research Reagent Solutions for AMH Detection
| Reagent / Assay Kit | Manufacturer | Key Function & Characteristics |
|---|---|---|
| AMH Gen II ELISA | Beckman Coulter | Manual assay; measures proAMH and AMHN,C complex; uses monoclonal antibodies F2B/12H (capture) and F2B/7A (detector) [66]. |
| Elecsys Cobas AMH / AMH Plus Assay | Roche Diagnostics | Automated, electrochemiluminescence (ECLIA) technology; uses the same antibodies as the Gen II assay (F2B/12H and F2B/7A) [66]. |
| Access AMH Immunoassay | Beckman Coulter | Fully automated chemiluminescent assay on Access/DxI platforms; high precision and correlation with Gen II ELISA [2] [6]. |
| pico AMH ELISA | Ansh Labs | Ultra-sensitive manual assay; LoD of 1.3 pg/mL; capture antibody targets C-terminal region, detector targets AMHM region [23] [9]. |
| Lumipulse G AMH Assay | Fujirebio | Fully automated, chemiluminescence enzyme immunoassay (CLEIA) on compact analyzer; uses monoclonal antibodies A7 and A12 [66]. |
Signaling Pathways and Experimental Workflow
The accurate measurement of AMH is complicated by its complex molecular biology. AMH is secreted as a pro-hormone (proAMH) that undergoes proteolytic cleavage, resulting in multiple circulating isoforms. The specific isoforms detected by an immunoassay depend on the antibodies used, which is a primary source of inter-assay variability.
Different assays employ different antibody pairs targeting specific regions of the AMH molecule. The Gen II, Elecsys, and Access assays use a capture antibody against the N-terminal pro-region (AMHN) and a detector antibody against the C-terminal mature region (AMHC), measuring the proAMH and AMHN,C complex [66] [9]. In contrast, the picoAMH assay uses a C-terminal capture antibody and a detector antibody against the mid-region (AMHM), leading to potential differences in measured values [9].
The Expanding Landscape of Novel AMH Platforms
Beyond the core comparison between Gen II ELISA and Elecsys, the landscape of AMH detection continues to evolve with the introduction of new platforms.
- Ultra-Sensitive Detection: The pico AMH ELISA (Ansh Labs) represents a significant advancement for measuring very low AMH levels. With a LoD of 1.3 pg/mL, it is substantially more sensitive than standard clinical assays (e.g., Access: 0.02 ng/mL; Gen II: 0.08 ng/mL) [23]. This sensitivity is critical for applications like monitoring POI patients, where a 2025 study found that AMH levels as low as 2.45 pg/mL could predict follicular growth with high accuracy (AUC: 0.957) [23].
- New Automated Platforms: Recent evaluations include the Siemens Healthineers Atellica IM AMH assay. A 2025 study found it correlates strongly with the Beckman DxI assay (slope: 1.07, R²: 0.9881) but shows a minimal positive bias [67]. In the same study, the Roche Elecsys assay exhibited a negative bias versus Beckman DxI (slope: 0.74), reinforcing that systematic differences persist among automated methods [67].
- Inter-Assay Correlation is Level-Dependent: A large 2022 study in PCOS patients revealed that the correlation between different AMH assays (Gen II, picoAMH, Elecsys) is not constant but depends on the AMH concentration range. While correlations were strong over the total range (0.81-0.94), they were only moderate in the mid-range (2.80-7.04 ng/mL), highlighting the challenge of defining universal conversion factors [9].
The emergence of novel and ultra-sensitive AMH detection platforms marks a significant step forward in reproductive endocrinology. The shift from manual ELISA to automated immunoassays like the Elecsys Cobas and Access AMH has provided the improved precision, reliability, and throughput necessary for a high-volume clinical biomarker. However, the persistence of significant and non-constant biases between assays underscores a critical challenge: the lack of an international standard for AMH measurement. This necessitates the use of assay-specific reference intervals and cautions against the direct comparison of results obtained from different methods. The development of ultra-sensitive assays opens new clinical frontiers, particularly in the management of POI, where predicting ovarian activity was previously nearly impossible. For researchers and clinicians, the key takeaway is that an understanding of the technical principles, performance characteristics, and limitations of each platform is fundamental to the accurate interpretation of AMH values and their application in both clinical practice and research.
Conclusion
The comparative analysis unequivocally establishes the Elecsys Cobas AMH assay as a technologically advanced successor to the AMH Gen II ELISA, offering enhanced automation, superior precision, and a more clinically useful lower limit of quantitation. However, the presence of a significant and consistent bias between the assays underscores a critical lack of harmonization, necessitating assay-specific reference intervals and clinical cut-offs. For researchers and drug developers, these findings highlight that assay choice is not merely a technical decision but one that directly impacts clinical data interpretation and patient management. Future directions must prioritize the development of an international standard for AMH to enable result harmonization across platforms. Furthermore, the exploration of even more sensitive assays and novel, rapid detection technologies like hydrogel-based radio frequency immunosensors promises to expand the clinical utility of AMH into new frontiers of diagnostics and personalized medicine.
References
- 1. Anti‐Müllerian hormone as a marker of ovarian reserve [https://pmc.ncbi.nlm.nih.gov/articles/PMC5715856/]
- 2. Assessment of the Access AMH assay as an automated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4754992/]
- 3. Markers of ovarian reserve as predictors of future fertility [https://www.sciencedirect.com/science/article/pii/S0015028222019641]
- 4. Article Prediction of ovarian response using the automated ... [https://www.sciencedirect.com/science/article/pii/S1472648322007672]
- 5. Comparison of two different methods for measuring anti ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4580367/]
- 6. Performance characteristics of the Access AMH assay for ... [https://www.sciencedirect.com/science/article/pii/S0009912016301849]
- 7. Elecsys® AMH (Anti-Müllerian Hormone) [https://diagnostics.roche.com/us/en/products/lab/elecsys-amh-cps-000432.html]
- 8. Age Group-specific Reference Intervals for the Elecsys Anti ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9277038/]
- 9. Comparison of 3 Different AMH Assays With AMH Levels ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9387710/]
- 10. Multicenter analytical performance evaluation of a fully ... [https://pubmed.ncbi.nlm.nih.gov/26500002/]
- 11. Challenges in Measuring AMH in the Clinical Setting [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2021.691432/full]
- 12. A multicentre evaluation of the Elecsys® anti-Müllerian ... [https://www.sciencedirect.com/science/article/pii/S1472648319300069]
- 13. Optimization and validation of an ELISA assay for the ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0275927]
- 14. Comparison of Conventional ELISA with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3075593/]
- 15. Development and laboratory validation of an ... [https://pubmed.ncbi.nlm.nih.gov/40268228/]
- 16. Comparison of two different methods for measuring anti ... [https://rbej.biomedcentral.com/articles/10.1186/s12958-015-0101-5]
- 17. Comparability of AMH levels among commercially ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4334563/]
- 18. integrating high-specific assays for anti-mullerian hormone ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12515637/]
- 19. Development and validation of a multiplex ... [https://www.nature.com/articles/s41598-023-39397-8]
- 20. Performance of anti‐Müllerian hormone (AMH) levels ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6613014/]
- 21. A Fully Automated Assay for Serum Antimüllerian Hormone ... [https://www.preprints.org/manuscript/202507.2468]
- 22. Measurement of serum anti-Müllerian hormone by revised ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10258520/]
- 23. A highly sensitive Anti-Müllerian hormone test as ... [https://www.nature.com/articles/s41598-025-98808-0]
- 24. Development of a second generation anti-Müllerian ... [https://www.sciencedirect.com/science/article/abs/pii/S0022175910002383]
- 25. Introducing the NEW Demeditec AMH ELISA [https://www.abacusdx.com/biochemistry/introducing-new-demeditec-amh-elisa/]
- 26. New automated antimüllerian hormone assays are more ... [https://www.sciencedirect.com/science/article/pii/S0015028216627711]
- 27. Predictive value of AMH in late reproductive age [https://pmc.ncbi.nlm.nih.gov/articles/PMC12217941/]
- 28. Anti-Müllerian hormone as a biomarker of ovarian function ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1640583/full]
- 29. Establishment of a homogeneous immunoassay-light ... [https://www.sciencedirect.com/science/article/abs/pii/S0022175921001046]
- 30. cobas® e 801 analytical unit [https://diagnostics.roche.com/us/en/products/instruments/cobas-e-801-ins-2202.html]
- 31. cobas® e 411 analyzer [https://diagnostics.roche.com/us/en/products/instruments/cobas-e-411-ins-502.html]
- 32. of two different methods for measuring anti-mullerian... Comparison [https://pubmed.ncbi.nlm.nih.gov/26394617/]
- 33. AMH ELISA [https://www.anshlabs.com/product/amh-elisa/]
- 34. Performance characteristics of the Mindray ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8059718/]
- 35. Analytical Characteristics and Clinical Performance of Anti ... [https://www.mdpi.com/2673-396X/5/4/37]
- 36. Age-specific Reference Intervals for Anti-Müllerian Hormone [https://pmc.ncbi.nlm.nih.gov/articles/PMC9277044/]
- 37. Age-specific Reference Intervals for Anti-Müllerian Hormone [https://www.annlabmed.org/journal/view.html?doi=10.3343/alm.2022.42.6.617]
- 38. Evaluation of the Elecsys® anti-Müllerian hormone assay ... [https://www.sciencedirect.com/science/article/pii/S0301211519300909]
- 39. A Prospective Cohort Study With AMH Tested on Three ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2018.00603/full]
- 40. Variation in the Measurement of Anti-Müllerian Hormone [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2021.719029/full]
- 41. Prospective validation of anti-Müllerian hormone cutoff to ... [https://www.sciencedirect.com/science/article/pii/S0015028225004352]
- 42. Human Anti-Mullerian Hormone (AMH) ELISA Kit [https://www.reddotbiotech.com/elisa-kits/human-anti-mullerian-hormone-amh-elisa-kit-22330.html?srsltid=AfmBOooIMX4KSVEVpvxHEpnd74KUYydyu17DbTv35D75nDfyeoE0bJGg]
- 43. ELISA Kit for Anti-Mullerian Hormone (AMH) | CEA228Hu [https://www.cloud-clone.com/products/CEA228Hu.html]
- 44. Two new automated, compared with two enzyme-linked ... [https://www.sciencedirect.com/science/article/pii/S0015028215004513]
- 45. Cow AMH ELISA kit [https://www.antibodies-online.com/kit/1568317/Anti-Mullerian+Hormone+AMH+ELISA+Kit/?srsltid=AfmBOoqpSkAJdu7zDBhp4mI47z9CSgRdaHAQsrh0Vpd8Gq_D0g1GCvgp]
- 46. Variation in the Measurement of Anti-Müllerian Hormone [https://pmc.ncbi.nlm.nih.gov/articles/PMC8446602/]
- 47. Challenges in Measuring AMH in the Clinical Setting - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8183164/]
- 48. Hormone Immunoassay Interference: A 2021 Update - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8368230/]
- 49. Common errors in manual ELISA assays and how ... [https://automata.tech/case-studies/common-errors-in-manual-elisa-assays-and-how-automation-can-help/]
- 50. A Practical Guide to Immunoassay Method Validation - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4541289/]
- 51. Inter-cycle variability of anti-Müllerian hormone [https://ovarianresearch.biomedcentral.com/articles/10.1186/s13048-024-01517-x]
- 52. A highly sensitive Anti-Müllerian hormone test as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12015210/]
- 53. Can we predict menopause and premature ovarian ... [https://www.sciencedirect.com/science/article/pii/S0015028224001171]
- 54. Correlation between serum AMH, AFC, AND histologically ... [https://ovarianresearch.biomedcentral.com/articles/10.1186/s13048-025-01807-y]
- 55. Ovarian volume and antral follicle count for the prediction of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1847823/]
- 56. Antral Follicle Counts, Testing Ovarian Reserve Predicts ... [https://www.advancedfertility.com/infertility-testing/antral-follicle-counts]
- 57. Guide to Antral Follicle Count [https://dallasivf.com/post/guide-to-antral-follicle-count]
- 58. Guide to Antral Follicle Count [https://utahfertility.com/post/guide-to-antral-follicle-count]
- 59. Body size affects measures of ovarian reserve in late ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2821936/]
- 60. Antral follicle count in clinical practice: building the bridge ... [https://www.sciencedirect.com/science/article/pii/S0015028210028566#!]
- 61. Evaluation of a new automated immunoassay for the ... [https://www.sciencedirect.com/science/article/pii/S2352551721000202]
- 62. Antimüllerian hormone to determine polycystic ovarian ... [https://www.sciencedirect.com/science/article/pii/S0015028221004477]
- 63. Validation of an Anti-Müllerian Hormone Cutoff for ... [https://pubmed.ncbi.nlm.nih.gov/38319689/]
- 64. Bibliometric analysis of the research on anti-Müllerian ... [https://www.frontiersin.org/journals/reproductive-health/articles/10.3389/frph.2025.1519249/full]
- 65. Anti-mullerian Hormone (AMH) Strategic Insights [https://www.archivemarketresearch.com/reports/anti-mullerian-hormone-amh-344555]
- 66. Evaluation of a new automated immunoassay for the quantification of anti-Müllerian hormone - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8145750/]
- 67. Evaluation of Siemens Atellica AMH assay and comparison with Roche, Beckman and Ansh Labs - PubMed [https://pubmed.ncbi.nlm.nih.gov/39675655/]